10 Jun 2026
// GLOBENEWSWIRE
05 Jun 2026
// BUSINESSWIRE
04 Jun 2026
// GLOBENEWSWIRE
Latest Content by PharmaCompass

Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
About
PEGS Boston SummitPEGS Boston Summit
Industry Trade Show
Not Confirmed
11 May-15 November, 2026
Industry Trade Show
Not Confirmed
09-11 June, 2026
World Vape ShowWorld Vape Show
Industry Trade Show
Not Confirmed
09-11 June, 2026
CONTACT DETAILS




Events
Webinars & Exhibitions
PEGS Boston SummitPEGS Boston Summit
Industry Trade Show
Not Confirmed
11 May-15 November, 2026
Industry Trade Show
Not Confirmed
09-11 June, 2026
World Vape ShowWorld Vape Show
Industry Trade Show
Not Confirmed
09-11 June, 2026
CORPORATE CONTENT #SupplierSpotlight
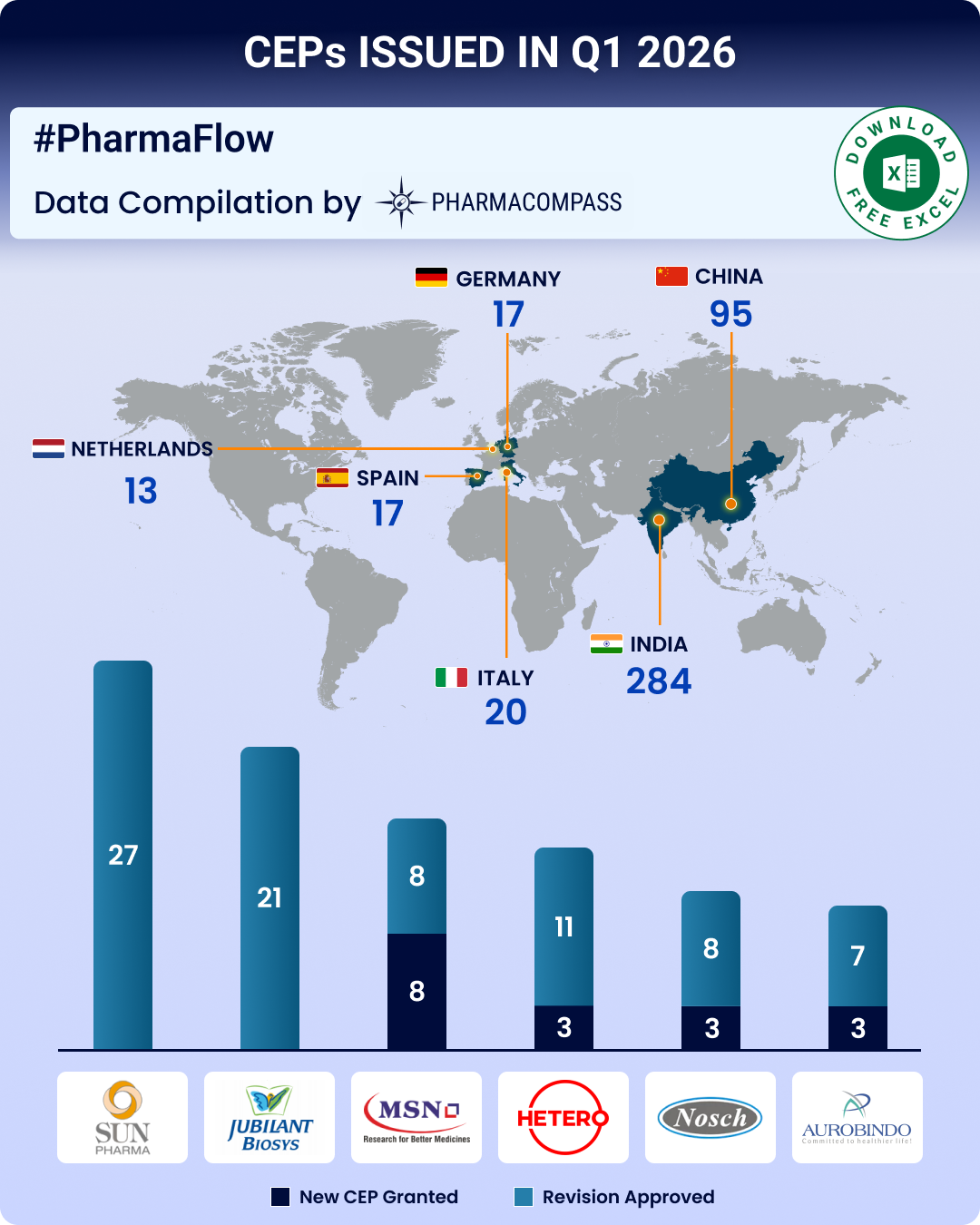
https://www.pharmacompass.com/radio-compass-blog/cep-q1-2026-update-cep-2-0-edqm-s-new-guidelines-strengthen-ecosystem-indian-firms-top-list-of-ceps-issued
https://www.pharmacompass.com/radio-compass-blog/fda-approvals-drop-8-in-2025-with-fewer-blockbusters-brinsupri-rhapsido-make-it-to-first-in-class-list
https://www.pharmacompass.com/radio-compass-blog/top-news-of-2025-drugmakers-invest-in-us-capacities-agree-to-lower-medicaid-prices-pfizer-buys-obesity-focused-biotech-metsera
https://www.pharmacompass.com/radio-compass-blog/fda-approvals-drop-24-in-h1-2025-gsk-s-uti-med-vertex-s-non-opioid-painkiller-lead-pack-of-first-in-class-meds
https://www.pharmacompass.com/radio-compass-blog/j-j-s-intra-cellular-buyout-bms-oncology-gambit-sanofi-s-blueprint-acquisition-drive-mega-deals-in-h1-2025
https://www.pharmacompass.com/radio-compass-blog/us-drug-shortages-reduce-16-yoy-in-q1-2025-cns-drugs-antimicrobials-face-highest-scarcities
https://www.pharmacompass.com/radio-compass-blog/top-pharma-companies-drugs-in-2024-merck-s-keytruda-maintains-top-spot-as-novo-s-semaglutide-nips-at-its-heels
https://www.pharmacompass.com/radio-compass-blog/top-first-in-class-drug-candidates-of-2025-ionis-donidalorsen-sanofi-s-fitusiran-cytokinetics-aficamten-await-fda-approval
https://www.pharmacompass.com/radio-compass-blog/fda-okays-50-new-drugs-in-2024-bms-cobenfy-lilly-s-kisunla-lead-pack-of-breakthrough-therapies
https://www.pharmacompass.com/radio-compass-blog/bms-j-j-bayer-lead-25-000-pharma-layoffs-in-2024-amylyx-fibrogen-kronos-bio-hit-by-trial-failures-cash-crunch
https://www.pharmacompass.com/radio-compass-blog/fda-s-landmark-approvals-of-bms-schizo-med-madrigal-s-mash-drug-us-16-5-bn-catalent-buyout-make-it-to-top-10-news-of-2024

10 Jun 2026
// GLOBENEWSWIRE
https://www.sanofi.com/en/media-room/press-releases/2026/2026-06-08-05-00-00-3307781

05 Jun 2026
// BUSINESSWIRE
https://www.businesswire.com/news/home/20260605704506/en/Owkin-to-Build-AI-Agents-as-Part-of-a-Multi-Year-K-Pro-Collaboration-with-Sanofi

04 Jun 2026
// GLOBENEWSWIRE
https://www.globenewswire.com/news-release/2026/06/04/3306553/0/en/press-release-sanofi-launches-2026-global-employee-stock-purchase-plan.html

02 Jun 2026
// FIERCE PHARMA
https://www.fiercepharma.com/marketing/sanofi-taps-snowflake-ai-field-agent-help

29 May 2026
// PRESS RELEASE
https://www.gov.uk/government/news/rilzabrutinib-authorised-to-treat-adults-with-immune-thrombocytopenia-when-prior-treatments-have-been-insufficient#:~:text=The%20Medicines%20and%20Healthcare%20products,have%20not%20worked%20well%20enough.

29 May 2026
// EXPRESSPHARMA
https://www.expresspharma.in/sanofi-launches-accelrare-reduce-indias-rare-disease-diagnosis-challenge/
Excipients
Inspections and registrations
ABOUT THIS PAGE
Sanofi is a supplier offers 203 products (APIs, Excipients or Intermediates).
Find Irbesartan bulk with DMF, CEP, JDMF offered by Sanofi
Find Chlorpromazine Hydrochloride bulk with DMF, CEP offered by Sanofi
Find Promethazine Hydrochloride bulk with DMF, CEP offered by Sanofi
Find Zolpidem Tartrate bulk with DMF, CEP offered by Sanofi
Find Clopidogrel bulk with CEP offered by Sanofi
Find Clorazepate Dipotassium bulk with CEP offered by Sanofi
Find Trimipramine bulk with CEP offered by Sanofi
Find Zopiclone bulk with CEP offered by Sanofi
Find Acebutolol Hydrochloride bulk offered by Sanofi
Find Alfuzosin Hydrochloride bulk with DMF offered by Sanofi
Find Amiodarone Hydrochloride bulk with CEP offered by Sanofi
Find Amisulpride bulk with CEP offered by Sanofi
Find Avanafil bulk with DMF offered by Sanofi
Find Cabazitaxel bulk with DMF offered by Sanofi
Find Carbaspirin Calcium bulk with CEP offered by Sanofi
Find Ciprofibrate bulk with CEP offered by Sanofi
Find Clopidogrel bulk with DMF offered by Sanofi
Find Divalproex Sodium bulk with CEP offered by Sanofi
Find Docetaxel bulk with CEP offered by Sanofi
Find Dronedarone Hydrochloride bulk with DMF offered by Sanofi
Find Heptaminol bulk with CEP offered by Sanofi
Find Irbesartan bulk with CEP offered by Sanofi
Find Ketoprofen bulk offered by Sanofi
Find Meglumine Antimonate bulk with DMF offered by Sanofi
Find Methylprednisolone Acetate bulk with DMF offered by Sanofi
Find Polio Vaccine bulk with JDMF offered by Sanofi
Find Sodium Hyaluronate bulk with JDMF offered by Sanofi
Find Sodium Valproate bulk with CEP offered by Sanofi
Find Spiramycin bulk offered by Sanofi
Find Thyrotropin-Releasing Hormone bulk offered by Sanofi
Find Tiapride Hydrochloride bulk with CEP offered by Sanofi
Find Ticlopidine bulk with CEP offered by Sanofi
Find SAR441236 bulk with DMF offered by Sanofi
Find Defibrinated sheep blood bulk with CEP offered by Sanofi
Find Citrated Ovine Blood bulk with CEP offered by Sanofi
Find 2-Hydroxymethyl-4-3-Methoxypropoxy-3-Methylpyrid bulk offered by Sanofi
Find 5 6-Dimethoxy-1-Indanone bulk offered by Sanofi
Find 5-Hydroxy Tryptophan bulk offered by Sanofi
Find Acesulfamo bulk offered by Sanofi
Find Alendronate Sodium bulk offered by Sanofi
Find Alfuzosin Hydrochloride bulk offered by Sanofi
Find Alimemazine bulk offered by Sanofi
Find Allethrin bulk offered by Sanofi
Find Alvocidib bulk offered by Sanofi
Find Amisulpride bulk offered by Sanofi
Find Amlodipine Besylate bulk offered by Sanofi
Find Aprotinin bulk offered by Sanofi
Find Artesunate bulk offered by Sanofi
Find Articaine Hydrochloride bulk offered by Sanofi
Find Beclomethasone Dipropionate bulk offered by Sanofi
Find Betamethasone Acetate bulk offered by Sanofi
Find Betaxolol bulk offered by Sanofi
Find Buprenorphine Hydrochloride bulk offered by Sanofi
Find Calcitonin bulk offered by Sanofi
Find CAS 150975-95-4 bulk offered by Sanofi
Find CAS 50-28-2 bulk offered by Sanofi
Find CAS 62063-19-8 bulk offered by Sanofi
Find Chenodeoxycholic Acid bulk offered by Sanofi
Find Chlormezanone bulk offered by Sanofi
Find Chloroquine Phosphate bulk offered by Sanofi
Find Cholesteryl Sodium Sulfate bulk offered by Sanofi
Find Cholic Acid bulk offered by Sanofi
Find Ciramadol bulk offered by Sanofi
Find Clemastine bulk offered by Sanofi
Find Clindamycin Phosphate bulk offered by Sanofi
Find Clopidogrel bulk offered by Sanofi
Find Clotrimazole bulk offered by Sanofi
Find Codeine bulk offered by Sanofi
Find Colchicine bulk offered by Sanofi
Find Cortisone bulk offered by Sanofi
Find Dalteparin sodium bulk offered by Sanofi
Find Daunorubicin HCl bulk offered by Sanofi
Find Deserpidine bulk offered by Sanofi
Find Dextropropoxyphene bulk offered by Sanofi
Find Diatrizoate Sodium bulk offered by Sanofi
Find Dihydroergotamine Mesylate bulk offered by Sanofi
Find Dihydrotachysterol bulk offered by Sanofi
Find Disopyramide bulk offered by Sanofi
Find Dobutamine Hydrochloride bulk offered by Sanofi
Find DPPC bulk offered by Sanofi
Find Eplivanserin Hemifumarate Salt bulk offered by Sanofi
Find Estradiol Valerate bulk offered by Sanofi
Find Estrovarin bulk offered by Sanofi
Find Ethambutol Dihydrochloride bulk offered by Sanofi
Find Ethinyl Estradiol bulk offered by Sanofi
Find Ethionamide bulk offered by Sanofi
Find Fluprednidene Acetate bulk offered by Sanofi
Find Flurazepam bulk offered by Sanofi
Find Fluticasone Propionate bulk offered by Sanofi
Find Fondaparinux Sodium bulk offered by Sanofi
Find gamma-BHC bulk offered by Sanofi
Find Guaifenesin bulk offered by Sanofi
Find Hematoporphyrin Dihydrochloride bulk offered by Sanofi
Find Hematoporphyrins bulk offered by Sanofi
Find Heparin Sodium bulk offered by Sanofi
Find Hydrochlorothiazide bulk offered by Sanofi
Find Hydrocortisone bulk offered by Sanofi
Find Insulin Lispro bulk offered by Sanofi
Find Iodine bulk offered by Sanofi
Find Isoetharine bulk offered by Sanofi
Find Leuprolide Acetate bulk offered by Sanofi
Find Levonordefrin bulk offered by Sanofi
Find Levonorgestrel Oxime bulk offered by Sanofi
Find Lidocaine bulk offered by Sanofi
Find Melatonin bulk offered by Sanofi
Find Mephentermine bulk offered by Sanofi
Find Meso-2,3-Dimercaptosuccinic Acid bulk offered by Sanofi
Find Mestranol bulk offered by Sanofi
Find Methadone Hydrochloride bulk offered by Sanofi
Find Methyl Testosterone bulk offered by Sanofi
Find Methylprednisolone Hemisuccinate bulk offered by Sanofi
Find Metronidazole bulk offered by Sanofi
Find Minaprine bulk offered by Sanofi
Find Nalmefene Hydrochloride bulk offered by Sanofi
Find Nedocromil Sodium bulk offered by Sanofi
Find Neomycin Sulfate bulk offered by Sanofi
Find Norandrostenedione bulk offered by Sanofi
Find Norethisterone bulk offered by Sanofi
Find Norethisterone Acetate bulk offered by Sanofi
Find Norgestimate bulk offered by Sanofi
Find Octoxynol bulk offered by Sanofi
Find Omeprazole bulk offered by Sanofi
Find Oxybutynin Hydrochloride bulk offered by Sanofi
Find Paracetamol bulk offered by Sanofi
Find Pentamidine Isethionate bulk offered by Sanofi
Find Phospholipon Mc bulk offered by Sanofi
Find POPC bulk offered by Sanofi
Find Prochlorperazine Edisylate bulk offered by Sanofi
Find Promazine bulk offered by Sanofi
Find Propionylpromazine bulk offered by Sanofi
Find Propyphenazone bulk offered by Sanofi
Find Pyrazinamide bulk offered by Sanofi
Find R-Zacopride bulk offered by Sanofi
Find Rifampicin bulk offered by Sanofi
Find Rifamycin Sodium bulk offered by Sanofi
Find Rimonabant bulk offered by Sanofi
Find Salicylic Acid bulk offered by Sanofi
Find Saredutant Succinate bulk offered by Sanofi
Find Satavaptan Phosphate bulk offered by Sanofi
Find Sodium Cromoglicate bulk offered by Sanofi
Find Streptomycin bulk offered by Sanofi
Find Suproclone bulk offered by Sanofi
Find Suriclone bulk offered by Sanofi
Find Teicoplanin bulk offered by Sanofi
Find Testosterone bulk offered by Sanofi
Find Testosterone Cypionate bulk offered by Sanofi
Find Testosterone Enanthate bulk offered by Sanofi
Find Testosterone Propionate bulk offered by Sanofi
Find Tetrazepam bulk offered by Sanofi
Find Tiludronic Acid bulk offered by Sanofi
Find Triamcinolone bulk offered by Sanofi
Find Triamcinolone Acetonide bulk offered by Sanofi
Find Triamcinolone Diacetate bulk offered by Sanofi
Find Triamcinolone Hexacetonide bulk offered by Sanofi
Find Triamterene bulk offered by Sanofi
Find Trifluoperazine Hydrochloride bulk offered by Sanofi
Find Trimegestone bulk offered by Sanofi
Find Triprolidine Hydrochloride bulk offered by Sanofi
Find Tyropanoate bulk offered by Sanofi
Find Verapamil Hydrochloride bulk offered by Sanofi
Find Vitamin B12 bulk offered by Sanofi
Find BEFLOXATONE bulk offered by Sanofi
Find MANUFACTURING SITE, FACILITIES, PERSONNEL, AND GENERAL OPERATING PROCEDURES IN FAWDON, NEWCASTLE TYNE NE3 3TT, ENGLAND. bulk offered by Sanofi
Find PEGYLATED RECOMBINANT ANTI-PSEUDOMONAS AERUGINOSA PCRV FAB' ANTIBODY (KB001-A) DRUG SUBSTANCE bulk offered by Sanofi
Find HEMATOROPHYRINE DIHYDROCHLORIDE,PACKAGED AND TESTED IN VERTOLAYE,FRANCE bulk offered by Sanofi
Find NORTON PLASTIC S-500-304 bulk offered by Sanofi
Find MANUFACTURING SITE, FACILITIES, PERSONNEL, AND GENERAL OPERATING PROCEDURES IN LIESTAL, SWITZERLAND. bulk offered by Sanofi
Find ADENO-ASSOCIATED VIRUS (AAV) VECTOR CONTAINING THE GENE FOR HUMAN BLOOD COAGULATION FACTOR IX (COAGULIN-B) bulk offered by Sanofi
Find ELS(ELASTASE) bulk offered by Sanofi
Find MANUFACTURING FACILITIES AT NOTRE DAME DE BONDEVILLE, FRANCE bulk offered by Sanofi
Find FACILITIES LOCATED IN SISTERON, FRNACE bulk offered by Sanofi
Find 1-PALMITOYL-2-OLEOYL-SN-GLYCERO-3-PHOSPHOGLYCEROL, AMMONIUM SALT (POPG, NH4) bulk offered by Sanofi
Find MANUFACTURING SITE, FACILITIES, PERSONNEL, AND GENERAL OPERATING PROCEDURES IN LABEGE CEDEX, FRANCE bulk offered by Sanofi
Find MANUFACTURING SITE, FACILITIES, PERSONNEL, AND GENERAL OPERATING PROCEDURES IN ARAMON. bulk offered by Sanofi
Find 1-(A-HYDROXYETHYL)-2-METHYL-5-NITRO-IMIDAZOLE,A NEW TRICHOMONOCIDE bulk offered by Sanofi
Find SR 27417A bulk offered by Sanofi
Find SEMULOPARIN SODIUM bulk offered by Sanofi
Find MANUFACTURING SITE, FACILITIES, PERSONNEL, AND GENERAL OPERATING PROCEDURES IN TOULOUSE, FRANCE. bulk offered by Sanofi
Find HYDROXY ACID bulk offered by Sanofi
Find CERAMIDETRIHEXOSIDASE bulk offered by Sanofi
Find MDL 104036 bulk offered by Sanofi
Find FACILITY LOCATED IN SISTERON, FRANCE bulk offered by Sanofi
Find CONTRACT PACKAGING OPERATIONS ONLY, LOCATED IN FENTON, MO bulk offered by Sanofi
Find METHYLOXINE BROMOMETHYLOXINE METHYLOXINE N-DODECYLSULFATE bulk offered by Sanofi
Find MDL 101,731 bulk offered by Sanofi
Find MANUFACTURING SITE, FACILITIES, PERSONNEL AND GENERAL OPERATING PROCEDURES IN AMBARES, FRANCE. bulk offered by Sanofi
Find PEGYLATED RECOMBINANT ANIT-PSEUDOMONAS AERUGINOSA PCRV FAB' ANTIBODY (KB001-A) DRUG PRODUCT bulk offered by Sanofi
Find MANUFACTURING SITE, FACILITIES, PERSONNEL AND GENERAL OPERATING PROCEDURES AT COLLEGEVIEW, PA bulk offered by Sanofi
Find MANUFACTURING FACILITIES AND CONTROLS bulk offered by Sanofi
Find FACILITIES, PERSONNEL AND GENERAL OPERATING PROCEDURES AT BUDAPEST, HUNGARY bulk offered by Sanofi
Find SPIROMETHYL-BIPHENYL-NITRILE (SR47563) bulk offered by Sanofi
Find MDL 105074 bulk offered by Sanofi
Find FACILITIES UTILIZED IN MONTPELLIER, FRANCE bulk offered by Sanofi
Find MANUFACTURING SITE, FACILITIES, PERSONNEL, AND GENERAL OPERATING PROCEDURES OF 5,6-DIMETHOXY-1-INDANONE bulk offered by Sanofi
Find MANUF RESCH AND PERSONNEL FILE RSCH FACILS, EQUIPMENT & CONTROLS 11-16 bulk offered by Sanofi
Find Apomorphine Hydrochloride bulk offered by Sanofi
Find Grape Seed Extract bulk offered by Sanofi
Find Hydroxocobalamin bulk offered by Sanofi
Find Hydroxocobalamin Acetate bulk offered by Sanofi
Find Nalbuphine Hydrochloride bulk offered by Sanofi
Find Naloxone Hydrochloride bulk offered by Sanofi
Find Naltrexone Hydrochloride bulk offered by Sanofi
Find Trenbolone bulk offered by Sanofi


 Sanofi
Sanofi