
Synopsis
Synopsis
0
CEP/COS
0
VMF
0
EDQM
0
USP
0
JP
0
Others
DRUG PRODUCT COMPOSITIONS
Regulatory FDF Prices
NA
0
Weekly News Recap #Phispers
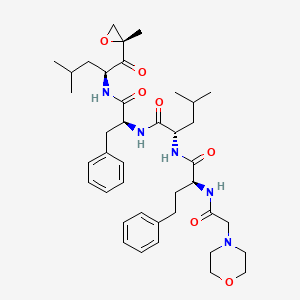

1. (2s)-n-((1s)-1-benzyl-2-(((1s)-3-methyl-1-(((2r)-2-methyloxiran-2-yl)carbonyl)butyl)amino)-2-oxoethyl)-4-methyl-2-(((2s)-2-((morpholin-4-ylacetyl)amino)-4-phenylbutanoyl)amino)pentanamide
2. Kyprolis
3. Pr-171
4. Pr171
1. 868540-17-4
2. Kyprolis
3. Carfilzomib (pr-171)
4. Pr-171
5. Carfilzomib (pr171)
6. Unii-72x6e3j5ar
7. Nsc-758252
8. 72x6e3j5ar
9. Chembl451887
10. Chebi:65347
11. Ncgc00249613-01
12. Dsstox_cid_28616
13. Dsstox_rid_82886
14. Dsstox_gsid_48690
15. (2s)-4-methyl-n-[(2s)-1-[[(2s)-4-methyl-1-[(2r)-2-methyloxiran-2-yl]-1-oxopentan-2-yl]amino]-1-oxo-3-phenylpropan-2-yl]-2-[[(2s)-2-[(2-morpholin-4-ylacetyl)amino]-4-phenylbutanoyl]amino]pentanamide
16. (2s)-n-((1s)-1-benzyl-2-(((1s)-3-methyl-1-(((2r)-2-methyloxiran-2-yl)carbonyl)butyl)amino)-2-oxoethyl)-4-methyl-2-(((2s)-2-((morpholin-4-ylacetyl)amino)-4-phenylbutanoyl)amino)pentanamide
17. (alphas)-alpha-[[2-(4-morpholinyl)acetyl]amino]benzenebutanoyl-l-leucyl-n-[(1s)-3-methyl-1-[[(2r)-2-methyl-2-oxiranyl]carbonyl]butyl]-l-phenylalaninamide
18. (s)-4-methyl-n-((s)-1-(((s)-4-methyl-1-((r)-2-methyloxiran-2-yl)-1-oxopentan-2-yl)amino)-1-oxo-3-phenylpropan-2-yl)-2-((s)-2-(2-morpholinoacetamido)-4-phenylbutanamido)pentanamide
19. N-{(2s)-2-[(morpholin-4-ylacetyl)amino]-4-phenylbutanoyl}-l-leucyl-n-{(2s)-4-methyl-1-[(2r)-2-methyloxiran-2-yl]-1-oxopentan-2-yl}-l-phenylalaninamide
20. (s)-4-methyl-n-((s)-1-((s)-4-methyl-1-((r)-2-methyloxiran-2-yl)-1-oxopentan-2-ylamino)-1-oxo-3-phenylpropan-2-yl)-2-((s)-2-(2-morpholinoacetamido)-4-phenylbutanamido)pentanamide
21. Carfilzomib [usan]
22. Cas-868540-17-4
23. Carfilzomib [usan:inn]
24. Mfcd11040997
25. Kyprolis (tn)
26. Carfilzomib; Pr171
27. Carfilzomib [mi]
28. Carfilzomib [inn]
29. Carfilzomib [jan]
30. Carfilzomib [vandf]
31. Schembl85165
32. Carfilzomib [who-dd]
33. Mls006011102
34. Carfilzomib (jan/usan/inn)
35. Gtpl7420
36. Dtxsid4048690
37. Amy4357
38. Carfilzomib [orange Book]
39. Ex-a2037
40. Ono-7057
41. Tox21_113079
42. Bdbm50277889
43. Nsc756640
44. Nsc758252
45. S2853
46. Zinc49841054
47. Akos025401910
48. Tox21_113079_1
49. Ccg-270405
50. Cs-0984
51. Cs-w004540
52. Db08889
53. Nsc 758252
54. Nsc-756640
55. Ncgc00249613-02
56. Ncgc00249613-03
57. Ncgc00249613-08
58. Ncgc00249613-11
59. Ncgc00249613-13
60. Ac-27051
61. As-17059
62. Hy-10455
63. Smr004660024
64. Sw218090-2
65. D08880
66. Ab01565867_02
67. Sr-01000941582
68. J-501773
69. Sr-01000941582-1
70. Q15366934
71. (alphas)-alpha-((4-morpholinylacetyl)amino)benzenebutanoyl-l-leucyl-n-((1s)-3-methyl-1-(((2r)-2-methyloxiranyl)carbonyl)butyl)-l-phe Nylalaninamide
72. (s)-4-methyl-n-((s)-1-((s)-4-methyl-1-((r)-2- Methyloxiran-2-yl)-1 -oxopentan-2-ylamino)-1-oxo-3-phenylpropan-2-yl)-2-((s)-2-(2-morpholinoacetamido)-4-phenylbutanamido)pentanamide
73. L-phenylalaninamide, (.alpha.s)-.alpha.-((4-morpholinylacetyl)amino)benzenebutanoyl-l-leucyl-n-((1s)-3-methyl-1-(((2r)-2-methyloxiranyl)carbonyl)butyl)-
74. L-phenylalaninamide, (alphas)-alpha-((4-morpholinylacetyl)amino)benzenebutanoyl-l-leucyl-n-((1s)-3-methyl-1-(((2r)-2-methyloxiranyl)carbonyl)butyl)-
| Molecular Weight | 719.9 g/mol |
|---|---|
| Molecular Formula | C40H57N5O7 |
| XLogP3 | 4.7 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 20 |
| Exact Mass | 719.42579917 g/mol |
| Monoisotopic Mass | 719.42579917 g/mol |
| Topological Polar Surface Area | 159 Ų |
| Heavy Atom Count | 52 |
| Formal Charge | 0 |
| Complexity | 1180 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 5 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Kyprolis |
| PubMed Health | Carfilzomib (Injection) |
| Drug Classes | Antineoplastic Agent |
| Drug Label | KYPROLIS (carfilzomib) for Injection is an antineoplastic agent available for intravenous use only. KYPROLIS is a sterile, white to off-white lyophilized powder and is available as a single-use vial. Each vial of KYPROLIS contains 60mg of carfilzom... |
| Active Ingredient | Carfilzomib |
| Dosage Form | Powder |
| Route | Intravenous |
| Strength | 60mg/vial |
| Market Status | Prescription |
| Company | Onyx Pharms |
| 2 of 2 | |
|---|---|
| Drug Name | Kyprolis |
| PubMed Health | Carfilzomib (Injection) |
| Drug Classes | Antineoplastic Agent |
| Drug Label | KYPROLIS (carfilzomib) for Injection is an antineoplastic agent available for intravenous use only. KYPROLIS is a sterile, white to off-white lyophilized powder and is available as a single-use vial. Each vial of KYPROLIS contains 60mg of carfilzom... |
| Active Ingredient | Carfilzomib |
| Dosage Form | Powder |
| Route | Intravenous |
| Strength | 60mg/vial |
| Market Status | Prescription |
| Company | Onyx Pharms |
Carfilzomib is indicated for the treatment of adult patients with relapsed or refractory multiple myeloma who have received one to three lines of therapy in combination with lenalidomide and dexamethasone; or dexamethasone; or daratumumab and dexamethasone; or daratumumab and hyaluronidase-fihj and dexamethasone. It is also indicated as a single agent for the treatment of patients with relapsed or refractory multiple myeloma who have received one or more lines of therapy.
FDA Label
Kyprolis in combination with daratumumab and dexamethasone, with lenalidomide and dexamethasone, or with dexamethasone alone is indicated for the treatment of adult patients with multiple myeloma who have received at least one prior therapy.
Treatment of Multiple Myeloma
Treatment of acute lymphoblastic leukaemia
Intravenous carfilzomib administration resulted in suppression of proteasome chymotrypsin-like activity when measured in blood 1 hour after the first dose. On Day 1 of Cycle 1, proteasome inhibition in peripheral blood mononuclear cells (PBMCs) ranged from 79% to 89% at 15 mg/m2, and from 82% to 83% at 20 mg/m2. In addition, carfilzomib administration resulted in inhibition of the LMP2 and MECL1 subunits of the immunoproteasome ranging from 26% to 32% and 41% to 49%, respectively, at 20 mg/m2. Proteasome inhibition was maintained for 48 hours following the first dose of carfilzomib for each week of dosing. Resistance against carfilzomib has been observed and although the mechanism has not been confirmed, it is thought that up-regulation of P-glycoprotein may be a contributing factor. Furthermore, studies suggest that carfilzomib is more potent than bortezomib.
L01XX45
L01XX45
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
L - Antineoplastic and immunomodulating agents
L01 - Antineoplastic agents
L01X - Other antineoplastic agents
L01XG - Proteasome inhibitors
L01XG02 - Carfilzomib
Absorption
Cmax, single IV dose of 27 mg/m^2 = 4232 ng/mL; AUC, single IV dose of 27 mg/m^2 = 379 nghr/mL; Carfilzomib does not accumulation in the systemic. At doses between 20 and 36 mg/m2, there was a dose-dependent increase in exposure.
Volume of Distribution
Vd, steady state, 20 mg/m^2 = 28 L
Clearance
Systemic clearance = 151 - 263 L/hour. As this value exceeds hepatic blood flow, it suggests that carfilozmib is cleared extrahepatically.
Carfilzomib was rapidly and extensively metabolized by the liver. The predominant metabolites were the peptide fragments and the diol of carfilzomib which suggests that the main metabolic pathways are peptidase cleavage and epoxide hydrolysis. The cytochrome P450 enzyme system is minimally involved in the metabolism of carfilzomib. All metabolites are inactive.
Following intravenous administration of doses 15 mg/m^2, carfilzomib was rapidly cleared from the systemic circulation with a half-life of 1 hour on Day 1 of Cycle 1.
Carfilzomib is made up of four modified peptides and acts as a proteasome inhibitor. Carfilzomib irreversibly and selectively binds to N-terminal threonine-containing active sites of the 20S proteasome, the proteolytic core particle within the 26S proteasome. This 20S core has 3 catalytic active sites: the chymotrypsin, trypsin, and caspase-like sites. Inhibition of the chymotrypsin-like site by carfilzomib (5 and 5i subunits) is the most effective target in decreasing cellular proliferation, ultimately resulting in cell cycle arrest and apoptosis of cancerous cells. At higher doses, carfilzomib will inhibit the trypsin-and capase-like sites.
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
 Click Us!
Click Us! 
GDUFA
DMF Review : Reviewed
Rev. Date : 2016-07-06
Pay. Date : 2016-05-04
DMF Number : 30402
Submission : 2016-03-29
Status : Active
Type : II

Registration Number : 307MF10126
Registrant's Address : 8-2-337, Road No. 3,Banjara Hills,Hyderabad 500 034,TELANGANA,INDIA
Initial Date of Registration : 2025-10-15
Latest Date of Registration :

Date of Issue : 2025-06-20
Valid Till : 2028-07-07
Written Confirmation Number : WC-0039
Address of the Firm :

 Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.

GDUFA
DMF Review : Reviewed
Rev. Date : 2024-09-19
Pay. Date : 2024-08-27
DMF Number : 32406
Submission : 2017-12-30
Status : Active
Type : II

 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
 Chunghwa provides cost-effective APIs & advanced intermediates with complete DMF or COS, ensuring quality & reliable production.
Chunghwa provides cost-effective APIs & advanced intermediates with complete DMF or COS, ensuring quality & reliable production.

NDC Package Code : 52076-6251
Start Marketing Date : 2012-10-16
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
 Omgene: R&D-based biopharmaceutical company with GMP facilities, focused on innovation and high-quality, affordable medicines.
Omgene: R&D-based biopharmaceutical company with GMP facilities, focused on innovation and high-quality, affordable medicines.
 TAPI, a leading global supplier of APIs, provides over 350 products and customized CDMO solutions for every stage of development.
TAPI, a leading global supplier of APIs, provides over 350 products and customized CDMO solutions for every stage of development.

GDUFA
DMF Review : Reviewed
Rev. Date : 2016-02-25
Pay. Date : 2015-12-21
DMF Number : 30124
Submission : 2016-01-21
Status : Active
Type : II


GDUFA
DMF Review : Reviewed
Rev. Date : 2023-07-05
Pay. Date : 2023-05-15
DMF Number : 38290
Submission : 2023-05-16
Status : Active
Type : II



GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 42402
Submission : 2025-08-21
Status : Active
Type : II


GDUFA
DMF Review : Reviewed
Rev. Date : 2024-12-26
Pay. Date : 2024-11-27
DMF Number : 40758
Submission : 2024-11-25
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 38169
Submission : 2023-04-03
Status : Inactive
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
GDUFA
DMF Review : Complete
Rev. Date : 2016-07-06
Pay. Date : 2016-05-04
DMF Number : 30402
Submission : 2016-03-29
Status : Active
Type : II
 Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
GDUFA
DMF Review : Complete
Rev. Date : 2024-09-19
Pay. Date : 2024-08-27
DMF Number : 32406
Submission : 2017-12-30
Status : Active
Type : II
 TAPI, a leading global supplier of APIs, provides over 350 products and customized CDMO solutions for every stage of development.
TAPI, a leading global supplier of APIs, provides over 350 products and customized CDMO solutions for every stage of development.
GDUFA
DMF Review : Complete
Rev. Date : 2016-02-25
Pay. Date : 2015-12-21
DMF Number : 30124
Submission : 2016-01-21
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 30162
Submission : 2015-12-29
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 29750
Submission : 2016-01-30
Status : Inactive
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2020-10-09
Pay. Date : 2020-09-01
DMF Number : 35083
Submission : 2020-09-08
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2016-04-19
Pay. Date : 2016-01-20
DMF Number : 30115
Submission : 2016-03-22
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2015-11-16
Pay. Date : 2015-09-23
DMF Number : 29671
Submission : 2015-08-29
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 30357
Submission : 2016-02-26
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2016-02-05
Pay. Date : 2015-09-25
DMF Number : 29770
Submission : 2015-09-28
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Registration Number : 307MF10126
Registrant's Address : 8-2-337, Road No. 3,Banjara Hills,Hyderabad 500 034,TELANGANA,INDIA
Initial Date of Registration : 2025-10-15
Latest Date of Registration : 2025-10-15
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Date of Issue : 2025-06-20
Valid Till : 2028-07-07
Written Confirmation Number : WC-0039
Address of the Firm : UNIT-I, Plot Nos. 137, 138, 145 & 146, Sri Venkateswara Co-operative Industrial ...
Date of Issue : 2025-11-12
Valid Till : 2028-11-11
Written Confirmation Number : WC-0115
Address of the Firm : D-35, Industrial Area, Kalyani, Dist-Nadia-741235, West Bengal

Date of Issue : 2025-09-19
Valid Till : 2028-05-05
Written Confirmation Number : WC-0349
Address of the Firm : Unit-II, Sy. Nos. 50, 53, 53/A, 54 & 54/A,Kardanur (Village), Patancheru (Mandal...

Date of Issue : 2023-11-15
Valid Till : 2026-05-11
Written Confirmation Number : WC-0273
Address of the Firm : Plot No: 34A, Road No: 1, Jawaharlal Nehru Pharma City, Thanam Village, Parawada...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Registrant Name : Amgen Korea Co., Ltd.
Registration Date : 2016-08-18
Registration Number : Su36-1-ND
Manufacturer Name : Sterling Wisconsin, LLC
Manufacturer Address : W130 N10497 Washington Drive Germantown, WI 53022-4448, USA

Registrant Name : Amgen Korea Co., Ltd.
Registration Date : 2020-09-11
Registration Number : Su36-3-ND
Manufacturer Name : Amgen Singapore Manufacturin...
Manufacturer Address : 1 Tuas View Drive, Singapore, 637026

Registrant Name : Amgen Korea Co., Ltd.
Registration Date : 2016-08-18
Registration Number : Su36-2-ND
Manufacturer Name : HAS Healthcare Advanced Synt...
Manufacturer Address : Via Industria 24, 6710 Biasca, Switzerland

Registrant Name : Gukjeon Co., Ltd.
Registration Date : 2022-04-25
Registration Number : Su173-39-ND
Manufacturer Name : MSN Laboratories Private Lim...
Manufacturer Address : (Unit-ll) Sy. No. 50, 53, 53/A, 54 & 54/A, Kardanur (Village), Patancheru (Mandal), S...

Registrant Name : Masung LS Co., Ltd.
Registration Date : 2024-04-16
Registration Number : Su163-33-ND
Manufacturer Name : Sionc Pharmaceuticals Pvt. L...
Manufacturer Address : Plot No.34A, Road No.1, Jawaharlal Nehru Pharma City, Thanam(V), Parawada Mandal, Vis...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
About the Company : Founded in 1984, DRL is well-known for its generic APIs and its track record in drug product development. It is one of the earliest pharma API manufacturers with a diverse portfoli...
 Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
About the Company : Biophore, founded in 2007, develops and manufactures niche and complex pharmaceutical products. With USFDA- and EU-approved API facilities, a dedicated intermediates site and an R&...
 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
About the Company : LGM Pharma is a global leader in sourcing APIs, including hard-to-find drug substances, for pharmaceutical and biotech industries. LGM also operates as a full-service drug product ...
 Chunghwa provides cost-effective APIs & advanced intermediates with complete DMF or COS, ensuring quality & reliable production.
Chunghwa provides cost-effective APIs & advanced intermediates with complete DMF or COS, ensuring quality & reliable production.
About the Company : Chunghwa Chemical Synthesis & Biotech Co. Ltd. (CCSB) offers cost-effective APIs with speed, supported by a skilled team. As the first Southeast Asian company to receive FDA approv...
 Omgene: R&D-based biopharmaceutical company with GMP facilities, focused on innovation and high-quality, affordable medicines.
Omgene: R&D-based biopharmaceutical company with GMP facilities, focused on innovation and high-quality, affordable medicines.
About the Company : Omgene Life Sciences Pvt. Ltd. is an R&D-driven biopharmaceutical company specializing in biopharmaceuticals, peptides, semi-synthetic, and synthetic actives. As a vertically integ...
 TAPI, a leading global supplier of APIs, provides over 350 products and customized CDMO solutions for every stage of development.
TAPI, a leading global supplier of APIs, provides over 350 products and customized CDMO solutions for every stage of development.
About the Company : Founded in 1935, TAPI Technology & API Services has a long legacy of advancing health through innovation. Today, we offer one of the industry’s most comprehensive API portfolios ...
About the Company : Apicore LLC, a wholly owned subsidiary of RK Pharma Inc is a leading process R&D and API manufacturing service provider for the worldwide pharmaceutical industry. We offer a wide p...

About the Company : Laurus Labs is a leading research and development-driven pharmaceutical company in India. The company has grown consistently to become one of the leading manufacturers of Active Ph...

About the Company : Established in 2010, Mac-Chem is a NMM Group company. Mac-Chem is focused on oncology and other niche specialty APIs. It supplies quality APIs to the top 15 oncology players in Ind...

About the Company : Teva was established in 1901. Our global headquarters are based in Israel. Today we have a portfolio of more than 3,500 medicines, and produce approximately 120 billion tablets and...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Details:
Selinexor is a cytotoxic drug drug, which is currently being evaluated in Phase I/ Phase II clinical studies for the treatment of multiple myeloma.
Lead Product(s): Selinexor,Carfilzomib,Isatuximab,Dexamethasone
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: Phase I/ Phase IIProduct Type: Cytotoxic Drug
Sponsor: Natalie Callander | Karyopharm Therapeutics | University of Wisconsin, Madison
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable March 18, 2026
Lead Product(s) : Selinexor,Carfilzomib,Isatuximab,Dexamethasone
Therapeutic Area : Oncology
Highest Development Status : Phase I/ Phase II
Partner/Sponsor/Collaborator : Natalie Callander | Karyopharm Therapeutics | University of Wisconsin, Madison
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Selinexor is a cytotoxic drug drug, which is currently being evaluated in Phase I/ Phase II clinical studies for the treatment of multiple myeloma.
Product Name : Undisclosed
Product Type : Cytotoxic Drug
Upfront Cash : Inapplicable
March 18, 2026
Details:
Isatuximab is a Antibody drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Multiple Myeloma.
Lead Product(s): Isatuximab,Montelukast Sodium,Dexamethasone,Paracetamol,Diphenhydramine,Methylprednisolone,Carfilzomib
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: Phase IIProduct Type: Antibody, Unconjugated
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable April 10, 2024
Lead Product(s) : Montelukast Sodium, Dexamethasone, Paracetamol, Diphenhydramine, Methylprednisolone
Therapeutic Area : Oncology
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Isatuximab is a Antibody drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Multiple Myeloma.
Product Name : Undisclosed
Product Type : Antibody, Unconjugated
Upfront Cash : Inapplicable
April 10, 2024
Details:
Isatuximab is a Antibody drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Multiple Myeloma.
Lead Product(s): Isatuximab,Carfilzomib,Dexamethasone,Montelukast Sodium,Paracetamol,Diphenhydramine,Methylprednisolone
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: Phase IIProduct Type: Antibody, Unconjugated
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable January 30, 2023
Lead Product(s) : Carfilzomib, Dexamethasone, Montelukast Sodium, Paracetamol, Diphenhydramine
Therapeutic Area : Oncology
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Isatuximab is a Antibody drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Multiple Myeloma.
Product Name : Undisclosed
Product Type : Antibody, Unconjugated
Upfront Cash : Inapplicable
January 30, 2023
Details:
Isatuximab is a Antibody drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Multiple Myeloma.
Lead Product(s): Isatuximab,Cemiplimab,Dexamethasone,Lenalidomide,Pomalidomide,Carfilzomib
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: Phase IIProduct Type: Antibody, Unconjugated
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable January 03, 2023
Lead Product(s) : Isatuximab,Cemiplimab,Dexamethasone,Lenalidomide,Pomalidomide,Carfilzomib
Therapeutic Area : Oncology
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Isatuximab is a Antibody drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Multiple Myeloma.
Product Name : Undisclosed
Product Type : Antibody, Unconjugated
Upfront Cash : Inapplicable
January 03, 2023
Details:
CHMP issues positive opinion for second indication for Sarclisa in combination with carfilzomib and dexamethasone for adult patients with multiple myeloma who have received at least one prior therapy.
Lead Product(s): Isatuximab,Carfilzomib,Dexamethasone
Therapeutic Area: Oncology Brand Name: Sarclisa
Study Phase: Phase IIIProduct Type: Antibody, Unconjugated
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable February 26, 2021
Lead Product(s) : Isatuximab,Carfilzomib,Dexamethasone
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : CHMP issues positive opinion for second indication for Sarclisa in combination with carfilzomib and dexamethasone for adult patients with multiple myeloma who have received at least one prior therapy.
Product Name : Sarclisa
Product Type : Antibody, Unconjugated
Upfront Cash : Inapplicable
February 26, 2021
Details:
Sarclisa added to carfilzomib and dexamethasone (Sarclisa combination) reduced risk of disease progression or death by 47% versus standard of care carfilzomib and dexamethasone (Kd) alone.
Lead Product(s): Isatuximab,Carfilzomib,Dexamethasone
Therapeutic Area: Oncology Brand Name: Sarclisa
Study Phase: Phase IIIProduct Type: Antibody, Unconjugated
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable June 02, 2020
Lead Product(s) : Isatuximab,Carfilzomib,Dexamethasone
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Sarclisa added to carfilzomib and dexamethasone (Sarclisa combination) reduced risk of disease progression or death by 47% versus standard of care carfilzomib and dexamethasone (Kd) alone.
Product Name : Sarclisa
Product Type : Antibody, Unconjugated
Upfront Cash : Inapplicable
June 02, 2020
Details:
Addition of Sarclisa significantly reduced the risk of disease progression or death compared to carfilzomib and dexamethasone alone.
Lead Product(s): Isatuximab,Carfilzomib,Dexamethasone
Therapeutic Area: Oncology Brand Name: Sarclisa
Study Phase: Phase IIIProduct Type: Antibody, Unconjugated
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable May 12, 2020
Lead Product(s) : Isatuximab,Carfilzomib,Dexamethasone
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Sanofi's Sarclisa Cuts Death Risk In Mutiple Myeloma
Details : Addition of Sarclisa significantly reduced the risk of disease progression or death compared to carfilzomib and dexamethasone alone.
Product Name : Sarclisa
Product Type : Antibody, Unconjugated
Upfront Cash : Inapplicable
May 12, 2020
Details:
Isatuximab is a Antibody drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Multiple Myeloma.
Lead Product(s): Isatuximab,Carfilzomib,Dexamethasone
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: Phase IIIProduct Type: Antibody, Unconjugated
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable September 07, 2017
Lead Product(s) : Isatuximab,Carfilzomib,Dexamethasone
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Isatuximab is a Antibody drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Multiple Myeloma.
Product Name : Undisclosed
Product Type : Antibody, Unconjugated
Upfront Cash : Inapplicable
September 07, 2017
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Carfilzomib is a Other Small Molecule drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Multiple Myeloma.
Lead Product(s): Carfilzomib,Venetoclax,Dexamethasone
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: Phase IIProduct Type: Miscellaneous
Sponsor: Genentech
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable September 14, 2016
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Carfilzomib,Venetoclax,Dexamethasone
Therapeutic Area : Oncology
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : Genentech
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Carfilzomib is a Other Small Molecule drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Multiple Myeloma.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
September 14, 2016
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Ibrutinib is a Other Small Molecule drug candidate, which is currently being evaluated in phase I/ phase II clinical studies for the treatment of Multiple Myeloma.
Lead Product(s): Ibrutinib,Carfilzomib,Dexamethasone
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: Phase I/ Phase IIProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable October 14, 2013
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Ibrutinib,Carfilzomib,Dexamethasone
Therapeutic Area : Oncology
Highest Development Status : Phase I/ Phase II
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Ibrutinib is a Other Small Molecule drug candidate, which is currently being evaluated in phase I/ phase II clinical studies for the treatment of Multiple Myeloma.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
October 14, 2013
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results](S)-2-((S)-4-methyl-2-((S)-2-(2-morpholinoacetamid...
CAS Number : 868540-16-3
End Use API : Carfilzomib
About The Company : Cohance Lifesciences is a leading CDMO and API platform delivering products and services across the full molecule lifecycle, from development to commercializati...
(S)-2-amino-4-methyl-1-((R)-2-methyloxiran-2-yl)pe...
CAS Number : 247068-85-5
End Use API : Carfilzomib
About The Company : Cohance Lifesciences is a leading CDMO and API platform delivering products and services across the full molecule lifecycle, from development to commercializati...
CAS Number : 247068-85-5
End Use API : Carfilzomib
About The Company : Founded in 2006, ChemExpress provides CRO and CDMO services to pharmaceutical and biotech companies. The company supports small molecules, biologics, and new mo...
CAS Number : 247068-82-2
End Use API : Carfilzomib
About The Company : Founded in 2006, ChemExpress provides CRO and CDMO services to pharmaceutical and biotech companies. The company supports small molecules, biologics, and new mo...
CAS Number : 868539-98-4
End Use API : Carfilzomib
About The Company : Founded in 2006, ChemExpress provides CRO and CDMO services to pharmaceutical and biotech companies. The company supports small molecules, biologics, and new mo...
CAS Number : 868539-99-5
End Use API : Carfilzomib
About The Company : Founded in 2006, ChemExpress provides CRO and CDMO services to pharmaceutical and biotech companies. The company supports small molecules, biologics, and new mo...
CAS Number : 868540-15-2
End Use API : Carfilzomib
About The Company : Founded in 2006, ChemExpress provides CRO and CDMO services to pharmaceutical and biotech companies. The company supports small molecules, biologics, and new mo...
CAS Number : 1140908-89-9
End Use API : Carfilzomib
About The Company : Founded in 2006, ChemExpress provides CRO and CDMO services to pharmaceutical and biotech companies. The company supports small molecules, biologics, and new mo...
(alphaS)-alpha-[(4-Morpholinylacetyl)aMino]benzene...
CAS Number : 868540-16-3
End Use API : Carfilzomib
About The Company : Established in 2011 and situated in Hangzhou, Zhejiang, China, Hangzhou Longshine Bio-Tech CO., Ltd is dedicated to providing services for pharmaceutical and ch...

Morpholin-4-yl-acetic acid Hydrochloride
CAS Number : 89531-58-8
End Use API : Carfilzomib
About The Company : Established in 2011 and situated in Hangzhou, Zhejiang, China, Hangzhou Longshine Bio-Tech CO., Ltd is dedicated to providing services for pharmaceutical and ch...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Regulatory Info : RX
Registration Country : USA
Brand Name : CARFILZOMIB
Dosage Form : POWDER;INTRAVENOUS
Dosage Strength : 60MG/VIAL
Packaging :
Approval Date : 2019-09-09
Application Number : 209422
Regulatory Info : RX
Registration Country : USA
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Regulatory Info :
Registration Country : India
Brand Name : Carfilzomib
Dosage Form : Injectable
Dosage Strength : 60 mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : DISCN
Registration Country : USA
Brand Name : CARFILZOMIB
Dosage Form : POWDER;INTRAVENOUS
Dosage Strength : 10MG/VIAL
Packaging :
Approval Date : 2021-06-11
Application Number : 209330
Regulatory Info : DISCN
Registration Country : USA
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : DISCN
Registration Country : USA
Brand Name : CARFILZOMIB
Dosage Form : POWDER;INTRAVENOUS
Dosage Strength : 60MG/VIAL
Packaging :
Approval Date : 2021-06-11
Application Number : 209330
Regulatory Info : DISCN
Registration Country : USA
Regulatory Info :
Registration Country : Iran
Brand Name : Filzoma
Dosage Form : Vial
Dosage Strength : 60MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Iran
 Omgene: R&D-based biopharmaceutical company with GMP facilities, focused on innovation and high-quality, affordable medicines.
Omgene: R&D-based biopharmaceutical company with GMP facilities, focused on innovation and high-quality, affordable medicines.
Regulatory Info :
Registration Country : India
Brand Name : Carfilzomib
Dosage Form : Injection
Dosage Strength : 10MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
 Omgene: R&D-based biopharmaceutical company with GMP facilities, focused on innovation and high-quality, affordable medicines.
Omgene: R&D-based biopharmaceutical company with GMP facilities, focused on innovation and high-quality, affordable medicines.
Regulatory Info :
Registration Country : India
Brand Name : Carfilzomib
Dosage Form : Injection
Dosage Strength : 30MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
 Omgene: R&D-based biopharmaceutical company with GMP facilities, focused on innovation and high-quality, affordable medicines.
Omgene: R&D-based biopharmaceutical company with GMP facilities, focused on innovation and high-quality, affordable medicines.
Regulatory Info :
Registration Country : India
Brand Name : Carfilzomib
Dosage Form : Injection
Dosage Strength : 60MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : RX
Registration Country : USA
Brand Name : KYPROLIS
Dosage Form : POWDER;INTRAVENOUS
Dosage Strength : 60MG/VIAL
Packaging :
Approval Date : 2012-07-20
Application Number : 202714
Regulatory Info : RX
Registration Country : USA

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : RX
Registration Country : USA
Brand Name : KYPROLIS
Dosage Form : POWDER;INTRAVENOUS
Dosage Strength : 30MG/VIAL
Packaging :
Approval Date : 2016-06-03
Application Number : 202714
Regulatory Info : RX
Registration Country : USA

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
RLD : No
TE Code : AP
Brand Name : CARFILZOMIB
Dosage Form : POWDER;INTRAVENOUS
Dosage Strength : 60MG/VIAL
Approval Date : 2019-09-09
Application Number : 209422
RX/OTC/DISCN : RX
RLD : No
TE Code : AP
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code :
Brand Name : CARFILZOMIB
Dosage Form : POWDER;INTRAVENOUS
Dosage Strength : 10MG/VIAL
Approval Date : 2021-06-11
Application Number : 209330
RX/OTC/DISCN : DISCN
RLD : No
TE Code :
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code :
Brand Name : CARFILZOMIB
Dosage Form : POWDER;INTRAVENOUS
Dosage Strength : 60MG/VIAL
Approval Date : 2021-06-11
Application Number : 209330
RX/OTC/DISCN : DISCN
RLD : No
TE Code :
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD :
TE Code :
Brand Name : KITPROZY
Dosage Form : VIAL; SINGLE-USE
Dosage Strength : 60MG/ML, 2/ML
Approval Date :
Application Number : 220712
RX/OTC/DISCN :
RLD :
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD :
TE Code :
Brand Name : KITPROZY
Dosage Form : VIAL; SINGLE-USE
Dosage Strength : 120MG/2ML (60MG/ML), 2ML
Approval Date :
Application Number : 220712
RX/OTC/DISCN :
RLD :
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code :
Brand Name : CARFILZOMIB
Dosage Form : POWDER;INTRAVENOUS
Dosage Strength : 30MG/VIAL
Approval Date : 2020-03-20
Application Number : 211185
RX/OTC/DISCN : DISCN
RLD : No
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD :
TE Code :
Brand Name : CARFILZOMIB
Dosage Form : POWDER;INTRAVENOUS
Dosage Strength : 60MG/VIAL
Approval Date :
Application Number : 210823
RX/OTC/DISCN :
RLD :
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD :
TE Code :
Brand Name : CARFILZOMIB
Dosage Form : POWDER
Dosage Strength : 60MG/1VIAL
Approval Date :
Application Number : 219276
RX/OTC/DISCN :
RLD :
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : Yes
TE Code : AP
Brand Name : KYPROLIS
Dosage Form : POWDER;INTRAVENOUS
Dosage Strength : 60MG/VIAL
Approval Date : 2012-07-20
Application Number : 202714
RX/OTC/DISCN : RX
RLD : Yes
TE Code : AP

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : Yes
TE Code :
Brand Name : KYPROLIS
Dosage Form : POWDER;INTRAVENOUS
Dosage Strength : 30MG/VIAL
Approval Date : 2016-06-03
Application Number : 202714
RX/OTC/DISCN : RX
RLD : Yes
TE Code :

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Reply
03 Mar 2026
Reply
09 Dec 2025
Reply
24 May 2023
Reply
21 Aug 2020
Reply
29 Nov 2019
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Patents & EXCLUSIVITIES
ABOUT THIS PAGE
12
PharmaCompass offers a list of Carfilzomib API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Carfilzomib manufacturer or Carfilzomib supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Carfilzomib manufacturer or Carfilzomib supplier.
A Kyprolis manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Kyprolis, including repackagers and relabelers. The FDA regulates Kyprolis manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Kyprolis API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Kyprolis manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Kyprolis supplier is an individual or a company that provides Kyprolis active pharmaceutical ingredient (API) or Kyprolis finished formulations upon request. The Kyprolis suppliers may include Kyprolis API manufacturers, exporters, distributors and traders.
click here to find a list of Kyprolis suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Kyprolis DMF (Drug Master File) is a document detailing the whole manufacturing process of Kyprolis active pharmaceutical ingredient (API) in detail. Different forms of Kyprolis DMFs exist exist since differing nations have different regulations, such as Kyprolis USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Kyprolis DMF submitted to regulatory agencies in the US is known as a USDMF. Kyprolis USDMF includes data on Kyprolis's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Kyprolis USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Kyprolis suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Kyprolis Drug Master File in Japan (Kyprolis JDMF) empowers Kyprolis API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Kyprolis JDMF during the approval evaluation for pharmaceutical products. At the time of Kyprolis JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Kyprolis suppliers with JDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Kyprolis Drug Master File in Korea (Kyprolis KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Kyprolis. The MFDS reviews the Kyprolis KDMF as part of the drug registration process and uses the information provided in the Kyprolis KDMF to evaluate the safety and efficacy of the drug.
After submitting a Kyprolis KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Kyprolis API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Kyprolis suppliers with KDMF on PharmaCompass.
A Kyprolis written confirmation (Kyprolis WC) is an official document issued by a regulatory agency to a Kyprolis manufacturer, verifying that the manufacturing facility of a Kyprolis active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Kyprolis APIs or Kyprolis finished pharmaceutical products to another nation, regulatory agencies frequently require a Kyprolis WC (written confirmation) as part of the regulatory process.
click here to find a list of Kyprolis suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Kyprolis as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Kyprolis API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Kyprolis as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Kyprolis and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Kyprolis NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Kyprolis suppliers with NDC on PharmaCompass.
Kyprolis Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Kyprolis GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Kyprolis GMP manufacturer or Kyprolis GMP API supplier for your needs.
A Kyprolis CoA (Certificate of Analysis) is a formal document that attests to Kyprolis's compliance with Kyprolis specifications and serves as a tool for batch-level quality control.
Kyprolis CoA mostly includes findings from lab analyses of a specific batch. For each Kyprolis CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Kyprolis may be tested according to a variety of international standards, such as European Pharmacopoeia (Kyprolis EP), Kyprolis JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Kyprolis USP).

