
Synopsis
Synopsis
0
USDMF
0
JDMF
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents

1. Hyperlipen
2. Lipanor
3. Modalim
4. Oroxadin
5. Win 35,833
1. 52214-84-3
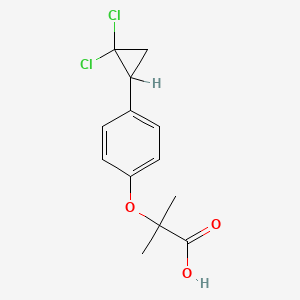
2. 2-(4-(2,2-dichlorocyclopropyl)phenoxy)-2-methylpropanoic Acid
3. Lipanor
4. Ciprofibratum
5. 2-[4-(2,2-dichlorocyclopropyl)phenoxy]-2-methylpropanoic Acid
6. Ciprofibrato
7. Modalim
8. Win 35833
9. Win-35833
10. Propanoic Acid, 2-[4-(2,2-dichlorocyclopropyl)phenoxy]-2-methyl-
11. Mfcd00467135
12. 2-(p-(2,2-dichlorocyclopropyl)phenoxy)-2-methylpropionic Acid
13. Nsc-759617
14. F8252jgo9s
15. Chebi:50867
16. Propanoic Acid, 2-(4-(2,2-dichlorocyclopropyl)phenoxy)-2-methyl-
17. Ncgc00164447-01
18. 2-{[4-(2,2-dichlorocyclopropyl)phenyl]oxy}-2-methylpropanoic Acid
19. Dsstox_cid_331
20. Dsstox_rid_75519
21. Dsstox_gsid_20331
22. Ciprofibratum [inn-latin]
23. Ciprofibrato [inn-spanish]
24. Hiperlipen
25. Ciprol
26. Smr000857190
27. Ccris 173
28. Sr-01000075734
29. Einecs 257-744-6
30. Ciprofibrate (usan/inn)
31. Brn 1984981
32. Unii-f8252jgo9s
33. 2-[p-(2,2-dichlorocyclopropyl)phenoxy]-2-methylpropionic Acid
34. Ciprofibrate?
35. Ciprofibrate [usan:inn:ban]
36. 2-(4-(2,2-dichlorocyclopropyl)phenoxy)2-methylpropanoic Acid
37. 2-[p-(2,2-dichlorocyclopropyl)phenoxy]-2-methylpropanoic Acid
38. Cas-52214-84-3
39. Ciprofibrate- Bio-x
40. Prestwick0_001076
41. Prestwick1_001076
42. Prestwick2_001076
43. Prestwick3_001076
44. Ciprofibrate [mi]
45. Ciprofibrate [inn]
46. C 0330
47. Ciprofibrate [usan]
48. Lopac0_000168
49. Schembl26870
50. Bspbio_001092
51. Ciprofibrate [mart.]
52. Mls002154143
53. Mls006011758
54. Ciprofibrate [who-dd]
55. Win35833
56. Spbio_002996
57. Bpbio1_001202
58. Chembl557555
59. Gtpl3438
60. Dtxsid8020331
61. Bcpp000277
62. Ciprofibrate [ep Impurity]
63. Ciprofibrate For System Suitability
64. Hms1571g14
65. Hms2098g14
66. Hms2235j14
67. Hms3260b17
68. Hms3369b22
69. Hms3656p09
70. Hms3715g14
71. Pharmakon1600-01502008
72. Ciprofibrate [ep Monograph]
73. Bcp03728
74. Hy-b0664
75. Tox21_112121
76. Tox21_201359
77. Tox21_302840
78. Tox21_500168
79. Bbl010821
80. Bdbm50371235
81. Nsc759617
82. S2665
83. Stk624758
84. Akos005557843
85. Tox21_112121_1
86. Ac-1018
87. Bcp9000533
88. Ccg-204263
89. Db09064
90. Ks-1201
91. Lp00168
92. Nsc 759617
93. Sdccgsbi-0050156.p002
94. Ncgc00015202-02
95. Ncgc00015202-03
96. Ncgc00015202-04
97. Ncgc00015202-05
98. Ncgc00015202-06
99. Ncgc00015202-08
100. Ncgc00015202-20
101. Ncgc00015202-21
102. Ncgc00093652-01
103. Ncgc00093652-02
104. Ncgc00256361-01
105. Ncgc00258911-01
106. Ncgc00260853-01
107. Bd164311
108. Bcp0726000148
109. Ab00514032
110. Eu-0100168
111. Ft-0602946
112. Sw197242-4
113. D03521
114. O10468
115. Ab00514032_06
116. 214c843
117. A828985
118. J-520054
119. Q3496452
120. Sr-01000075734-1
121. Sr-01000075734-4
122. Sr-01000075734-7
123. Brd-a49358627-001-03-9
124. Brd-a49358627-001-10-4
125. 2-[4-(2,2-dichlorocyclopropyl)phenoxy]-2-methylpropionic Acid
126. Ciprofibrate, European Pharmacopoeia (ep) Reference Standard
127. 2-[4-[2,2-bis(chloranyl)cyclopropyl]phenoxy]-2-methyl-propanoic Acid
128. Ciprofibrate For System Suitability, European Pharmacopoeia (ep) Reference Standard
| Molecular Weight | 289.15 g/mol |
|---|---|
| Molecular Formula | C13H14Cl2O3 |
| XLogP3 | 3.4 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 4 |
| Exact Mass | 288.0319997 g/mol |
| Monoisotopic Mass | 288.0319997 g/mol |
| Topological Polar Surface Area | 46.5 Ų |
| Heavy Atom Count | 18 |
| Formal Charge | 0 |
| Complexity | 333 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Peroxisome Proliferators
A class of nongenotoxic CARCINOGENS that induce the production of hepatic PEROXISOMES and induce hepatic neoplasms after long-term administration. (See all compounds classified as Peroxisome Proliferators.)
Hypolipidemic Agents
Substances that lower the levels of certain LIPIDS in the BLOOD. They are used to treat HYPERLIPIDEMIAS. (See all compounds classified as Hypolipidemic Agents.)
C10AB08
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
C - Cardiovascular system
C10 - Lipid modifying agents
C10A - Lipid modifying agents, plain
C10AB - Fibrates
C10AB08 - Ciprofibrate
 Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.

Certificate Number : CEP 2011-120 - Rev 05
Issue Date : 2024-04-17
Type : Chemical
Substance Number : 2013
Status : Valid

 Zhejiang Hengkang Pharm Group is a dynamic pharmaceutical entity, spanning drug research, large-scale production, and global marketing.
Zhejiang Hengkang Pharm Group is a dynamic pharmaceutical entity, spanning drug research, large-scale production, and global marketing.



Date of Issue : 2025-06-11
Valid Till : 2027-12-09
Written Confirmation Number : WC-0321
Address of the Firm :



Certificate Number : CEP 2013-112 - Rev 03
Issue Date : 2024-05-03
Type : Chemical
Substance Number : 2013
Status : Valid



 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
 Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
 Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Certificate Number : CEP 2011-120 - Rev 05
Status : Valid
Issue Date : 2024-04-17
Type : Chemical
Substance Number : 2013
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : CEP 2013-112 - Rev 03
Status : Valid
Issue Date : 2024-05-03
Type : Chemical
Substance Number : 2013

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : R0-CEP 2021-263 - Rev 00
Status : Valid
Issue Date : 2023-06-12
Type : Chemical
Substance Number : 2013

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Date of Issue : 2025-06-11
Valid Till : 2027-12-09
Written Confirmation Number : WC-0321
Address of the Firm : NS 13-16, N.I.E.C.S. Ltd., Uppalwadi, Nagpur-440026, Taluka: Nagpur Rural, Distr...

Date of Issue : 2022-12-30
Valid Till : 2026-01-05
Written Confirmation Number : WC-0384
Address of the Firm : UNIT-IV, Sy No. 332, 333 & 335 Veliminedu (Village), Chityal (Mandal), Nalgonda ...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
64
PharmaCompass offers a list of Ciprofibrate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ciprofibrate manufacturer or Ciprofibrate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Ciprofibrate manufacturer or Ciprofibrate supplier.
A Ciprofibrate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Ciprofibrate, including repackagers and relabelers. The FDA regulates Ciprofibrate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Ciprofibrate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Ciprofibrate manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Ciprofibrate supplier is an individual or a company that provides Ciprofibrate active pharmaceutical ingredient (API) or Ciprofibrate finished formulations upon request. The Ciprofibrate suppliers may include Ciprofibrate API manufacturers, exporters, distributors and traders.
click here to find a list of Ciprofibrate suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Ciprofibrate CEP of the European Pharmacopoeia monograph is often referred to as a Ciprofibrate Certificate of Suitability (COS). The purpose of a Ciprofibrate CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Ciprofibrate EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Ciprofibrate to their clients by showing that a Ciprofibrate CEP has been issued for it. The manufacturer submits a Ciprofibrate CEP (COS) as part of the market authorization procedure, and it takes on the role of a Ciprofibrate CEP holder for the record. Additionally, the data presented in the Ciprofibrate CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Ciprofibrate DMF.
A Ciprofibrate CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Ciprofibrate CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Ciprofibrate suppliers with CEP (COS) on PharmaCompass.
A Ciprofibrate written confirmation (Ciprofibrate WC) is an official document issued by a regulatory agency to a Ciprofibrate manufacturer, verifying that the manufacturing facility of a Ciprofibrate active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Ciprofibrate APIs or Ciprofibrate finished pharmaceutical products to another nation, regulatory agencies frequently require a Ciprofibrate WC (written confirmation) as part of the regulatory process.
click here to find a list of Ciprofibrate suppliers with Written Confirmation (WC) on PharmaCompass.
Ciprofibrate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Ciprofibrate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ciprofibrate GMP manufacturer or Ciprofibrate GMP API supplier for your needs.
A Ciprofibrate CoA (Certificate of Analysis) is a formal document that attests to Ciprofibrate's compliance with Ciprofibrate specifications and serves as a tool for batch-level quality control.
Ciprofibrate CoA mostly includes findings from lab analyses of a specific batch. For each Ciprofibrate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Ciprofibrate may be tested according to a variety of international standards, such as European Pharmacopoeia (Ciprofibrate EP), Ciprofibrate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Ciprofibrate USP).

