
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Europe
0
Australia
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Bol 303259-x
2. Bol-303259-x
3. Bol303259-x
4. Ncx 116
5. Ncx-116
6. Ncx116
7. Pf 3187207
8. Pf-3187207
9. Pf3187207
1. Vyzulta
2. 860005-21-6
3. Pf-3187207
4. Bol-303259-x
5. Ncx 116
6. Ncx-116
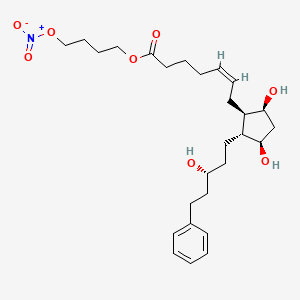
7. 4-nitrooxybutyl (z)-7-[(1r,2r,3r,5s)-3,5-dihydroxy-2-[(3r)-3-hydroxy-5-phenylpentyl]cyclopentyl]hept-5-enoate
8. I6393o0922
9. 4-(nitrooxy)butyl (5z)-7-((1r,2r,3r,5s)-3,5-dihydroxy-2-((3r)-3-hydroxy-5-phenylpentyl)cyclopentyl)hept-5-enoate
10. Vesneo
11. Latanoprostene Bunod [inn]
12. Unii-i6393o0922
13. Latanoprostene Bunod [usan:inn]
14. Lbn
15. Vyzulta (tn)
16. Lbnncx116
17. Gtpl9635
18. Ncx116
19. Chembl2364612
20. Schembl12119560
21. Latanoprostene Bunod [mi]
22. Chebi:177703
23. Latanoprostene Bunod (usan/inn)
24. Dtxsid101027765
25. Latanoprostene Bunod [usan]
26. Bcp29385
27. Latanoprostene Bunod [who-dd]
28. Db11660
29. Hy-19518
30. Latanoprostene Bunod [orange Book]
31. Cs-0015617
32. D10441
33. Q27280492
| Molecular Weight | 507.6 g/mol |
|---|---|
| Molecular Formula | C27H41NO8 |
| XLogP3 | 4.4 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 18 |
| Exact Mass | 507.28321727 g/mol |
| Monoisotopic Mass | 507.28321727 g/mol |
| Topological Polar Surface Area | 142 Ų |
| Heavy Atom Count | 36 |
| Formal Charge | 0 |
| Complexity | 646 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 5 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 1 | |
|---|---|
| Drug Name | VYZULTA |
| Active Ingredient | LATANOPROSTENE BUNOD |
| Company | BAUSCH AND LOMB (Application Number: N207795. Patents: 6211233, 7273946, 7629345, 7910767, 8058467) |
Latanoprostene bunod opthalmic solution is indicated for the reduction of intraocular pressure in patients with open-angle glaucoma or ocular hypertension.
FDA Label
Upon applying an appropriate dose of latanoprost bunod, reduction in intraocular pressure begins approximately 1 to 3 hours later with a maximum intraocular pressure reduction effect demonstrated after 11 to 13 hours.
S - Sensory organs
S01 - Ophthalmologicals
S01E - Antiglaucoma preparations and miotics
S01EE - Prostaglandin analogues
S01EE06 - Latanoprostene bunod
Absorption
In a study with 22 healthy subjects monitored for 28 days, there were no quantifiable plasma concentrations of latanoprostene bunod (Lower Limit Of Quantitation, LLOQ, of 10.0 pg/mL) or butanediol mononitrate (LLOQ of 200 pg/mL) post daily dose of one drop bilaterally in the morning on Day 1 and 28. The mean time of maximum plasma concentration (Tmax) for latanoprost acid was about 5 minutes post dosage on both Day 1 and 28 of therapy. The mean maximum plasma concentrations (Cmax) of latanoprost acid (LLOQ of 30 pg/mL) were 59.1 pg/mL on Day 1 and 28, respectively.
Route of Elimination
The latanoprost acid component of latanoprostene bunod is predominantly metabolized by the liver and excreted primarily in the urine.
Volume of Distribution
Unfortunately there have been no formal ocular distribution studies performed in humans at this time.
Clearance
Since latanoprost acid plasma concentration dropped below the LLOQ (Lower Limit Of Quantitation) of 30 pg/mL in the majority of study subjects by 15 minutes following ordinary ocular administration, the elimination of latanoprost acid from human plasma is considered rapid.
Upon topical administration at the ocular surface, latanoprostene bunod undergoes rapid carboxyl ester hydrolysis by endogenous corneal esterases into latanoprost acid and butanediol mononitrate. After the latanoprost acid reaches the systemic circulation, it is largely metabolized by the liver to the 1,2-dinor and 1,2,3,4-tetranor metabolites by way of fatty acid beta-oxidation. The butanediol monohidrate undergoes further metabolism (reduction) to 1,4-butanediol and nitric oxide (NO). Furthermore, this 1,4-butanediol metabolite is further oxidized to succinic acid that is subsequently then primarily taken up as a component in the tricarboxylic acid (TCA) cycle in cellular aerobic respiration.
The half-life after application of latanoprostene bunod in rabbits was 1.8 hours in cornea, 2.1 hours in aqueous humor, and 4.6 hours in the iris/ciliary body.
Open-angle glaucoma (OAG) is a medical condition that is associated with progressive visual field damage and the loss of vision. Occular hypertension (OHT) is considered a key risk factor for OAG and reducing intraocular pressure (IOP) and being able to maintain unique and appropriate target IOPs for various different patients having OHT can delay or prevent the onset of primary OAG or slow the disease progression of established glaucoma. Ordinary physiological IOP results from aqueous humor produced by the ocular ciliary body and its outflow through a) the trabecular meshwork (TM) and Schlemm's canal (SC) in what is called the conventional pathway, and b) the uveoscleral pathway via the ciliary muscle/choroid/sclera in what is refered to as the unconventional pathway. In patients with OHT or OAG there is increased resistance to aqueous humor outflow by way of the TM/SC pathway, which causes increased IOP. This increase in IOP is believed to be the cause of mechanical stress on the posterior structures of the eye which can result in the dysfunction of optic nerve fibers and the destruction of retinal ganglion cells - all of which ultimately contributes to vision loss. As there is no cure for glaucoma, therapeutic management is predominantly focused on minimizing disease progression and clinical sequelae via the reduction and maintainenance of appropriate target IOPs. Subsequently, latanoprostene bunod is thought to lower intraocular pressure via a dual mechanism of action since the medication is metabolized into two relevant moieties upon administration: (1) latanoprost acid, and (2) butanediol mononitrate. As a prostaglandin F2-alpha analog, the latanoprost acid moiety operates as a selective PGF2-alpha (FP) receptor agonist. Since FP receptors occur in the ciliary muscle, ciliary epithelium, and sclera the latanoprost acid moiety primarily acts in the uveoscleral pathway where it increases the expression of matrix metalloproteinases (MMPs) like MMP-1, -3, and -9 which promote the degradation of collagen types I, III, and IV in the longitudinal bundles of the ciliary musicle and surrounding sclera. The resultant extracellular matrix remodeling of the ciliary muscle consequently produces reduced outflow resistance via increased permeability and increased aqueous humor outflow through the uveoscleral route. Conversely, the butanediol mononitrate undergoes further metabolism to NO and an inactive 1,4-butanediol moiety. As a gas that can freely diffuse across plasma membranes, it is proposed that the relaxing effect of NO to induce reductions in the cell volume and contractility of vascular smooth muscle like cells is dependant upon activation of the sGC/cGMP/PKG cascade pathway. NO released from butanediol mononitrate consequently enters the cells of the TM and inner wall of SC, causing decreases in myosin light chain-2 phosphorylation, increased phosphorylation of large-conductance calcium-activated potassium (BKCa) channels, and a subsequent efflux of potassium ions through such BKCa channels. All of these changes serve to decrease the cell contractility and volume, as well as to rearrange the actin cytoskeleton of the TM and SC cells. These biomechanical changes ultimately allow for enhanced conventional outflow of aqueous humor.
 Click Us!
Click Us! 
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 37976
Submission : 2023-04-24
Status : Active
Type : II

GDUFA
DMF Review : Reviewed
Rev. Date : 2024-01-24
Pay. Date : 2023-04-27
DMF Number : 37429
Submission : 2023-05-30
Status : Active
Type : II

Registrant Name : Clasia Co., Ltd.
Registration Date : 2024-01-15
Registration Number : Su758-10-ND
Manufacturer Name : Cayman Pharma sro
Manufacturer Address : Práce 657, 277 11 Neratovice, Czech Republic

 Century has been an API manufacturer for over 40 years & is the partner of choice for multipurpose custom manufacturing projects.
Century has been an API manufacturer for over 40 years & is the partner of choice for multipurpose custom manufacturing projects.
 Delivering quality and niche Active Pharmaceutical Ingredients across the global from our USFDA-approved facility.
Delivering quality and niche Active Pharmaceutical Ingredients across the global from our USFDA-approved facility.

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 42791
Submission : 2025-11-18
Status : Active
Type : II

Registrant Name : Sungjin Exim Co., Ltd.
Registration Date : 2025-11-07
Registration Number : Su133-23-ND
Manufacturer Name : NEWCHEM SPA
Manufacturer Address : Via Parco del Ticino, 10-27100 San Martino Siccomario-Pavia Italy



GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39153
Submission : 2023-12-27
Status : Active
Type : II



GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 40478
Submission : 2024-09-27
Status : Active
Type : II

NDC Package Code : 40016-015
Start Marketing Date : 2024-12-17
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1g/g)
Marketing Category : BULK INGREDIENT

Registrant Name : YS Life Science Co., Ltd.
Registration Date : 2024-10-09
Registration Number : 2247-1-ND
Manufacturer Name : YS Life Science Co., Ltd.
Manufacturer Address : 207 Sujeong-ro, Jangan-myeon, Hwaseong-si, Gyeonggi-do



GDUFA
DMF Review : Reviewed
Rev. Date : 2020-12-16
Pay. Date : 2020-09-30
DMF Number : 34033
Submission : 2019-08-01
Status : Active
Type : II

Date of Issue : 2025-09-19
Valid Till : 2028-05-05
Written Confirmation Number : WC-0349
Address of the Firm :

NDC Package Code : 54893-0091
Start Marketing Date : 2019-08-01
End Marketing Date : 2027-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Registrant Name : MPK Korea Co., Ltd.
Registration Date : 2025-02-28
Registration Number : Su812-25-ND
Manufacturer Name : MSN Laboratories Private Limited
Manufacturer Address : (Unit-ll) Sy. No. 50, 53, 53/A, 54 & 54/A, Kardanur (Village), Patancheru (Mandal), Sangareddy District, Telangana, Pincode: 502 300, India.



 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
57
PharmaCompass offers a list of Latanoprostene Bunod API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Latanoprostene Bunod manufacturer or Latanoprostene Bunod supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Latanoprostene Bunod manufacturer or Latanoprostene Bunod supplier.
A Latanoprostene Bunod manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Latanoprostene Bunod, including repackagers and relabelers. The FDA regulates Latanoprostene Bunod manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Latanoprostene Bunod API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Latanoprostene Bunod manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Latanoprostene Bunod supplier is an individual or a company that provides Latanoprostene Bunod active pharmaceutical ingredient (API) or Latanoprostene Bunod finished formulations upon request. The Latanoprostene Bunod suppliers may include Latanoprostene Bunod API manufacturers, exporters, distributors and traders.
click here to find a list of Latanoprostene Bunod suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Latanoprostene Bunod DMF (Drug Master File) is a document detailing the whole manufacturing process of Latanoprostene Bunod active pharmaceutical ingredient (API) in detail. Different forms of Latanoprostene Bunod DMFs exist exist since differing nations have different regulations, such as Latanoprostene Bunod USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Latanoprostene Bunod DMF submitted to regulatory agencies in the US is known as a USDMF. Latanoprostene Bunod USDMF includes data on Latanoprostene Bunod's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Latanoprostene Bunod USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Latanoprostene Bunod suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Latanoprostene Bunod Drug Master File in Korea (Latanoprostene Bunod KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Latanoprostene Bunod. The MFDS reviews the Latanoprostene Bunod KDMF as part of the drug registration process and uses the information provided in the Latanoprostene Bunod KDMF to evaluate the safety and efficacy of the drug.
After submitting a Latanoprostene Bunod KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Latanoprostene Bunod API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Latanoprostene Bunod suppliers with KDMF on PharmaCompass.
A Latanoprostene Bunod written confirmation (Latanoprostene Bunod WC) is an official document issued by a regulatory agency to a Latanoprostene Bunod manufacturer, verifying that the manufacturing facility of a Latanoprostene Bunod active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Latanoprostene Bunod APIs or Latanoprostene Bunod finished pharmaceutical products to another nation, regulatory agencies frequently require a Latanoprostene Bunod WC (written confirmation) as part of the regulatory process.
click here to find a list of Latanoprostene Bunod suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Latanoprostene Bunod as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Latanoprostene Bunod API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Latanoprostene Bunod as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Latanoprostene Bunod and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Latanoprostene Bunod NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Latanoprostene Bunod suppliers with NDC on PharmaCompass.
Latanoprostene Bunod Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Latanoprostene Bunod GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Latanoprostene Bunod GMP manufacturer or Latanoprostene Bunod GMP API supplier for your needs.
A Latanoprostene Bunod CoA (Certificate of Analysis) is a formal document that attests to Latanoprostene Bunod's compliance with Latanoprostene Bunod specifications and serves as a tool for batch-level quality control.
Latanoprostene Bunod CoA mostly includes findings from lab analyses of a specific batch. For each Latanoprostene Bunod CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Latanoprostene Bunod may be tested according to a variety of international standards, such as European Pharmacopoeia (Latanoprostene Bunod EP), Latanoprostene Bunod JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Latanoprostene Bunod USP).

