By PharmaCompass
2020-02-13
Impressions: 12950
In 2019, concerns over quality of medicines continued to dominate news headlines. The ‘sartan’ recall saga, which was triggered in July 2018 after the European Medicines Agency (EMA) began reviewing medicines containing valsartan, for the presence of the carcinogenic impurity N-nitrosodimethylamine (NDMA), continued well through the year and had a major impact on the global pharmaceutical industry.
View our Interactive 2019 cGMP Compliance Recap Dashboard (Free Excel Available)
In 2019, French drugmaker Sanofi announced it would recall popular heartburn medicine Zantac in the United States and Canada, after medicines containing the active ingredient ranitidine were also linked with the presence of NDMA.
Several generic drugmakers followed suit and as concerns mounted over cancer-causing impurities in commonly used antacid, diabetes and blood pressure medicines, the EMA’s human medicines committee (CHMP) requested that marketing authorization holders (MAHs) for human medicines containing chemically synthesized active substances review their medicines for the possible presence of nitrosamines and test all products at risk. The review will include all generics and over-the counter (OTC) products.
View our Interactive 2019 cGMP Compliance Recap Dashboard (Free Excel Available)
In Spain, a drug mixup caused children to develop a form of werewolf syndrome (a rare and curious condition that causes excessive hair growth) after they were given a wrong medication to treat heartburn.
Swiss drugmaker Novartis battled data manipulation allegations involving its US$ 2.1 million gene therapy Zolgensma and a new book titled ‘Bottle of Lies’, by investigative journalist Katherine Eban revealed how quality and efficacy of generic drugs is being compromised by companies in India and China, the two main countries that produce these drugs for the US consumer.
The book release sent the FDA in damage control mode, with senior officials issuing statements supporting the agency’s generic drug framework.
Inspection statistics
PharmaCompass reviewed the FDA and EDQM’s inspection statistics for the calendar year 2019 and found that the FDA’s Drug Quality Assurance division conducted 1,138 inspections with 2,280 inspections being performed by the member states of the EDQM.
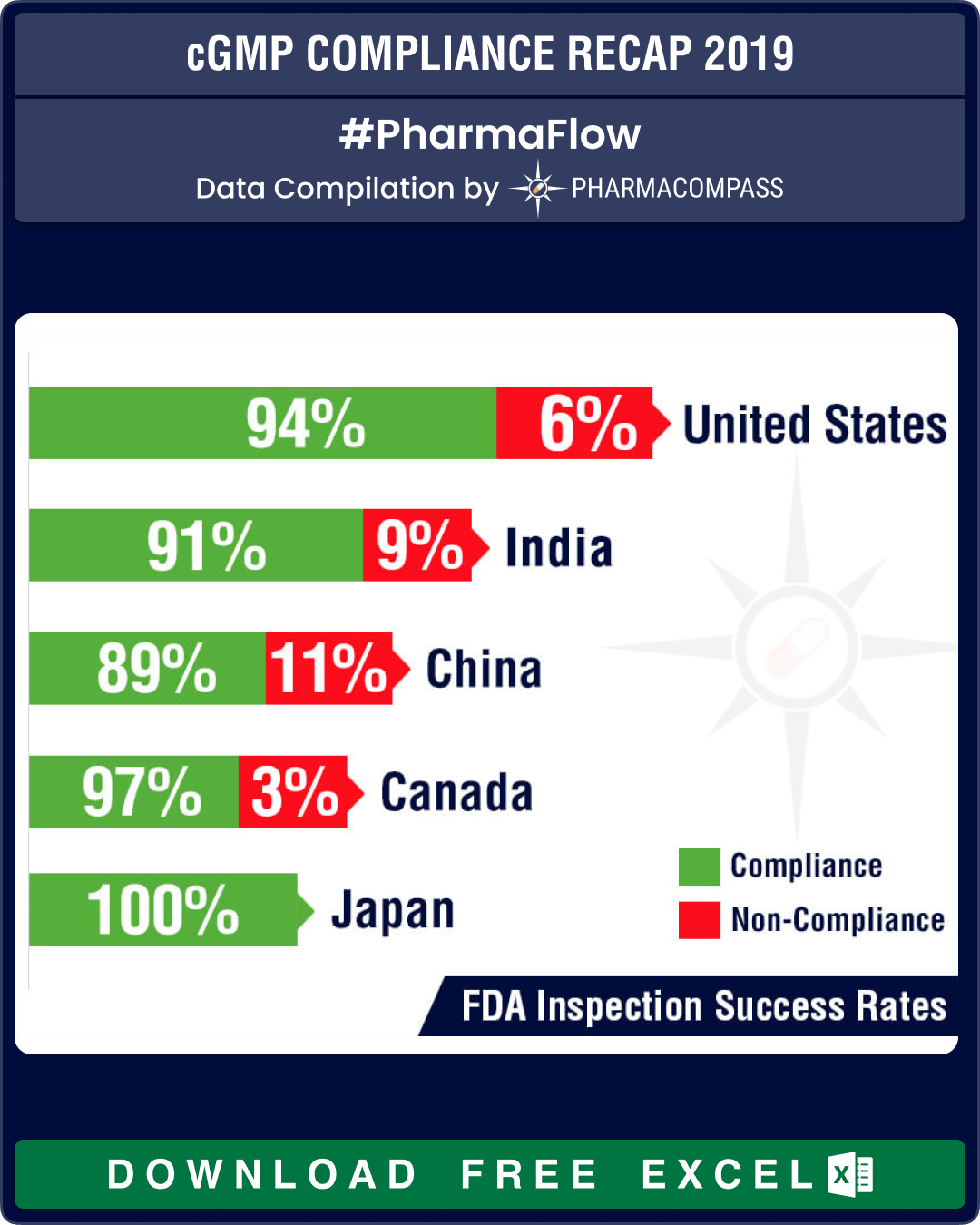
The FDA was the most active in the United States with 478 inspections followed by India where the inspection count was 193 and then in China where the count stood at 117.
With regard to non-compliance, there were 73 inspections which were classified as OAI (6.4 percent of the total number of inspections) by the FDA while the EDQM issued 20 non-compliance reports (0.87 percent of all inspections).
The highest number of facilities which failed to meet FDA’s standards were in the United States (29), followed by India (18) and then China (13). From an inspection success rate perspective, firms in the United States passed 94 percent of their inspections while the success rates in India and China were 91 percent and 89 percent respectively.
View our Interactive 2019 cGMP Compliance Recap Dashboard (Free Excel Available)
FDA investigators have identified persistent challenges while conducting foreign inspections, raising questions about the equivalence of foreign to domestic inspections. For example, while domestic inspections are almost always unannounced, FDA’s practice of pre-announcing foreign inspections up to 12 weeks in advance gives manufacturers the opportunity to fix problems.
A US Government Accountability Office preliminary analysis of FDA data revealed that from fiscal year 2012 through 2016, the number of foreign drug manufacturing establishment inspections increased. However, from fiscal year 2016 through 2018, both foreign and domestic inspections decreased—by about 10 percent and 13 percent, respectively. FDA officials attributed the decline, in part, to difficulty in filling jobs abroad and on the paucity of inspectors.
View our Interactive 2019 cGMP Compliance Recap Dashboard (Free Excel Available)
While the FDA is actively working on increasing the number of inspectors, according to FDA officials, it could take two to three years before the new staff is experienced enough to conduct foreign inspections.
Concerns over manufacturing quality in India re-emerge
In 2019, there was a surge of OAI classifications and FDA warning letters issued to the manufacturing sites of many major Indian pharmaceutical companies. The FDA issued warning letters to Aurobindo Pharma, Cadila Healthcare, Emcure Pharmaceuticals, Glenmark, Indoco Remedies, Jubilant, Lupin, Mylan, Strides and Torrent Pharmaceuticals.
There are more warning letters expected as Form 483s issued after inspections at the facilities of Sun Pharma, Cipla and Lupin continue to reveal problems.
View our Interactive 2019 cGMP Compliance Recap Dashboard (Free Excel Available)
The Form 483 issued by the FDA, following the inspection at Sun Pharma’s Halol facility, highlighted that Sun “failed to establish and implement controls which ensure data integrity” while Cipla’s finished pharmaceuticals manufacturing facility in Goa got classified as Official Action Indicated (OAI) by the FDA following a September 2019 inspection in which the FDA investigators had issued a 38-page Form 483.
Regulatory actions lead to potential supply disruptions into US
These regulatory actions have now begun to impact the supplies of generic pharmaceuticals into the United States. Injectable drugs, which constantly feature on the drug shortage list, had Cadila and Pfizer announce the discontinuation of their supplies to the United States. Following FDA’s warning letter to Cadila, the firm informed the FDA that it would permanently cease production of injectable drug products for the United States.
In 2019, Pfizer announced that two manufacturing sites in India, which it had acquired through its US$ 17 billion acquisition of Hospira, will cease manufacturing operations. The sites located near Chennai (Irungattukottai) and Aurangabad employed 1,700 people.
View our Interactive 2019 cGMP Compliance Recap Dashboard (Free Excel Available)
The Irungattukottai site received an FDA warning letter in 2013 and in 2016, Pfizer halted production at the plant after a PIC/S (short for Pharmaceutical Inspection Convention and Pharmaceutical Inspection Co-operation Scheme) joint inspection with regulators from four international agencies — MHRA (Medicines and Healthcare products Regulatory Agency of the UK), USFDA (United States Food and Drug Administration), TGA (Therapeutic Goods Administration of Australia) and Health Canada — found various quality control problems.
Firms like Vital Laboratories and Alchymars ICM SM Private Limited, which had been issued warning letters in the past, were placed on import alert by the FDA.
Concerns over operations at firms responsible for valsartan recalls
The FDA and EDQM both raised concerns over the operations at Lantech Pharmaceuticals Limited, a firm which undertakes contract solvent recovery for valsartan API manufacturing operations. Solvents recovered by Lantech and samples collected from Lantech’s equipment were found to contain mutagenic impurities.
Inspections at Lantech revealed that the firm failed to implement a procedure for investigating unknown peaks in recovered solvent chromatograms observed during analytical testing — an oversight which had led to the cancer-causing impurities not being detected in the ‘sartan’ APIs.
View our Interactive 2019 cGMP Compliance Recap Dashboard (Free Excel Available)
The FDA also raised data-integrity concerns at the facility as it was “routinely deleting recovered solvents gas chromatography (GC) data older than three months permanently, without any backup” and adequate controls.
As a fallout of the “sartan” recall, warning letters were issued to Mylan and Jubilant over their valsartan manufacturing operations.
Novartis’ data manipulation scandal
In a shocking announcement, the US Food and Drug Administration (FDA) issued a press release on data accuracy issues with Novartis’ gene therapy — Zolgensma.
Zolgensma was acquired by Novartis in April 2018 when in a bid to secure its leadership position in gene therapy, Novartis struck a deal to acquire Illinois-based AveXis Inc for US$ 8.7 billion.
The gene therapy, intended to treat children less than two years of age with the most severe form of spinal muscular atrophy (SMA), is priced at US$ 2.1 million, making it the world’s most expensive drug.
View our Interactive 2019 cGMP Compliance Recap Dashboard (Free Excel Available)
On May 24, 2019, the FDA approved Zolgensma and a month later, on June 28, the agency was informed by AveXis (the product's manufacturer) that its personnel had manipulated data from an in-vivo murine potency assay.
The FDA used this information to evaluate product comparability and nonclinical (animal) pharmacology as part of the biologics license application (BLA), which was submitted and reviewed by the FDA.
According to information shared by the FDA, the product that was administered in the Phase 1 clinical trial was manufactured by a different process than the product that was administered in the Phase 3 clinical trial and the animal toxicology studies.
Because the manufacturing processes were different, interpretation of the overall clinical trial and nonclinical study results depends on understanding the characteristics of the Phase 1 version of the product in relationship to the characteristics of the Phase 3 version of the product.
View our Interactive 2019 cGMP Compliance Recap Dashboard (Free Excel Available)
AveXis’ investigation report revealed that the firm became aware of the data manipulation as early as on March 14, 2019, more than two months prior to the BLA approval. However, AveXis did not inform the FDA of the issue until over a month after the BLA approval. If AveXis had informed FDA of this issue prior to the BLA approval, there was a possibility that the approval would have been delayed beyond the PDUFA goal date of May 31, 2019.
The FDA assessment, however, stated that although the BLA would have eventually been approved, it is carefully assessing this situation and remains confident that Zolgensma should remain on the market.
While managing the crisis, Novartis CEO Vas Narasimhan acknowledged that the company could have handled the furor surrounding Zolgensma better.
Novartis’ AveXis fired its former chief scientific officer Brian Kaspar in connection with the data manipulation scandal, although Kaspar’s lawyer said he did nothing wrong and is ready to defend his name as needed.
The Swiss drugmaker could face possible civil or criminal penalties, the FDA has said.
Our view
Over the past years, PharmaCompass has provided on-time coverage of quality concerns that have emerged at manufacturing operations around the world. With the widespread use of generic drugs and the majority of them being manufactured in India and China, data-integrity within manufacturing operations and assurance of product quality continue to remain key concerns.
View our Interactive 2019 cGMP Compliance Recap Dashboard (Free Excel Available)
While streamlined, compliant operations still seem like a distant dream, in 2019, the former promoters of Ranbaxy, the Singh brothers were finally arrested for misappropriating funds. The legal action was in connection with their stake sale in the Indian drug behemoth to Japanese drugmaker Daiichi Sankyo for US$ 2.4 billion. The sale took place in 2008, months before the US Food and Drug Administration (FDA) banned imports from two of Ranbaxy’s Indian plants as a result of widespread data falsification by the firm. That was the start of troubles for the duo, as Daiichi later took them to court for allegedly suppressing and misrepresenting facts at the time of sale.
We hope there is a lesson in this for the industry so that companies improve their practices of falsifying data and the focus shifts towards patient safety rather than minimizing cost and maximizing profit.
View our Interactive 2019 cGMP Compliance Recap Dashboard (Free Excel Available)
The PharmaCompass Newsletter – Sign Up, Stay Ahead
Feedback, help us to improve. Click here
Image Credit : cGMP Compliance Recap 2019 by PharmaCompass is licensed under CC BY 2.0
“ The article is based on the information available in public and which the author believes to be true. The author is not disseminating any information, which the author believes or knows, is confidential or in conflict with the privacy of any person. The views expressed or information supplied through this article is mere opinion and observation of the author. The author does not intend to defame, insult or, cause loss or damage to anyone, in any manner, through this article.”







