By PharmaCompass
2025-08-28
Impressions: 7675
It has been a turbulent year for the US Food and Drug Administration (FDA), marked by reductions in both staff and budget, shake-ups at the top and a reduction in inspections. This upheaval has clearly impacted the functioning of the agency, with reports of missed deadlines and reduced responsiveness.
This disruption resulted in a slowdown in new drug approvals in the first half of 2025 (H1 2025), as opposed to the same period last year. Incidentally, drug approvals in H1 2024 were also down by 19 percent compared to H1 2023.
FDA’s Center for Drug Evaluation and Research (CDER) approved 16 novel drugs in H1 2025, down from 21 in H1 2024, a decline of nearly 24 percent. Of these, nine were small molecules and seven were biologics. The period saw eight first-in-class therapies get CDER’s approval, as opposed to 17 in H1 2024.
FDA’s Center for Biologics Evaluation and Research (CBER) approved seven biologics in H1 2025, compared to eight in the same period last year.
In comparison, both Health Canada and the European Medicines Agency (EMA) reported a strong rebound in approvals and authorizations, respectively. Health Canada approved 20 drugs in H1 2025, as opposed to 10 in H1 2024. Similarly, EMA authorized 19 new therapies in H1 2025, compared to 15 in H1 2024. Notably, EMA’s pending decisions increased to 25 in H1 2025, from 14 in the same period last year, while conditional authorizations remained static at five. This underscores EMA’s active regulatory pipeline, suggesting that more drugs could be approved in Europe soon.
View New Drug Approvals in H1 2025 with Estimated Sales (Free Excel Available)
Vertex’s non-opioid painkiller, GSK’s antibiotic for UTI, Merck’s RSV antibody bag FDA nods
Oncology remained the single largest category in H1 2025 with seven new drugs being approved by the FDA, followed by four rare diseases and disorders and as many infections and infective diseases.
The eight drugs that were designated first-in-class in H1 2025 are: Journavx (Suzetrigine), Imaavy (nipocalimab), Emrelis (telisotuzumab vedotin), Blujepa (gepotidacin mesylate), Qfitlia (fitusiran), Avmapki Fakzynja Co-Pack (avutometinib potassium/defactinib hydrochloride), Tryptyr (acoltremon), and Andembry (garadacimab).
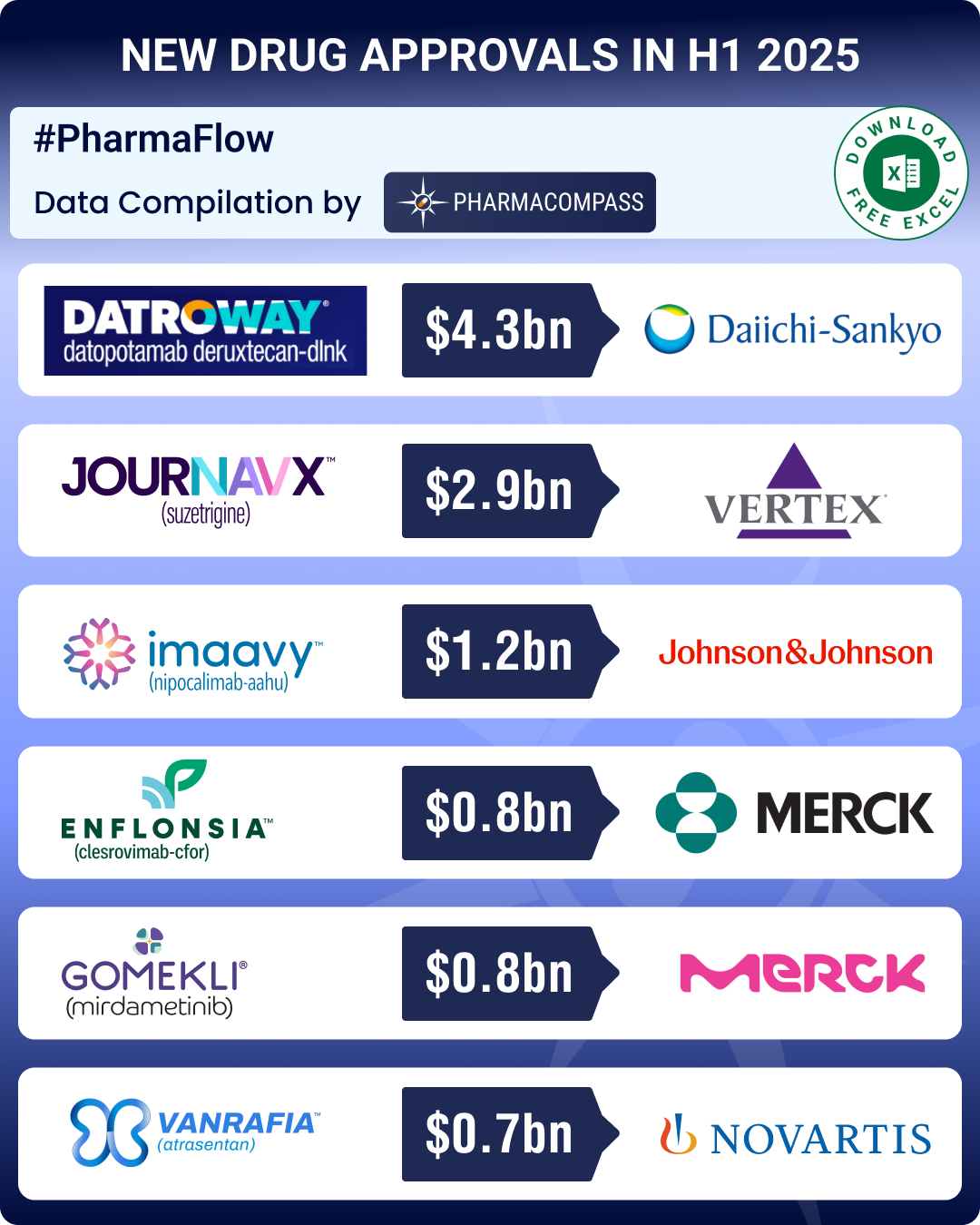
In January, a new class of non-opioid pain therapy made a debut — FDA approved Vertex Pharmaceuticals’ Journavx (suzetrigine). This first-in-class oral analgesic could bring in US$ 2.9 billion in sales for Vertex by 2030.
The half year also saw a new oral antibiotic — GSK’s Blujepa (gepotidacin) — for treating uncomplicated urinary tract infections (uUTIs) bag FDA approval. This is the first new antibiotic for uUTIs in nearly three decades. It treats infections caused by resistant pathogens such as E. coli and K. pneumoniae.
In June, Merck’s Enflonsia (clesrovimab), a long-acting monoclonal antibody, won FDA approval to prevent respiratory syncytial virus (RSV) lower respiratory tract disease in neonates and infants entering their first RSV season. A single 105 mg dose provides protection for about five months, the length of a typical RSV season. Enflonsia could bring in US$844 million in annual sales by 2030.
View New Drug Approvals in H1 2025 with Estimated Sales (Free Excel Available)
FDA approves four meds for rare diseases, including SpringWorks’ Gomekli, Novartis’ Vanrafia
In H1 2024, rare genetic diseases had witnessed some significant approvals. The trend continued in H1 2025. In February, Gomekli (mirdametinib), developed by SpringWorks Therapeutics, became the first approved therapy for neurofibromatosis type 1 (a genetic disorder that causes tumors to grow on nerve tissues) in adult and pediatric patients who have symptomatic plexiform neurofibromas (benign, complex nerve tumors) not amenable to complete resection. Analysts expect peak sales of around US$ 0.8 billion by 2030. Post this approval, SpringWorks got acquired by Merck KGaA for approximately US$ 3.9 billion.
The same month, Ono Pharma-owned Deciphera’s Romvimza (vimseltinib) became the first oral therapy approved for another rare disease known as symptomatic tenosynovial giant cell tumor. This is a benign but aggressive growth that can be painful. This new drug offers patients an option beyond surgery.
In March, Sanofi’s Qfitlia (fitusiran) was approved for routine prophylaxis in hemophilia A or B, with or without inhibitors. And in April, Novartis’ Vanrafia (atrasentan) was granted accelerated approval for IgA nephropathy (a chronic autoimmune kidney disease), which is at the risk of rapid progression. Vanrafia could bring in sales of over US$ 735 million by 2030.
Johnson & Johnson secured approval for Imaavy (nipocalimab-aahu) for generalized myasthenia gravis (a chronic autoimmune disease that causes muscle weakness) in adults and adolescents. Imaavy’s sales could cross US$1.17 billion by 2030.
Rare diseases continue to attract regulatory attention, with the trend carrying into the current quarter. July witnessed two noteworthy rare-disease approvals — Ekterly (garadacimab), CSL’s once-monthly prophylactic treatment for hereditary angioedema (a genetic disorder that causes recurrent episodes of swellings), and Sephience (pegvaliase-abcx), BioMarin’s new therapy for phenylketonuria (a rare metabolic disorder).
View New Drug Approvals in H1 2025 with Estimated Sales (Free Excel Available)
FDA greenlights new oncology therapies, including Astra’s Datroway, AbbVie’s Emrelis
The field of oncology saw some notable new drug approvals. In January, Datroway (datopotamab deruxtecan), an antibody-drug conjugate (ADC) developed by AstraZeneca and Daiichi Sankyo, was granted approval for adults with unresectable or metastatic, hormone receptor-positive, HER2-negative breast cancer. Later, in June, the FDA granted accelerated approval to AstraZeneca;s Datroway for adults with locally advanced or metastatic, EGFR-mutated non-small cell lung cancer. The drug’s peak annual sales are projected at about US$ 4.2 billion by 2030.
In May, Emrelis (telisotuzumab vedotin-tllv), AbbVie’s ADC was granted FDA’s accelerated approval. Emrelis treats locally advanced or metastatic non-squamous non-small cell lung cancer (NSCLC) characterized by significantly increased levels of the c-Met protein in cancer cells. FDA also approved a companion diagnostic test — Roche's Ventana — in May to determine the c-Met protein biomarker status in order to identify patients eligible for Emrelis.
The same month, FDA also granted accelerated approval to Verastem’s Avmapki Fakzynja Co-pack, a dual oral therapy for adults with KRAS‑mutated recurrent low‑grade serous ovarian cancer (LGSOC). This novel regimen combines two inhibitors — avutometinib and defactinib — to disrupt critical signaling pathways that fuel tumor growth.
In ophthalmology, Alcon re-entered the prescription pharmaceutical market with Tryptry (acoltremon), a first-in-class TRPM8 agonist for dry eye disease, which stimulates corneal nerves to increase tear production.
View New Drug Approvals in H1 2025 with Estimated Sales (Free Excel Available)
Our view
The changes in the FDA are clearly having a far-reaching impact on the global pharmaceutical industry. There have been reports of drugmakers losing confidence in the FDA, which has been a gold standard for drug regulation. Some American biotechs are shifting early trials outside of the US. With EMA and Health Canada authorizing/approving a larger number of drugs in H1 2025, we do see some changes underway that may eventually reshape drug development.
The PharmaCompass Newsletter – Sign Up, Stay Ahead
Feedback, help us to improve. Click here
Image Credit : NEW DRUG APPROVALS IN H1 2025 by PharmaCompass license under CC BY 2.0
“ The article is based on the information available in public and which the author believes to be true. The author is not disseminating any information, which the author believes or knows, is confidential or in conflict with the privacy of any person. The views expressed or information supplied through this article is mere opinion and observation of the author. The author does not intend to defame, insult or, cause loss or damage to anyone, in any manner, through this article.”







