
Synopsis
Synopsis
0
VMF
0
Australia
Annual Reports
NA
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Amlodipine
2. Amlodipine Maleate
3. Amlodipine Maleate (1:1)
4. Amlodipine, (+-)-isomer
5. Amlodipine, (+-)-isomer, Maleate (1:1)
6. Amlodipine, (r)-isomer
7. Amlodipine, (s)-isomer, Maleate (1:1)
8. Amlodis
9. Amlor
10. Astudal
11. Istin
12. Norvasc
1. 111470-99-6
2. Amlodipine Besilate
3. Amlodipine Benzenesulfonate
4. Antacal
5. Monopina
6. Istin
7. Amlodipine (besylate)
8. Amdepin
9. Amdipin
10. Amlodin
11. Pelmec
12. 3-ethyl 5-methyl 2-((2-aminoethoxy)methyl)-4-(2-chlorophenyl)-6-methyl-1,4-dihydropyridine-3,5-dicarboxylate Benzenesulfonate
13. Lodipressin
14. Amlor
15. Amlodipine (as Besilate)
16. Nsc-758922
17. (s)-amlodipine;levoamlodipine
18. Amlodipine Besylate (norvasc)
19. Chebi:2669
20. Cardiorex
21. Uk-48,340-26
22. 111470-99-6 (besylate)
23. 864v2q084h
24. 3,5-pyridinedicarboxylic Acid, 2-((2-aminoethoxy)methyl)-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-, 3-ethyl 5-methyl Ester, (+-)-, Monobenzenesulfonate
25. Ncgc00095835-01
26. Amlogard
27. Amlopin
28. Amlosyn
29. Astudar
30. Cordarene
31. Myodura
32. Norlopin
33. Norvask
34. Tensivask
35. Amcard
36. Norvas
37. Terloc
38. Norvasc (tn)
39. Uk-48340-26
40. (s)-amlodipine Besylate;levoamlodipine Besylate
41. Dsstox_cid_23909
42. Dsstox_rid_80087
43. Dsstox_gsid_43909
44. Benzenesulfonic Acid;3-o-ethyl 5-o-methyl 2-(2-aminoethoxymethyl)-4-(2-chlorophenyl)-6-methyl-1,4-dihydropyridine-3,5-dicarboxylate
45. Stamlo
46. Amlodipine Besylate [usan]
47. Cas-111470-99-6
48. C20h25cln2o5.c6h6o3s
49. Ncgc00165957-04
50. Amlodipine 100 Microg/ml In Acetonitrile
51. Unii-864v2q084h
52. Amlodipine Besylate [usan:usp]
53. Uk 48340-26
54. 3-ethyl 5-methyl 2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-6-methyl-1,4-dihydropyridine-3,5-dicarboxylate Benzenesulfonate
55. Mfcd00887594
56. Norliqva
57. Spectrum_000573
58. Amlodipine Benzenesulphonate
59. Amlodipine Besylate (usp)
60. Amlodipine Besylate- Bio-x
61. Amlodipine Besilate (jp17)
62. Schembl41282
63. Kbioss_001053
64. 3-ethyl 5-methyl (+-)-2-((2-aminoethoxy)methyl)-4-(o-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarboxylate, Monobenzenesulfonate
65. Mls001361394
66. Spectrum1505202
67. Chembl1200402
68. Chembl3193637
69. Chembl3207778
70. Dtxsid2043909
71. Hy-b0317b
72. Kbio2_001053
73. Kbio2_003621
74. Kbio2_006189
75. Amlodipine Besilate [jan]
76. Amlodipine Besylate [hsdb]
77. Hms2094e13
78. Hms2232d22
79. Hms2235b19
80. Hms3269b19
81. Hms3372o18
82. Hms3373f15
83. Hms3413h11
84. Hms3677h11
85. Hms3884m10
86. Pharmakon1600-01505202
87. Amlodipine Besylate [vandf]
88. Amlodipine Besilate [mart.]
89. Bcp21993
90. Tox21_111525
91. Tox21_113476
92. Ac-079
93. Amlodipine Besilate [who-dd]
94. Amlodipine Besylate [usp-rs]
95. Ccg-39531
96. Nsc758922
97. S1813
98. Akos001260256
99. Akos016340493
100. Amlodipine Besylate [ema Epar]
101. Amlodipine Besylate, >=98% (hplc)
102. Bcp9000059
103. Cs-3988
104. Ks-1029
105. Nsc 758922
106. Amlodipine Benzenesulfonate [mi]
107. Amlodipine Besylate [orange Book]
108. Azor Component Amlodipine Besylate
109. 3,5-pyridinedicarboxylic Acid, 2-((2-aminoethoxy)methyl)-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-, 3-ethyl-5-methyl Ester, Monobenzenesulfonate
110. Amlodipine Besilate [ep Monograph]
111. Amlodipine Besylate [usp Impurity]
112. Ba164162
113. Smr000875356
114. Amlodipine Besylate [usp Monograph]
115. Caduet Component Amlodipine Besylate
116. Dafiro Component Amlodipine Besilate
117. Lotrel Component Amlodipine Besylate
118. Amlessa Component Amlodipine Besylate
119. Copalia Component Amlodipine Besylate
120. Exforge Component Amlodipine Besilate
121. Exforge Component Amlodipine Besylate
122. Imprida Component Amlodipine Besilate
123. Onduarp Component Amlodipine Besylate
124. Tekamlo Component Amlodipine Besylate
125. Twynsta Component Amlodipine Besylate
126. Amlodipine Besylate Component Of Azor
127. Consensi Component Amlodipine Besylate
128. Ft-0622301
129. Sw198880-2
130. Amlodipine Besilate Component Of Dafiro
131. Amlodipine Besylate Component Of Caduet
132. Amlodipine Besylate Component Of Lotrel
133. Amturnide Component Amlodipine Besylate
134. Tribenzor Component Amlodipine Besylate
135. Vu0244594-3
136. Amlodipine Besilate Component Of Exforge
137. Amlodipine Besylate Component Of Copalia
138. Amlodipine Besylate Component Of Exforge
139. Amlodipine Besylate Component Of Onduarp
140. Amlodipine Besylate Component Of Tekamlo
141. Amlodipine Besylate Component Of Twynsta
142. D00615
143. Dafiro-hct Component Amlodipine Besilate
144. F17340
145. Amlodipine Besylate [ema Epar Veterinary]
146. Amlodipine Besylate Component Of Amturnide
147. Amlodipine Besylate Component Of Consensi
148. Amlodipine Besylate Component Of Tribenzor
149. Exforge Hct Component Amlodipine Besylate
150. Exforge-hct Component Amlodipine Besilate
151. Imprida-hct Component Amlodipine Besilate
152. 470a996
153. A802368
154. Amlodipine Besilate Component Of Dafiro-hct
155. Amlodipine Besilate Component Of Exforge-hct
156. Amlodipine Besilate Component Of Imprida-hct
157. Amlodipine Besylate Component Of Exforge Hct
158. Q-101934
159. Q-200627
160. Sr-05000001461-2
161. Q27105760
162. F6170-0024
163. Amlodipine Besylate, European Pharmacopoeia (ep) Reference Standard
164. Amlodipine Besylate, United States Pharmacopeia (usp) Reference Standard
165. Amlodipine Besylate, Pharmaceutical Secondary Standard; Certified Reference Material
166. Amlodipine For Peak Identification, European Pharmacopoeia (ep) Reference Standard
167. (+/-)-3-ethyl 5-methyl 2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarboxylate Monobenzenesulfonate
168. 2-(2-aminoethoxy)methyl-4-(2-chlorophenyl)-6-methyl-1,4-dihydropyridine-3,5-dicarboxylic Acid 3-ethyl 5-methyl Ester Benzenesulfona
169. 2-(2-aminoethoxymethyl)-4-(2-chlorophenyl)-6-methyl-1,4-dihydropyridine-3,5-dicarboxylic Acid O3-ethyl Ester O5-methyl Ester; Benzenesulfonic Acid
170. 2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarbo Xylic Acid 3-ethyl 5-methyl Ester Benzenesulfonate
171. 2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarboxylic Acid 3-ethyl 5-methyl Ester Benzenesulfonate
172. 2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-3-ethoxycarbonyl-5-methoxycarbonyl-6-methyl-1,4-dihydropyridine Benzenesulfonate
173. 3,5-pyridinedicarboxylic Acid, 2-((2-aminoethoxy)methyl)-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-, 3-ethyl 5-methyl Ester, (+/-)-, Monobenzenesulfonate
174. 3,5-pyridinedicarboxylic Acid, 2-((2-aminoethoxy)methyl)-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-, 3-ethyl 5-methyl Ester, (+/-)-, Monobenzenesulphonate
175. 3,5-pyridinedicarboxylic Acid, 2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-, 3-ethyl 5-methyl Ester, Benzenesulfonate (1:1)
176. 3-ethyl 5-methyl (+/-)-2-((2-aminoethoxy)methyl)-4-(o-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarboxylate, Monobenzenesulfonate
177. 3-ethyl 5-methyl (+/-)-2-((2-aminoethoxy)methyl)-4-(o-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarboxylate, Monobenzenesulphonate
178. 3-ethyl 5-methyl 2-(2-aminoethoxy)methyl-4-(2-chlorophenyl)-6-methyl-1,4-dihydropyridine-3,5-dicarboxylate Benzenesulfonate
179. 3-ethyl 5-methyl 2-(2-aminoethoxymethyl)-4-(2-chlorophenyl)-1,4-dihydro-6-methylpyridine-3,5-dicarboxylate Monobenzenesulphonate
180. 3-ethyl5-methyl2-((2-aminoethoxy)methyl)-4-(2-chlorophenyl)-6-methyl-1,4-dihydropyridine-3,5-dicarboxylatebenzenesulfonate
181. Benzenesulfonic Acid; O3-ethyl O5-methyl 2-(2-azanylethoxymethyl)-4-(2-chlorophenyl)-6-methyl-1,4-dihydropyridine-3,5-dicarboxylate
1. Prestalia
| Molecular Weight | 567.1 g/mol |
|---|---|
| Molecular Formula | C26H31ClN2O8S |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 11 |
| Exact Mass | 566.1489648 g/mol |
| Monoisotopic Mass | 566.1489648 g/mol |
| Topological Polar Surface Area | 163 Ų |
| Heavy Atom Count | 38 |
| Formal Charge | 0 |
| Complexity | 830 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 4 | |
|---|---|
| Drug Name | Amlodipine besylate |
| PubMed Health | Amlodipine (By mouth) |
| Drug Classes | Antianginal, Antihypertensive, Cardiovascular Agent |
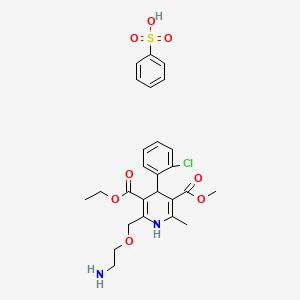
| Drug Label | Amlodipine besylate is the besylate salt of amlodipine, a long-acting calcium channel blocker. Amlodipine besylate is chemically described as 3-Ethyl-5-methyl ()-2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarbox... |
| Active Ingredient | Amlodipine besylate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 5mg base; eq 2.5mg base; eq 10mg base |
| Market Status | Prescription |
| Company | Watson Labs; Wockhardt; Ranbaxy; Amneal Pharms Ny; Upsher Smith; Secan Pharms; Matrix Labs; Epic Pharma; Macleods Pharms; Vivimed Labs; Teva; Apotex; Accord Hlthcare; Vintage; Alkem; Sun Pharm Inds; Aurobindo Pharma; Torrent Pharms; Zydus Pharms Usa; Lupi |
| 2 of 4 | |
|---|---|
| Drug Name | Norvasc |
| Drug Label | NORVASC is the besylate salt of amlodipine, a long-acting calcium channel blocker.Amlodipine besylate is chemically described as 3-Ethyl-5-methyl ()-2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarboxylate, monobe... |
| Active Ingredient | Amlodipine besylate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 5mg base; eq 2.5mg base; eq 10mg base |
| Market Status | Prescription |
| Company | Pfizer |
| 3 of 4 | |
|---|---|
| Drug Name | Amlodipine besylate |
| PubMed Health | Amlodipine (By mouth) |
| Drug Classes | Antianginal, Antihypertensive, Cardiovascular Agent |
| Drug Label | Amlodipine besylate is the besylate salt of amlodipine, a long-acting calcium channel blocker. Amlodipine besylate is chemically described as 3-Ethyl-5-methyl ()-2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarbox... |
| Active Ingredient | Amlodipine besylate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 5mg base; eq 2.5mg base; eq 10mg base |
| Market Status | Prescription |
| Company | Watson Labs; Wockhardt; Ranbaxy; Amneal Pharms Ny; Upsher Smith; Secan Pharms; Matrix Labs; Epic Pharma; Macleods Pharms; Vivimed Labs; Teva; Apotex; Accord Hlthcare; Vintage; Alkem; Sun Pharm Inds; Aurobindo Pharma; Torrent Pharms; Zydus Pharms Usa; Lupi |
| 4 of 4 | |
|---|---|
| Drug Name | Norvasc |
| Drug Label | NORVASC is the besylate salt of amlodipine, a long-acting calcium channel blocker.Amlodipine besylate is chemically described as 3-Ethyl-5-methyl ()-2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarboxylate, monobe... |
| Active Ingredient | Amlodipine besylate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 5mg base; eq 2.5mg base; eq 10mg base |
| Market Status | Prescription |
| Company | Pfizer |
Treatment of systemic arterial hypertension in cats.
Antihypertensive Agents
Drugs used in the treatment of acute or chronic vascular HYPERTENSION regardless of pharmacological mechanism. Among the antihypertensive agents are DIURETICS; (especially DIURETICS, THIAZIDE); ADRENERGIC BETA-ANTAGONISTS; ADRENERGIC ALPHA-ANTAGONISTS; ANGIOTENSIN-CONVERTING ENZYME INHIBITORS; CALCIUM CHANNEL BLOCKERS; GANGLIONIC BLOCKERS; and VASODILATOR AGENTS. (See all compounds classified as Antihypertensive Agents.)
Calcium Channel Blockers
A class of drugs that act by selective inhibition of calcium influx through cellular membranes. (See all compounds classified as Calcium Channel Blockers.)
Vasodilator Agents
Drugs used to cause dilation of the blood vessels. (See all compounds classified as Vasodilator Agents.)
QC08CA01
 Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Certificate Number : R1-CEP 2005-052 - Rev 05
Issue Date : 2021-02-25
Type : Chemical
Substance Number : 1491
Status : Withdrawn by Holder

 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.

GDUFA
DMF Review : Reviewed
Rev. Date : 2013-05-21
Pay. Date : 2013-05-10
DMF Number : 16473
Submission : 2003-03-19
Status : Active
Type : II

Registration Number : 219MF10352
Registrant's Address : 8-2-337, Road No. 3, Banjara Hills, Hyderabad 500 034, TELANGANA, INDIA
Initial Date of Registration : 2007-12-10
Latest Date of Registration :

Date of Issue : 2025-05-28
Valid Till : 2028-06-25
Written Confirmation Number : WC-0035
Address of the Firm :

NDC Package Code : 55111-025
Start Marketing Date : 2003-03-19
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Registrant Name : Boryeong Co., Ltd.
Registration Date : 2016-01-04
Registration Number : 20110414-130-H-86-20(11)
Manufacturer Name : Dr. Reddy's Laboratories Limited
Manufacturer Address : Unit-III, Plot No. 116, Sri Venkateswara Co-operative Industrial Estate, IDA Bollaram, Jinnaram Mandal, Sangareddy District- 502 325, Telangana State, India

| Available Reg Filing : CDMF |

 Expand your portfolio with Egis- value-added generics, API capabilities, and reliable contract services to drive your growth.
Expand your portfolio with Egis- value-added generics, API capabilities, and reliable contract services to drive your growth.

Certificate Number : R1-CEP 2007-185 - Rev 02
Issue Date : 2016-12-19
Type : Chemical
Substance Number : 1491
Status : Valid

Registrant Name : Servier Korea Co., Ltd.
Registration Date : 2016-03-23
Registration Number : 20160323-130-H-293-45
Manufacturer Name : EGIS PHARMACEUTICALS PRIVATE LIMITED COMPANY(Egis Pharmaceuticals PLC)
Manufacturer Address : Site 1, H-1106 Budapest, X. Kereszturi Ut 30-38

| Available Reg Filing : CN, BR |

 Gonane has API manufacturing expertise in new-age Corticosteroids, Hormones and other pharma raw materials.
Gonane has API manufacturing expertise in new-age Corticosteroids, Hormones and other pharma raw materials.
 Malladi is a leader in Ephedrine, Pseudoephedrine Salts & Phenylephrine HCl // USFDA, EDQM, ANSM, KFDA, and TGA inspected.
Malladi is a leader in Ephedrine, Pseudoephedrine Salts & Phenylephrine HCl // USFDA, EDQM, ANSM, KFDA, and TGA inspected.

NDC Package Code : 57218-936
Start Marketing Date : 2017-08-07
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 17070
Submission : 2003-12-29
Status : Active
Type : II

Certificate Number : R1-CEP 2005-237 - Rev 02
Issue Date : 2020-06-29
Type : Chemical
Substance Number : 1491
Status : Valid

Registration Number : 304MF10142
Registrant's Address : Cesar Martinell i Brunet 12A, Poligono Rubi Sur, Rubi (Barcelona), Spain
Initial Date of Registration : 2022-10-19
Latest Date of Registration :

NDC Package Code : 52932-0723
Start Marketing Date : 2010-07-23
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Registrant Name : New One Science Co., Ltd.
Registration Date : 2012-08-24
Registration Number : 20101116-130-H-25-05(1)
Manufacturer Name : Moehs Catalana, SL
Manufacturer Address : Poligono Rubi Sur, Cesar Martinell I Brunet n12A, 08191, Rubi (Barcelona), Spain

| Available Reg Filing : ASMF |

 Century has been an API manufacturer for over 40 years & is the partner of choice for multipurpose custom manufacturing projects.
Century has been an API manufacturer for over 40 years & is the partner of choice for multipurpose custom manufacturing projects.
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
CAS Number : 88150-62-3
End Use API : Amlodipine Besylate
About The Company : Aurore Life Sciences is a pure-play API manufacturer with a diverse portfolio of capabilities in delivering efficiencies to generic players globally. Founded in...

CAS Number : 88150-42-9
End Use API : Amlodipine Besylate
About The Company : Aurore Life Sciences is a pure-play API manufacturer with a diverse portfolio of capabilities in delivering efficiencies to generic players globally. Founded in...

CAS Number : 88150-62-3
End Use API : Amlodipine Besylate
About The Company : Cadila Pharmaceuticals Ltd. is one of the largest privately-held pharmaceutical companies in India. Over the past six decades, we have been developing and manuf...

CAS Number : 88150-62-3
End Use API : Amlodipine Besylate
About The Company : Prudence Pharmachem, founded in 2003, operates in the production and advancement of a diverse array of premium pharmaceutical bulk drugs (API) and their advance...

CAS Number : 88150-42-9
End Use API : Amlodipine Besylate
About The Company : Prudence Pharmachem, founded in 2003, operates in the production and advancement of a diverse array of premium pharmaceutical bulk drugs (API) and their advance...

S-Amlodipine Base [Leva Amlodipine Base]
CAS Number : 103129-82-4
End Use API : Amlodipine Besylate
About The Company : Prudence Pharmachem, founded in 2003, operates in the production and advancement of a diverse array of premium pharmaceutical bulk drugs (API) and their advance...

CAS Number : 88150-62-3
End Use API : Amlodipine Besylate
About The Company : Smilax Laboratories Limited is a research-driven, vertically integrated pharmaceutical company manufacturing Active Pharmaceutical Ingredients, API Intermediate...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : PRESCRIPTION
Registration Country : Canada
AMLODIPINE (AMLODIPINE BESYLATE)
Brand Name : AG-AMLODIPINE
Dosage Form : TABLET
Dosage Strength : 5MG
Packaging : 100/500
Approval Date :
Application Number : 2369230
Regulatory Info : PRESCRIPTION
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : PRESCRIPTION
Registration Country : Canada
ATORVASTATIN (ATORVASTATIN CALCIUM PROPYLENE GLYCOL SOLVATE)
Brand Name : APO-AMLODIPINE-ATORVASTATIN
Dosage Form : TABLET
Dosage Strength : 40MG
Packaging : 100
Approval Date :
Application Number : 2411288
Regulatory Info : PRESCRIPTION
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : PRESCRIPTION
Registration Country : Canada
AMLODIPINE (AMLODIPINE BESYLATE)
Brand Name : APO-PERINDOPRIL/AMLODIPINE
Dosage Form : TABLET
Dosage Strength : 5MG
Packaging : 30/100/500
Approval Date :
Application Number : 2468573
Regulatory Info : PRESCRIPTION
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : PRESCRIPTION
Registration Country : Canada
AMLODIPINE (AMLODIPINE BESYLATE)
Brand Name : JAMP-AMLODIPINE
Dosage Form : TABLET
Dosage Strength : 5MG
Packaging : 30/100/250/500
Approval Date :
Application Number : 2357194
Regulatory Info : PRESCRIPTION
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : PRESCRIPTION
Registration Country : Canada
AMLODIPINE (AMLODIPINE BESYLATE)
Brand Name : RIVA-TELMISARTAN/AMLODIPINE
Dosage Form : TABLET
Dosage Strength : 10MG
Packaging :
Approval Date :
Application Number : 2559889
Regulatory Info : PRESCRIPTION
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : PRESCRIPTION
Registration Country : Canada
AMLODIPINE (AMLODIPINE BESYLATE)
Brand Name : MAR-AMLODIPINE
Dosage Form : TABLET
Dosage Strength : 10MG
Packaging : 100/250/500
Approval Date :
Application Number : 2371723
Regulatory Info : PRESCRIPTION
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : PRESCRIPTION
Registration Country : Canada
AMLODIPINE (AMLODIPINE BESYLATE)
Brand Name : MINT-AMLODIPINE
Dosage Form : TABLET
Dosage Strength : 10MG
Packaging : 100/250
Approval Date :
Application Number : 2362678
Regulatory Info : PRESCRIPTION
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : PRESCRIPTION
Registration Country : Canada
AMLODIPINE (AMLODIPINE BESYLATE)
Brand Name : PRZ-AMLODIPINE
Dosage Form : TABLET
Dosage Strength : 10MG
Packaging :
Approval Date :
Application Number : 2522527
Regulatory Info : PRESCRIPTION
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : PRESCRIPTION
Registration Country : Canada
AMLODIPINE (AMLODIPINE BESYLATE)
Brand Name : SANDOZ AMLODIPINE
Dosage Form : TABLET
Dosage Strength : 5MG
Packaging : 100/500
Approval Date :
Application Number : 2284383
Regulatory Info : PRESCRIPTION
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : PRESCRIPTION
Registration Country : Canada
ATORVASTATIN (ATORVASTATIN CALCIUM PROPYLENE GLYCOL SOLVATE)
Brand Name : ATORVASTATIN-80
Dosage Form : TABLET
Dosage Strength : 80MG
Packaging : 30/90/100
Approval Date :
Application Number : 2411385
Regulatory Info : PRESCRIPTION
Registration Country : Canada

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
13
PharmaCompass offers a list of Amlodipine Besylate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Amlodipine Besylate manufacturer or Amlodipine Besylate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Amlodipine Besylate manufacturer or Amlodipine Besylate supplier.
A Amlodipine Besylate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Amlodipine Besylate, including repackagers and relabelers. The FDA regulates Amlodipine Besylate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Amlodipine Besylate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Amlodipine Besylate manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Amlodipine Besylate supplier is an individual or a company that provides Amlodipine Besylate active pharmaceutical ingredient (API) or Amlodipine Besylate finished formulations upon request. The Amlodipine Besylate suppliers may include Amlodipine Besylate API manufacturers, exporters, distributors and traders.
click here to find a list of Amlodipine Besylate suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Amlodipine Besylate DMF (Drug Master File) is a document detailing the whole manufacturing process of Amlodipine Besylate active pharmaceutical ingredient (API) in detail. Different forms of Amlodipine Besylate DMFs exist exist since differing nations have different regulations, such as Amlodipine Besylate USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Amlodipine Besylate DMF submitted to regulatory agencies in the US is known as a USDMF. Amlodipine Besylate USDMF includes data on Amlodipine Besylate's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Amlodipine Besylate USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Amlodipine Besylate suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Amlodipine Besylate Drug Master File in Japan (Amlodipine Besylate JDMF) empowers Amlodipine Besylate API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Amlodipine Besylate JDMF during the approval evaluation for pharmaceutical products. At the time of Amlodipine Besylate JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Amlodipine Besylate suppliers with JDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Amlodipine Besylate Drug Master File in Korea (Amlodipine Besylate KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Amlodipine Besylate. The MFDS reviews the Amlodipine Besylate KDMF as part of the drug registration process and uses the information provided in the Amlodipine Besylate KDMF to evaluate the safety and efficacy of the drug.
After submitting a Amlodipine Besylate KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Amlodipine Besylate API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Amlodipine Besylate suppliers with KDMF on PharmaCompass.
A Amlodipine Besylate CEP of the European Pharmacopoeia monograph is often referred to as a Amlodipine Besylate Certificate of Suitability (COS). The purpose of a Amlodipine Besylate CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Amlodipine Besylate EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Amlodipine Besylate to their clients by showing that a Amlodipine Besylate CEP has been issued for it. The manufacturer submits a Amlodipine Besylate CEP (COS) as part of the market authorization procedure, and it takes on the role of a Amlodipine Besylate CEP holder for the record. Additionally, the data presented in the Amlodipine Besylate CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Amlodipine Besylate DMF.
A Amlodipine Besylate CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Amlodipine Besylate CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Amlodipine Besylate suppliers with CEP (COS) on PharmaCompass.
A Amlodipine Besylate written confirmation (Amlodipine Besylate WC) is an official document issued by a regulatory agency to a Amlodipine Besylate manufacturer, verifying that the manufacturing facility of a Amlodipine Besylate active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Amlodipine Besylate APIs or Amlodipine Besylate finished pharmaceutical products to another nation, regulatory agencies frequently require a Amlodipine Besylate WC (written confirmation) as part of the regulatory process.
click here to find a list of Amlodipine Besylate suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Amlodipine Besylate as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Amlodipine Besylate API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Amlodipine Besylate as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Amlodipine Besylate and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Amlodipine Besylate NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Amlodipine Besylate suppliers with NDC on PharmaCompass.
Amlodipine Besylate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Amlodipine Besylate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Amlodipine Besylate GMP manufacturer or Amlodipine Besylate GMP API supplier for your needs.
A Amlodipine Besylate CoA (Certificate of Analysis) is a formal document that attests to Amlodipine Besylate's compliance with Amlodipine Besylate specifications and serves as a tool for batch-level quality control.
Amlodipine Besylate CoA mostly includes findings from lab analyses of a specific batch. For each Amlodipine Besylate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Amlodipine Besylate may be tested according to a variety of international standards, such as European Pharmacopoeia (Amlodipine Besylate EP), Amlodipine Besylate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Amlodipine Besylate USP).

