03 Apr 2025
// INDPHARMAPOST
27 Jan 2025
// PRESS FRELEASE
24 Jan 2025
// BUSINESS STD
 KEY PRODUCTS
KEY PRODUCTS
Delivering quality and niche Active Pharmaceutical Ingredients across the global from our USFDA-approved facility.
About
CPhI North America CPhI North America
Industry Trade Show
Not Confirmed
20-22 May, 2025
BIO International Conv...BIO International Convention
Industry Trade Show
Attending
16-19 June, 2025
Industry Trade Show
Attending
28-30 August, 2025
CONTACT DETAILS




Events
Webinars & Exhibitions
CPhI North America CPhI North America
Industry Trade Show
Not Confirmed
20-22 May, 2025
BIO International Conv...BIO International Convention
Industry Trade Show
Attending
16-19 June, 2025
Industry Trade Show
Attending
28-30 August, 2025
CORPORATE CONTENT #SupplierSpotlight
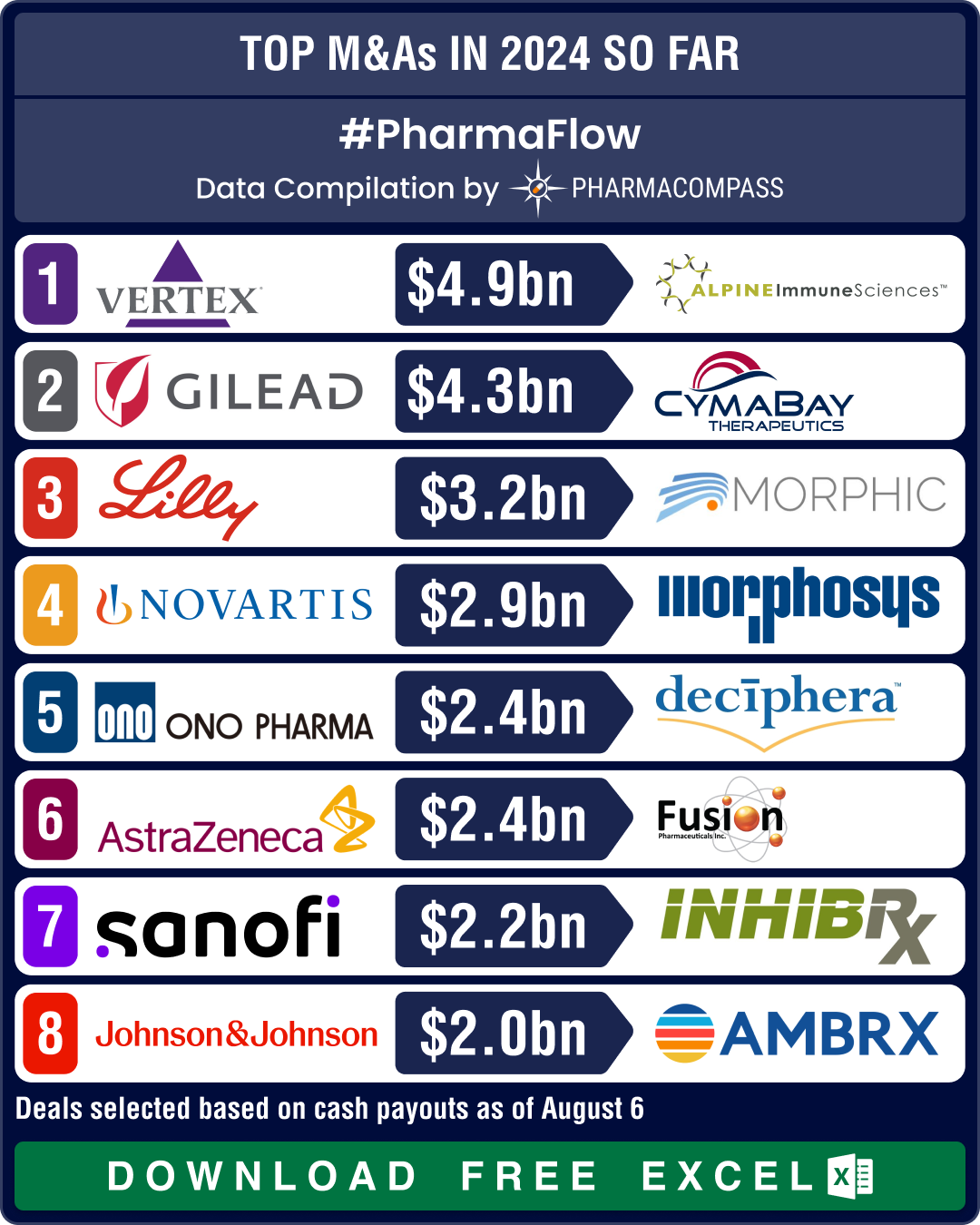
https://www.pharmacompass.com/radio-compass-blog/novartis-gsk-sanofi-bms-shell-out-over-us-10-bn-in-dealmaking-as-mid-size-deals-take-centerstage-in-2024

03 Apr 2025
// INDPHARMAPOST
https://www.indianpharmapost.com/news/mankind-unveils-healthoks-100-vegetarian-multivitamin-tablets-campaign-with-archana-puran-singh-16968

27 Jan 2025
// PRESS FRELEASE
https://www.mankindpharma.com/media/press-release/mankind-pharma-launches-45-day-healthcare-mission-at-mahakumbh-serving-thousands-of-pilgrims-with-free-health-check-ups

24 Jan 2025
// BUSINESS STD
https://www.business-standard.com/markets/capital-market-news/mankind-pharma-slides-after-q3-pat-slips-16-yoy-to-rs-380-cr-125012400521_1.html

24 Jan 2025
// BUSINESS STD
https://www.business-standard.com/markets/news/mankind-pharma-shares-crack-6-after-q3-profit-drops-16-know-more-125012400322_1.html

24 Jan 2025
// BUSINESS STD
https://www.business-standard.com/markets/capital-market-news/mankind-pharma-consolidated-net-profit-declines-16-20-in-the-december-2024-quarter-125012400174_1.html

23 Jan 2025
// BUSINESS STD
https://www.business-standard.com/companies/news/mankind-pharma-q3fy25-results-pat-falls-16-5-revenue-rises-24-125012301463_1.html
Inspections and registrations
ABOUT THIS PAGE
Mankind Pharma is a supplier offers 45 products (APIs, Excipients or Intermediates).
Find a price of Atorvastatin bulk with DMF, CEP offered by Mankind Pharma
Find a price of Clomipramine Hydrochloride bulk with DMF, CEP offered by Mankind Pharma
Find a price of Haloperidol bulk with DMF, CEP offered by Mankind Pharma
Find a price of Haloperidol Decanoate bulk with DMF, CEP offered by Mankind Pharma
Find a price of Nitrofurantoin bulk with DMF, CEP offered by Mankind Pharma
Find a price of Pantoprazole Sodium bulk with DMF, CEP offered by Mankind Pharma
Find a price of Propranolol Hydrochloride bulk with DMF, CEP offered by Mankind Pharma
Find a price of Rosuvastatin Calcium bulk with DMF, CEP offered by Mankind Pharma
Find a price of Ticagrelor bulk with DMF, CEP offered by Mankind Pharma
Find a price of Atorvastatin bulk with DMF offered by Mankind Pharma
Find a price of Baclofen bulk with DMF offered by Mankind Pharma
Find a price of Cilnidipine bulk with DMF offered by Mankind Pharma
Find a price of Clozapine bulk with DMF offered by Mankind Pharma
Find a price of Midodrine bulk with DMF offered by Mankind Pharma
Find a price of Pantoprazole Sodium bulk with DMF offered by Mankind Pharma
Find a price of Ranolazine bulk with DMF offered by Mankind Pharma
Find a price of Revefenacin bulk with DMF offered by Mankind Pharma
Find a price of Rocuronium Bromide bulk with DMF offered by Mankind Pharma
Find a price of Sacubitril Sodium bulk with DMF offered by Mankind Pharma
Find a price of Sacubitril-Valsartan bulk with DMF offered by Mankind Pharma
Find a price of Sitagliptin Phosphate bulk with DMF offered by Mankind Pharma
Find a price of Succinylcholine Chloride bulk with DMF offered by Mankind Pharma
Find a price of Sugammadex Sodium bulk with DMF offered by Mankind Pharma
Find a price of Telmisartan bulk with DMF offered by Mankind Pharma
Find a price of Valsartan bulk with DMF offered by Mankind Pharma
Find a price of EMPAGLIFLOZIN (PROCESS II) bulk with DMF offered by Mankind Pharma
Find a price of Brivaracetam bulk offered by Mankind Pharma
Find a price of Dydrogesterone bulk offered by Mankind Pharma
Find a price of Empagliflozin bulk offered by Mankind Pharma
Find a price of Ensifentrine bulk offered by Mankind Pharma
Find a price of Etoricoxib bulk offered by Mankind Pharma
Find a price of Fezolinetant bulk offered by Mankind Pharma
Find a price of Lasmiditan bulk offered by Mankind Pharma
Find a price of Latanoprostene Bunod bulk offered by Mankind Pharma
Find a price of Lifitegrast bulk offered by Mankind Pharma
Find a price of Netarsudil bulk offered by Mankind Pharma
Find a price of Netarsudil Mesylate bulk offered by Mankind Pharma
Find a price of Obeticholic Acid bulk offered by Mankind Pharma
Find a price of Perfluorohexyloctane bulk offered by Mankind Pharma
Find a price of Resmetirom bulk offered by Mankind Pharma
Find a price of Semaglutide bulk offered by Mankind Pharma
Find a price of Suzetrigine bulk offered by Mankind Pharma
Find a price of Upadacitinib bulk offered by Mankind Pharma
Find a price of Ursodeoxycholic Acid bulk offered by Mankind Pharma
Find a price of Xanomeline bulk offered by Mankind Pharma