10 Dec 2025
// PRESS RELEASE
01 Oct 2025
// PRESS RELEASE
29 Sep 2025
// PRESS RELEASE
Latest Content by PharmaCompass
 KEY SERVICES
KEY SERVICES
End-to-end drug development, specializing in high-potency softgels and liquids, providing global partners with a competitive advantage.
About
CPhI North AmericaCPhI North America
Industry Trade Show
Attending
02-04 June, 2026
Industry Trade Show
Attending
16-18 June, 2026
BIO International Conv...BIO International Convention
Industry Trade Show
Attending
22-25 June, 2026
CONTACT DETAILS





Events
Webinars & Exhibitions
CPhI North AmericaCPhI North America
Industry Trade Show
Attending
02-04 June, 2026
Industry Trade Show
Attending
16-18 June, 2026
BIO International Conv...BIO International Convention
Industry Trade Show
Attending
22-25 June, 2026
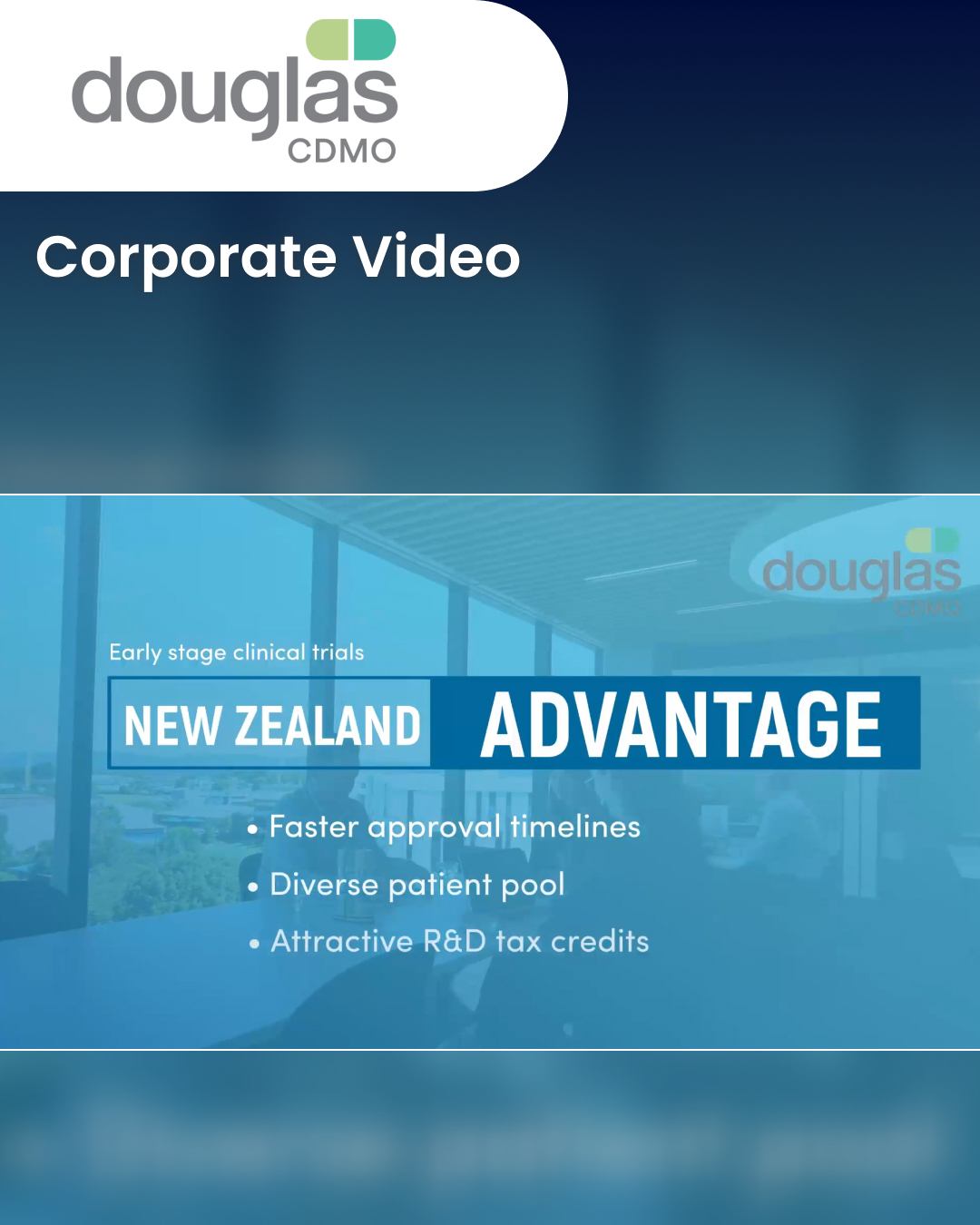
CORPORATE CONTENT #SupplierSpotlight
https://www.pharmacompass.com/radio-compass-blog/cdmo-activity-tracker-veranova-carbogen-lead-adc-investments-axplora-polfa-tarchomin-famar-expand-european-footprint
https://www.pharmacompass.com/radio-compass-blog/cdmo-activity-tracker-bora-polpharma-make-acquisitions-evonik-euroapi-porton-announce-technological-expansions
https://www.pharmacompass.com/radio-compass-blog/cdmo-activity-tracker-novo-s-parent-buys-catalent-for-us-16-5-bn-fujifilm-merck-kgaa-axplora-lonza-expand-capabilities

10 Dec 2025
// PRESS RELEASE
https://www.douglascdmo.com/insights/articles/douglas-cdmos-commitment-to-a-sustainable-future-in-global-healthcare

01 Oct 2025
// PRESS RELEASE
https://www.douglascdmo.com/insights/articles/softgels-proven-formula-for-pharma-commercial-success

29 Sep 2025
// PRESS RELEASE
https://www.douglascdmo.com/insights/articles/your-guide-to-minimizing-scope-creep

01 Jul 2025
// PRESS RELEASE
https://www.douglascdmo.com/insights/articles/the-role-of-cdmos-in-supporting-generic-drug-development-for-the-usa

20 May 2025
// PRESS RELEASE

12 May 2025
// PRESS RELEASE
Services
Packaging
Drug Product Manufacturing
API & Drug Product Development
Inspections and registrations
Country : New Zealand
City/Region : Lincoln
Audit Date : Aug-23
Audit Type : On-Site
Country : New Zealand
City/Region : Lincoln
Audit Date : Apr-26
Audit Type : On-Site
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]ABOUT THIS PAGE


 Douglas CDMO
Douglas CDMO