
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
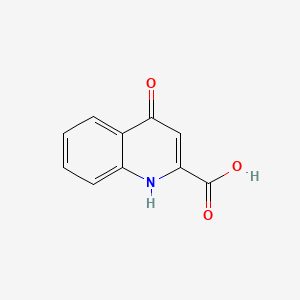

1. 492-27-3
2. 4-hydroxyquinoline-2-carboxylic Acid
3. Quinurenic Acid
4. 4-hydroxyquinaldic Acid
5. Kinurenic Acid
6. Kynuronic Acid
7. 2-quinolinecarboxylic Acid, 4-hydroxy-
8. 4-hydroxy-2-quinolinecarboxylic Acid
9. 4-hydroxyquinaldinic Acid
10. 4-oxo-1h-quinoline-2-carboxylic Acid
11. Chebi:18344
12. Nsc-58973
13. H030s2s85j
14. Dtxsid8075417
15. Acid, Kynurenic
16. Refchem:794384
17. Dtxcid5040675
18. 207-751-5
19. 13593-94-7
20. 4-oxo-1,4-dihydroquinoline-2-carboxylic Acid
21. Transtorine
22. 1,4-dihydro-4-oxoquinoline-2-carboxylic Acid
23. 4-hydroxy-2-quinolincarboxylic Acid
24. Quinaldic Acid, 4-hydroxy-
25. Mfcd00006753
26. Nsc 58973
27. Kyna
28. Nsc58973
29. Mfcd03197717
30. 2-carboxy-4-hydroxyquinoline
31. Chembl299155
32. Ccris 4428
33. Sr-01000075455
34. Einecs 207-751-5
35. Kynurensaeure
36. Kynurenic-acid
37. Unii-h030s2s85j
38. 4-hydroxy-2-quinolinecarboxylic Acid;4-hydroxy-quinaldic Acid;2-carboxy-4-hydroxyquinoline
39. Kynurenate; Kyna
40. 4-hydroxyquinaldate
41. Kya
42. 4-hydroxy-quinaldate
43. 4-hydroxyquinaldinate
44. Spectrum_001116
45. Tocris-0223
46. 4-hydroxy-quinaldic Acid
47. Spectrum2_001342
48. Spectrum3_001390
49. Spectrum4_000814
50. Spectrum5_001318
51. Lopac-k-3375
52. Quinurenic Acid
53. Kynurenate
54. Kynurenic Acid (standard)
55. Biomol-nt_000229
56. Bmse000410
57. Kynurenic Acid, >=98%
58. 4-oxo-1,4-dihydro-quinoline-2-carboxylic Acid
59. Kynurenic Acid [mi]
60. Lopac0_000716
61. Oprea1_032085
62. Schembl22979
63. Bspbio_002980
64. Kbiogr_001327
65. Kbioss_001596
66. Mls002172436
67. Divk1c_000309
68. Spectrum1500688
69. 1,4-dihydro-4-oxoquinoline-2-carboxylicacid
70. Spbio_001523
71. Bpbio1_001350
72. Gtpl2918
73. Orb1298768
74. Orb1980161
75. Schembl5144405
76. Schembl29366068
77. 4-hydroxy-2-chinolincarbonsaeure
78. Bdbm81975
79. Hczhheifkropdy-uhfffaoysa-
80. Hms500p11
81. Kbio1_000309
82. Kbio2_001596
83. Kbio2_004164
84. Kbio2_006732
85. Kbio3_002200
86. Ninds_000309
87. 4-hydroxyquinolinium-2-carboxylate
88. Hms1736a10
89. Hms1921c20
90. Hms2269g22
91. Hms3262o13
92. Hms3266c13
93. Hms3411c03
94. Hms3675c03
95. Hms3885d20
96. Albb-014130
97. Kynurenic Acid - Bio-x Trade Mark
98. Tox21_500716
99. Bbl027606
100. Bdbm50233945
101. Ccg-39280
102. Hb0362
103. Msk157317
104. Pdsp1_000132
105. Pdsp2_000131
106. S4719
107. Sbb003671
108. Stl294769
109. Stl301826
110. 4-hydroxy-quinoline-2-carboxylic Acid
111. Akos000118368
112. Akos000277721
113. Cs-w020664
114. Db11937
115. Fk24817
116. Gs-3763
117. Hy-100806r
118. Hy-w110662
119. Lp00716
120. Sb67494
121. Sb67643
122. Sdccgsbi-0050694.p003
123. Idi1_000309
124. Nsc_5280455
125. Smp1_000172
126. Ncgc00015581-01
127. Ncgc00015581-02
128. Ncgc00015581-03
129. Ncgc00015581-04
130. Ncgc00015581-05
131. Ncgc00015581-06
132. Ncgc00015581-07
133. Ncgc00015581-08
134. Ncgc00015581-09
135. Ncgc00015581-14
136. Ncgc00024505-01
137. Ncgc00024505-02
138. Ncgc00024505-03
139. Ncgc00024505-04
140. Ncgc00024505-05
141. Ncgc00024505-06
142. Ncgc00024505-07
143. Ncgc00261401-01
144. Bk166244
145. Cas_492-27-3
146. Smr000112310
147. St036704
148. St066912
149. Sy152524
150. Db-008408
151. Db-081634
152. Hy-100806
153. 4-oxo-1,4-dihydroquinoline-2-carboxylicacid
154. Cs-0168103
155. Eu-0100716
156. H0303
157. Ns00014859
158. En300-13998
159. C01717
160. K 3375
161. S12153
162. 593d947
163. Ae-641/00585057
164. F047165
165. F554963
166. Q642217
167. Sr-01000075455-1
168. Sr-01000075455-3
169. Brd-k85872723-001-15-9
170. Z94602408
171. 6f535706-b297-4930-a3fc-7a2823830118
172. Inchi=1/c10h7no3/c12-9-5-8(10(13)14)11-7-4-2-1-3-6(7)9/h1-5h,(h,11,12)(h,13,14)
| Molecular Weight | 189.17 g/mol |
|---|---|
| Molecular Formula | C10H7NO3 |
| XLogP3 | 1.3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 1 |
| Exact Mass | Da |
| Monoisotopic Mass | Da |
| Topological Polar Surface Area | 66.4 |
| Heavy Atom Count | 14 |
| Formal Charge | 0 |
| Complexity | 308 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Excitatory Amino Acid Antagonists
Drugs that bind to but do not activate excitatory amino acid receptors, thereby blocking the actions of agonists.
Uremic toxins tend to accumulate in the blood either through dietary excess or through poor filtration by the kidneys. Most uremic toxins are metabolic waste products and are normally excreted in the urine or feces.
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
31
PharmaCompass offers a list of DTXCID5040675 API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right DTXCID5040675 manufacturer or DTXCID5040675 supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred DTXCID5040675 manufacturer or DTXCID5040675 supplier.
A DTXCID5040675 manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of DTXCID5040675, including repackagers and relabelers. The FDA regulates DTXCID5040675 manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. DTXCID5040675 API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A DTXCID5040675 supplier is an individual or a company that provides DTXCID5040675 active pharmaceutical ingredient (API) or DTXCID5040675 finished formulations upon request. The DTXCID5040675 suppliers may include DTXCID5040675 API manufacturers, exporters, distributors and traders.
DTXCID5040675 Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of DTXCID5040675 GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right DTXCID5040675 GMP manufacturer or DTXCID5040675 GMP API supplier for your needs.
A DTXCID5040675 CoA (Certificate of Analysis) is a formal document that attests to DTXCID5040675's compliance with DTXCID5040675 specifications and serves as a tool for batch-level quality control.
DTXCID5040675 CoA mostly includes findings from lab analyses of a specific batch. For each DTXCID5040675 CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
DTXCID5040675 may be tested according to a variety of international standards, such as European Pharmacopoeia (DTXCID5040675 EP), DTXCID5040675 JP (Japanese Pharmacopeia) and the US Pharmacopoeia (DTXCID5040675 USP).

