13 Mar 2026
// PRESS RELEASE
12 Feb 2026
// PRESS RELEASE
24 Oct 2025
// PRESS RELEASE
Latest Content by PharmaCompass
 KEY PRODUCTS
KEY PRODUCTS KEY SERVICES
KEY SERVICES
European CDMO and Gx manufacturer with 75 years of experience in delivering premium APIs to pharmaceutical partners worldwide.
About
CPhI North AmericaCPhI North America
Industry Trade Show
Attending
02-04 June, 2026
Industry Trade Show
Booth #E2C71
16-18 June, 2026
BIO International Conv...BIO International Convention
Industry Trade Show
Attending
22-25 June, 2026
CONTACT DETAILS




Events
Webinars & Exhibitions
CPhI North AmericaCPhI North America
Industry Trade Show
Attending
02-04 June, 2026
Industry Trade Show
Booth #E2C71
16-18 June, 2026
BIO International Conv...BIO International Convention
Industry Trade Show
Attending
22-25 June, 2026
https://www.pharmacompass.com/speak-pharma/expert-take-on-cep-2-0-a-new-era-of-transparency-in-pharmaceutical-regulation
VLOG #PharmaReel
CORPORATE CONTENT #SupplierSpotlight
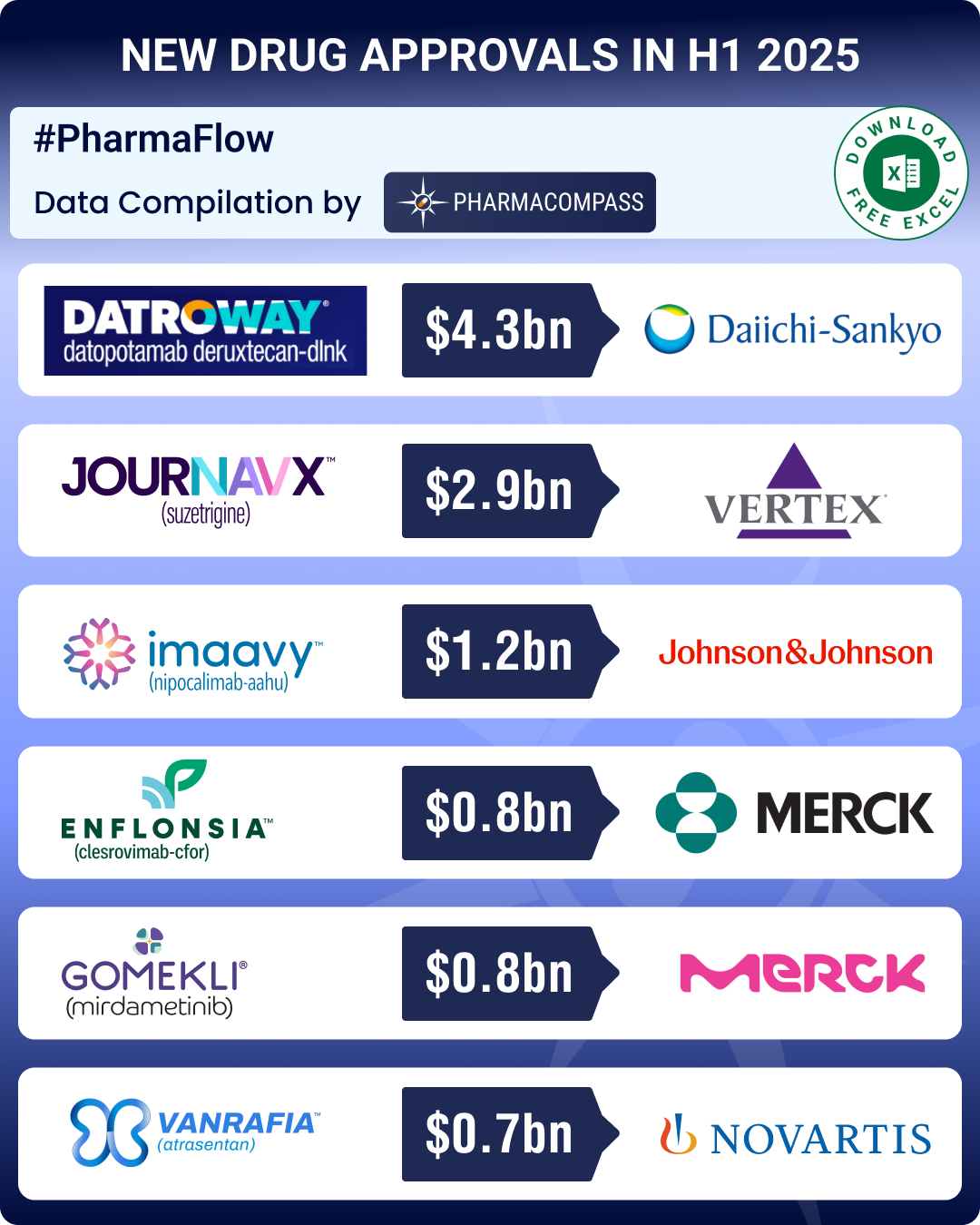
https://www.pharmacompass.com/radio-compass-blog/fda-approvals-drop-24-in-h1-2025-gsk-s-uti-med-vertex-s-non-opioid-painkiller-lead-pack-of-first-in-class-meds
https://www.pharmacompass.com/radio-compass-blog/cdmo-activity-tracker-veranova-carbogen-lead-adc-investments-axplora-polfa-tarchomin-famar-expand-european-footprint
https://www.pharmacompass.com/radio-compass-blog/cdmo-activity-tracker-bora-polpharma-make-acquisitions-evonik-euroapi-porton-announce-technological-expansions
https://www.pharmacompass.com/radio-compass-blog/fda-approves-record-eight-biosimilars-in-h1-2024-okays-first-interchangeable-biosimilars-for-eylea

13 Mar 2026
// PRESS RELEASE
https://polpharma.pl/en/polpharma-opens-an-office-in-india-strengthening-global-collaboration-and-api-supply-chain-resilience/

12 Feb 2026
// PRESS RELEASE
https://polpharma.pl/en/polpharma-sa-launches-a-project-under-the-step-initiative/

24 Oct 2025
// PRESS RELEASE
https://polpharma.pl/en/polpharma-group-announces-leadership-transition-sebastian-szymanek-appointed-as-its-new-president/#:~:text=At%20the%20same%20time%2C%20Polpharma,Farmaceutyczne%20Polpharma%20S.A.%20in%20Poland.

25 Jun 2025
// PRESS RELEASE
https://www.api.polpharma.com/articles/rising-to-the-solubility-challenge-collaborative-solutions-in-api-development

25 Jun 2025
// PRESS RELEASE
https://www.api.polpharma.com/articles/rising-to-the-solubility-challenge-collaborative-solutions-in-api-development

14 May 2025
// PRESS RELEASE
https://polpharma.pl/en/polpharma-group-joins-the-call-to-action-to-finalize-pharmaceutical-reform-and-secure-early-and-sustainable-healthcare-for-european-patients/
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 42990
Submission : 2025-12-05
Status : Active
Type : II
GDUFA
DMF Review : Complete
Rev. Date : 2013-01-30
Pay. Date : 2013-01-15
DMF Number : 10383
Submission : 1993-07-30
Status : Active
Type : II
GDUFA
DMF Review : Complete
Rev. Date : 2017-02-10
Pay. Date : 2016-12-20
DMF Number : 31166
Submission : 2016-11-16
Status : Active
Type : II
GDUFA
DMF Review : Complete
Rev. Date : 2017-04-07
Pay. Date : 2016-12-20
DMF Number : 31141
Submission : 2016-11-09
Status : Active
Type : II
GDUFA
DMF Review : Complete
Rev. Date : 2015-02-20
Pay. Date : 2013-12-19
DMF Number : 23314
Submission : 2009-11-27
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 33204
Submission : 2018-10-30
Status : Active
Type : II
GDUFA
DMF Review : Complete
Rev. Date : 2013-01-08
Pay. Date : 2012-12-11
DMF Number : 18014
Submission : 2005-01-20
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39562
Submission : 2024-12-16
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 32347
Submission : 2017-12-21
Status : Active
Type : II
GDUFA
DMF Review : Complete
Rev. Date : 2015-12-16
Pay. Date : 2015-06-25
DMF Number : 26266
Submission : 2012-07-31
Status : Active
Type : II
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Services
API Manufacturing
API & Drug Product Development
Inspections and registrations
Country : Poland
City/Region : Sieradz
Audit Date : 2025-10-14
Audit Type : On-Site
Country : Poland
City/Region : Starogard Gdański
Audit Date : Nov-25
Audit Type : On-Site
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]ABOUT THIS PAGE
Polpharma is a supplier offers 77 products (APIs, Excipients or Intermediates).
Find Alendronate Sodium bulk with DMF, CEP, JDMF offered by Polpharma
Find Hydrochlorothiazide bulk with DMF, CEP, JDMF offered by Polpharma
Find Ibandronate Sodium bulk with DMF, CEP, JDMF offered by Polpharma
Find Lamotrigine bulk with DMF, CEP, JDMF offered by Polpharma
Find Risedronate Sodium bulk with DMF, CEP, JDMF offered by Polpharma
Find Sildenafil Citrate bulk with DMF, CEP, JDMF offered by Polpharma
Find Tadalafil bulk with DMF, CEP, JDMF offered by Polpharma
Find Vardenafil Hydrochloride bulk with DMF, CEP, JDMF offered by Polpharma
Find Zoledronic Acid bulk with DMF, CEP, JDMF offered by Polpharma
Find Acetazolamide bulk with DMF, JDMF offered by Polpharma
Find Baclofen bulk with DMF, JDMF offered by Polpharma
Find Carbamazepine bulk with DMF, CEP offered by Polpharma
Find Dapagliflozin bulk with DMF, JDMF offered by Polpharma
Find Etodolac bulk with DMF, JDMF offered by Polpharma
Find Pentoxifylline bulk with DMF, CEP offered by Polpharma
Find Sodium Salicylate bulk with DMF, JDMF offered by Polpharma
Find Abemaciclib bulk with DMF offered by Polpharma
Find Apixaban bulk with DMF offered by Polpharma
Find Apremilast bulk with DMF offered by Polpharma
Find Aripiprazole bulk with DMF offered by Polpharma
Find Baricitinib bulk with DMF offered by Polpharma
Find Brexpiprazole bulk with DMF offered by Polpharma
Find Carbamazepine bulk with CEP offered by Polpharma
Find Carvedilol bulk with DMF offered by Polpharma
Find Carvedilol Phosphate bulk with DMF offered by Polpharma
Find Dapagliflozin Propanediol Monohydrate bulk with DMF offered by Polpharma
Find Empagliflozin bulk with DMF offered by Polpharma
Find Enzalutamide bulk with DMF offered by Polpharma
Find Ibandronate Sodium bulk with CEP offered by Polpharma
Find Isavuconazonium Sulfate bulk with DMF offered by Polpharma
Find Lamotrigine bulk with JDMF offered by Polpharma
Find Molsidomine bulk with CEP offered by Polpharma
Find Palbociclib bulk with DMF offered by Polpharma
Find Pimavanserin Tartrate bulk with DMF offered by Polpharma
Find Piracetam bulk with CEP offered by Polpharma
Find Rivaroxaban bulk with DMF offered by Polpharma
Find Sacubitril-Valsartan bulk with DMF offered by Polpharma
Find Safinamide Methanesulfonate bulk with DMF offered by Polpharma
Find Sildenafil Citrate bulk with CEP offered by Polpharma
Find Sitagliptin Hydrochloride bulk with DMF offered by Polpharma
Find Ticagrelor bulk with DMF offered by Polpharma
Find Tolterodine Tartrate bulk with CEP offered by Polpharma
Find Topiramate bulk with DMF offered by Polpharma
Find Trametinib bulk with DMF offered by Polpharma
Find Xylometazoline Hydrochloride bulk with CEP offered by Polpharma
Find Acenocoumarol bulk offered by Polpharma
Find Acetazolamide bulk offered by Polpharma
Find Alendronate Sodium bulk offered by Polpharma
Find Aniracetam bulk offered by Polpharma
Find Antazoline HCl bulk offered by Polpharma
Find Antazoline Mesylate bulk offered by Polpharma
Find Antazoline Sulfate bulk offered by Polpharma
Find Apixaban bulk offered by Polpharma
Find Aripiprazole bulk offered by Polpharma
Find Aripiprazole Lauroxil bulk offered by Polpharma
Find Baclofen bulk offered by Polpharma
Find Carbamazepine bulk offered by Polpharma
Find Carvedilol bulk offered by Polpharma
Find Carvedilol Phosphate bulk offered by Polpharma
Find CAS 66376-36-1 bulk offered by Polpharma
Find Clemastine bulk offered by Polpharma
Find Clopamide bulk offered by Polpharma
Find Denotivir bulk offered by Polpharma
Find Disulfiram bulk offered by Polpharma
Find Etodolac bulk offered by Polpharma
Find Hydrochlorothiazide bulk offered by Polpharma
Find Nefopam Hydrochloride bulk offered by Polpharma
Find Opipramol Dihydrochloride bulk offered by Polpharma
Find Phenyl Salicylate bulk offered by Polpharma
Find Risdiplam bulk offered by Polpharma
Find Risedronate Sodium bulk offered by Polpharma
Find Rivaroxaban bulk offered by Polpharma
Find Salicylamide bulk offered by Polpharma
Find Talazoparib bulk offered by Polpharma
Find Ticagrelor bulk offered by Polpharma
Find Topiramate bulk offered by Polpharma
Find Vismodegib bulk offered by Polpharma


 Polpharma
Polpharma