
Synopsis
Synopsis
0
VMF
0
Australia
Annual Reports
NA
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
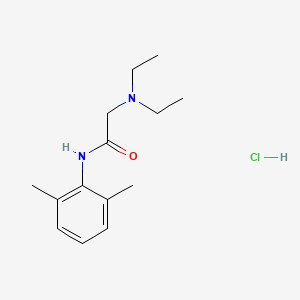

1. 2-(diethylamino)-n-(2,6-dimethylphenyl)acetamide
2. 2-2etn-2mephacn
3. Dalcaine
4. Lidocaine
5. Lidocaine Carbonate
6. Lidocaine Carbonate (2:1)
7. Lidocaine Hydrocarbonate
8. Lidocaine Monoacetate
9. Lidocaine Monohydrochloride
10. Lidocaine Monohydrochloride, Monohydrate
11. Lidocaine Sulfate (1:1)
12. Octocaine
13. Xylesthesin
14. Xylocaine
15. Xylocitin
16. Xyloneural
1. 73-78-9
2. Lidocaine Hcl
3. Lidothesin
4. Xyloneural
5. Lignocaine Hydrochloride
6. Lidocaine (hydrochloride)
7. Lidocaton
8. Xylocard
9. Lidocaine Hydrochloride Anhydrous
10. Lidopen
11. Acetamide, 2-(diethylamino)-n-(2,6-dimethylphenyl)-, Monohydrochloride
12. Lta Ii Kit
13. Laryng-o-jet Kit
14. Ec2cnf7xfp
15. Linocaine Hydrochloride
16. Mls000069665
17. Laryngotracheal Anesthesia Kit
18. 2-(diethylamino)-n-(2,6-dimethylphenyl)acetamide Hydrochloride
19. 2-diethylamino-2',6'-acetoxylidide Hydrochloride
20. Alphacaine Hydrochloride
21. Nsc-757420
22. Smr000058468
23. Xylocaine (tn)
24. Alpha-diethylamino-2,6-acetoxylidine Hydrochloride
25. Laryng-o-jet
26. Omega-diethylamino-2,6-dimethylacetanilide Hydrochloride
27. Xilina Hydrochloride
28. 73-78-9 (hcl); 6108-05-0 (monohydrate)
29. Rucaina Hydrochloride
30. Xycaine Hydrochloride
31. Xylotox Hydrochloride
32. Duncaine Hydrochloride
33. Isicaine Hydrochloride
34. Lidocain Hydrochloride
35. Anestacon Hydrochloride
36. Gravocain Hydrochloride
37. Leostesin Hydrochloride
38. Xylocitin Hydrochloride
39. Lidothesin Hydrochloride
40. Xylestesin Hydrochloride
41. Glydo
42. Lidocaine Viscous
43. Pediatric Lta Kit
44. Chebi:50512
45. Xylocaine Preservative Free
46. Sr-01000000189
47. Unii-ec2cnf7xfp
48. Einecs 200-803-8
49. Xylocaine 4% Preservative Free
50. Xylocaine 5% W/ Glucose 7.5%
51. Lidocaine Hydrochloride Viscous
52. S 202
53. V 262
54. Xylocaine 1.5% W/ Dextrose 7.5%
55. Dalcaine (tn)
56. Prestwick_296
57. N-(diethylaminoacetyl)-2,6-dimethylaniline Hydrochloride
58. Lidoca Ne Hydrochloride
59. Lidocaine Hydrochloride Preservative Free
60. Opera_id_351
61. Lidocaine Hydrochloride In Plastic Container
62. Lidocaine Hydrochloride 0.2% In Dextrose 5%
63. Lidocaine Hydrochloride 0.4% In Dextrose 5%
64. Lidocaine Hydrochloride 5% And Dextrose 7.5%
65. Spectrum1500689
66. Chembl541521
67. Dtxsid4058782
68. Hy-b0185a
69. 2',6'-acetoxylidide, 2-(diethylamino)-, Monohydrochloride
70. Hms1568i21
71. Hms1921c22
72. Lidocaine Hydrochloride Preservative Free In Plastic Container
73. Pharmakon1600-01500689
74. Lidocaine Hydrochloride (jan/usp)
75. 2',6'-acetoxylidide, 2-(diethylamino)-, Hydrochloride
76. Bcp30473
77. Lidocaine Hydrochloride 0.2% In Dextrose 5% In Plastic Container
78. Lidocaine Hydrochloride 0.4% In Dextrose 5% In Plastic Container
79. Lidocaine Hydrochloride 0.8% In Dextrose 5% In Plastic Container
80. Tox21_500669
81. Ccg-39281
82. Lidocaine Hydrochloride [jan]
83. Lidocaine Hydrochloride 0.1% And Dextrose 5% In Plastic Container
84. Lidocaine Hydrochloride 0.2% And Dextrose 5% In Plastic Container
85. Lidocaine Hydrochloride 0.4% And Dextrose 5% In Plastic Container
86. Lidocaine Hydrochloride 0.8% And Dextrose 5% In Plastic Container
87. Nsc757420
88. S4667
89. (unlabeled)lidocaine-d6 Hydrochloride
90. Akos015889456
91. Cs-3888
92. Lp00669
93. Nsc 757420
94. Sb19119
95. Anhydrous Lidocaine Hydrochloride
96. Lidocaine Hydrochloride [who-dd]
97. Ncgc00094030-01
98. Ncgc00094030-02
99. Ncgc00094030-03
100. Ncgc00094030-04
101. Ncgc00094030-05
102. Ncgc00261354-01
103. Ac-11712
104. As-35171
105. Eu-0100669
106. Lignocaine Hydrochloride Pound>>lidocaine Hcl
107. A16132
108. D02086
109. L 5647
110. Lidocaine Hydrochloride Anhydrous [mart.]
111. A837924
112. Q-201304
113. Sr-01000000189-3
114. Sr-01000000189-9
115. Q27122094
116. 2-(diethylamino)-2',6'-acetoxylidide Monohydrochloride
117. 2-(diethylamino)-n-(2,6-dimethylphenyl)acetamide;hydrochloride
118. 2-(diethylamino)-n-(2,6-dimethylphenyl)ethanamide Hydrochloride
119. Acetamide, 2-(diethylamino)-n-(2,6-dimethylphenyl)-, Hydrochloride (1:1)
| Molecular Weight | 270.80 g/mol |
|---|---|
| Molecular Formula | C14H23ClN2O |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 5 |
| Exact Mass | 270.1498911 g/mol |
| Monoisotopic Mass | 270.1498911 g/mol |
| Topological Polar Surface Area | 32.3 Ų |
| Heavy Atom Count | 18 |
| Formal Charge | 0 |
| Complexity | 228 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 18 | |
|---|---|
| Drug Name | Laryng-o-jet kit |
| Drug Label | DESCRIPTIONLidocaine Hydrochloride Oral Topical Solution, USP (Viscous) 2% contains a local anesthetic agent and is administered topically. Lidocaine Hydrochloride Oral Topical Solution, USP (Viscous) 2% contains lidocaine hydrochloride, which is che... |
| Active Ingredient | Lidocaine hydrochloride |
| Dosage Form | Solution |
| Route | Topical |
| Strength | 4% |
| Market Status | Prescription |
| Company | Intl Medication |
| 2 of 18 | |
|---|---|
| Drug Name | Lidocaine hydrochloride |
| Active Ingredient | Lidocaine hydrochloride |
| Dosage Form | Jelly; Injectable; Solution |
| Route | Injection; Oral; Topical |
| Strength | 0.5%; 1%; 20%; 2%; 4% |
| Market Status | Prescription |
| Company | Wockhardt; Igi Labs; Hospira; Roxane; Watson Labs; Intl Medication; Hi Tech Pharma; Vintage; Luitpold; Agila Speclts; Akorn |
| 3 of 18 | |
|---|---|
| Drug Name | Lidocaine hydrochloride in plastic container |
| Drug Label | Each mL contains:Lidocaine Hydrochloride . . . . . . . . . . 20 mg (2%)Inactive ingredients: flavoring, methylparaben, propylparaben, sodium carboxymethylcellulose, and sodium saccharin in an aqueous solution.Lidocaine Viscous (Lidocaine Hydrochlorid... |
| Active Ingredient | Lidocaine hydrochloride |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 0.5%; 1%; 2% |
| Market Status | Prescription |
| Company | Hospira; Fresenius Kabi Usa |
| 4 of 18 | |
|---|---|
| Drug Name | Lidocaine hydrochloride preservative free |
| Active Ingredient | Lidocaine hydrochloride |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 1%; 20%; 1.5%; 2%; 4% |
| Market Status | Prescription |
| Company | Hospira; Aurobindo Pharma; Intl Medication; Fresenius Kabi Usa; Agila Speclts |
| 5 of 18 | |
|---|---|
| Drug Name | Lidocaine hydrochloride preservative free in plastic container |
| Active Ingredient | Lidocaine hydrochloride |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 1%; 2% |
| Market Status | Prescription |
| Company | Hospira |
| 6 of 18 | |
|---|---|
| Drug Name | Lidocaine hydrochloride viscous |
| Active Ingredient | Lidocaine hydrochloride |
| Dosage Form | Solution |
| Route | Oral |
| Strength | 2% |
| Market Status | Prescription |
| Company | Vintage |
| 7 of 18 | |
|---|---|
| Drug Name | Lidocaine viscous |
| Active Ingredient | Lidocaine hydrochloride |
| Dosage Form | Solution |
| Route | Oral |
| Strength | 2% |
| Market Status | Prescription |
| Company | Roxane |
| 8 of 18 | |
|---|---|
| Drug Name | Lta ii kit |
| Active Ingredient | Lidocaine hydrochloride |
| Dosage Form | Solution |
| Route | Topical |
| Strength | 4% |
| Market Status | Prescription |
| Company | Hospira |
| 9 of 18 | |
|---|---|
| Drug Name | Xylocaine preservative free |
| Active Ingredient | Lidocaine hydrochloride |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 1%; 20%; 2%; 4% |
| Market Status | Prescription |
| Company | Fresenius Kabi Usa |
| 10 of 18 | |
|---|---|
| Drug Name | Laryng-o-jet kit |
| Drug Label | DESCRIPTIONLidocaine Hydrochloride Oral Topical Solution, USP (Viscous) 2% contains a local anesthetic agent and is administered topically. Lidocaine Hydrochloride Oral Topical Solution, USP (Viscous) 2% contains lidocaine hydrochloride, which is che... |
| Active Ingredient | Lidocaine hydrochloride |
| Dosage Form | Solution |
| Route | Topical |
| Strength | 4% |
| Market Status | Prescription |
| Company | Intl Medication |
| 11 of 18 | |
|---|---|
| Drug Name | Lidocaine hydrochloride |
| Active Ingredient | Lidocaine hydrochloride |
| Dosage Form | Jelly; Injectable; Solution |
| Route | Injection; Oral; Topical |
| Strength | 0.5%; 1%; 20%; 2%; 4% |
| Market Status | Prescription |
| Company | Wockhardt; Igi Labs; Hospira; Roxane; Watson Labs; Intl Medication; Hi Tech Pharma; Vintage; Luitpold; Agila Speclts; Akorn |
| 12 of 18 | |
|---|---|
| Drug Name | Lidocaine hydrochloride in plastic container |
| Drug Label | Each mL contains:Lidocaine Hydrochloride . . . . . . . . . . 20 mg (2%)Inactive ingredients: flavoring, methylparaben, propylparaben, sodium carboxymethylcellulose, and sodium saccharin in an aqueous solution.Lidocaine Viscous (Lidocaine Hydrochlorid... |
| Active Ingredient | Lidocaine hydrochloride |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 0.5%; 1%; 2% |
| Market Status | Prescription |
| Company | Hospira; Fresenius Kabi Usa |
| 13 of 18 | |
|---|---|
| Drug Name | Lidocaine hydrochloride preservative free |
| Active Ingredient | Lidocaine hydrochloride |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 1%; 20%; 1.5%; 2%; 4% |
| Market Status | Prescription |
| Company | Hospira; Aurobindo Pharma; Intl Medication; Fresenius Kabi Usa; Agila Speclts |
| 14 of 18 | |
|---|---|
| Drug Name | Lidocaine hydrochloride preservative free in plastic container |
| Active Ingredient | Lidocaine hydrochloride |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 1%; 2% |
| Market Status | Prescription |
| Company | Hospira |
| 15 of 18 | |
|---|---|
| Drug Name | Lidocaine hydrochloride viscous |
| Active Ingredient | Lidocaine hydrochloride |
| Dosage Form | Solution |
| Route | Oral |
| Strength | 2% |
| Market Status | Prescription |
| Company | Vintage |
| 16 of 18 | |
|---|---|
| Drug Name | Lidocaine viscous |
| Active Ingredient | Lidocaine hydrochloride |
| Dosage Form | Solution |
| Route | Oral |
| Strength | 2% |
| Market Status | Prescription |
| Company | Roxane |
| 17 of 18 | |
|---|---|
| Drug Name | Lta ii kit |
| Active Ingredient | Lidocaine hydrochloride |
| Dosage Form | Solution |
| Route | Topical |
| Strength | 4% |
| Market Status | Prescription |
| Company | Hospira |
| 18 of 18 | |
|---|---|
| Drug Name | Xylocaine preservative free |
| Active Ingredient | Lidocaine hydrochloride |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 1%; 20%; 2%; 4% |
| Market Status | Prescription |
| Company | Fresenius Kabi Usa |
Voltage-Gated Sodium Channel Blockers
A class of drugs that inhibit the activation of VOLTAGE-GATED SODIUM CHANNELS. (See all compounds classified as Voltage-Gated Sodium Channel Blockers.)
Anesthetics, Local
Drugs that block nerve conduction when applied locally to nerve tissue in appropriate concentrations. They act on any part of the nervous system and on every type of nerve fiber. In contact with a nerve trunk, these anesthetics can cause both sensory and motor paralysis in the innervated area. Their action is completely reversible. (From Gilman AG, et. al., Goodman and Gilman's The Pharmacological Basis of Therapeutics, 8th ed) Nearly all local anesthetics act by reducing the tendency of voltage-dependent sodium channels to activate. (See all compounds classified as Anesthetics, Local.)
Anti-Arrhythmia Agents
Agents used for the treatment or prevention of cardiac arrhythmias. They may affect the polarization-repolarization phase of the action potential, its excitability or refractoriness, or impulse conduction or membrane responsiveness within cardiac fibers. Anti-arrhythmia agents are often classed into four main groups according to their mechanism of action: sodium channel blockade, beta-adrenergic blockade, repolarization prolongation, or calcium channel blockade. (See all compounds classified as Anti-Arrhythmia Agents.)
 SWATI - Transforming science into solutions with 60+ years of expertise, global accreditations, and pioneering biotech innovation.
SWATI - Transforming science into solutions with 60+ years of expertise, global accreditations, and pioneering biotech innovation.
 Click Us!
Click Us! 
GDUFA
DMF Review : Reviewed
Rev. Date : 2015-01-05
Pay. Date : 2013-09-18
DMF Number : 25963
Submission : 2012-03-06
Status : Active
Type : II

Certificate Number : CEP 2013-103 - Rev 04
Issue Date : 2025-02-03
Type : Chemical
Substance Number : 227
Status : Valid

Date of Issue : 2025-09-24
Valid Till : 2028-07-07
Written Confirmation Number : WC-0165
Address of the Firm :

NDC Package Code : 52119-009
Start Marketing Date : 2014-03-20
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Registrant Name : Daeshin Muyak Co., Ltd.
Registration Date : 2020-01-08
Registration Number : 20200108-210-J-409
Manufacturer Name : Swati Spentose Private Limited (Unit-I)
Manufacturer Address : Plot No. - A-1/2102, 2103 & A-2/2120, Phase III, GIDC, Vapi - 396 195, Dist. - Valsad, Gujarat State, INDIA.

 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
 Gonane has API manufacturing expertise in new-age Corticosteroids, Hormones and other pharma raw materials.
Gonane has API manufacturing expertise in new-age Corticosteroids, Hormones and other pharma raw materials.

GDUFA
DMF Review : Reviewed
Rev. Date : 2014-06-05
Pay. Date : 2014-05-29
DMF Number : 5876
Submission : 1985-06-10
Status : Active
Type : II

Certificate Number : CEP 1996-020 - Rev 09
Issue Date : 2024-05-29
Type : Chemical
Substance Number : 227
Status : Valid

Registration Number : 219MF10205
Registrant's Address : Cesar Martinell i Brunet 12A, Poligono Rubi Sur, Rubi (Barcelona), Spain
Initial Date of Registration : 2007-06-13
Latest Date of Registration :

NDC Package Code : 71495-0001
Start Marketing Date : 2017-05-01
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

| Available Reg Filing : ASMF |

 China’s top API & intermediate manufacturer, delivering advanced CMO/CDMO solutions to 60+ countries, empowering global health progress
China’s top API & intermediate manufacturer, delivering advanced CMO/CDMO solutions to 60+ countries, empowering global health progress
 Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.

NDC Package Code : 61281-2000
Start Marketing Date : 2011-11-21
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

 Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
 Apex Healthcare Limited: ISO 9001:2008, WHO-GMP, US FDA-audited manufacturer & exporter of APIs, bulk drugs, and formulations.
Apex Healthcare Limited: ISO 9001:2008, WHO-GMP, US FDA-audited manufacturer & exporter of APIs, bulk drugs, and formulations.

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39139
Submission : 2023-12-01
Status : Active
Type : II

Certificate Number : CEP 2024-258 - Rev 00
Issue Date : 2025-10-02
Type : Chemical
Substance Number : 227
Status : Valid

Date of Issue : 2024-03-01
Valid Till : 2026-12-31
Written Confirmation Number : WC-0409
Address of the Firm :

NDC Package Code : 69617-010
Start Marketing Date : 2019-01-01
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (25kg/25kg)
Marketing Category : BULK INGREDIENT FOR HUMAN PRESCRIPTION COMPOUNDING
 Willow Birch Pharma delivers trusted, high-quality APIs nationwide with unmatched service, compliance, and competitive value.
Willow Birch Pharma delivers trusted, high-quality APIs nationwide with unmatched service, compliance, and competitive value.
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
 SWATI - Transforming science into solutions with 60+ years of expertise, global accreditations, and pioneering biotech innovation.
SWATI - Transforming science into solutions with 60+ years of expertise, global accreditations, and pioneering biotech innovation.
GDUFA
DMF Review : Complete
Rev. Date : 2015-01-05
Pay. Date : 2013-09-18
DMF Number : 25963
Submission : 2012-03-06
Status : Active
Type : II
GDUFA
DMF Review : Complete
Rev. Date : 2014-06-05
Pay. Date : 2014-05-29
DMF Number : 5876
Submission : 1985-06-10
Status : Active
Type : II
 Apex Healthcare Limited: ISO 9001:2008, WHO-GMP, US FDA-audited manufacturer & exporter of APIs, bulk drugs, and formulations.
Apex Healthcare Limited: ISO 9001:2008, WHO-GMP, US FDA-audited manufacturer & exporter of APIs, bulk drugs, and formulations.
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39139
Submission : 2023-12-01
Status : Active
Type : II
GDUFA
DMF Review : Complete
Rev. Date : 2015-10-07
Pay. Date : 2015-09-29
DMF Number : 19531
Submission : 2006-04-30
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2014-01-14
Pay. Date : 2013-11-29
DMF Number : 11338
Submission : 1995-02-10
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 17483
Submission : 2004-06-21
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 24051
Submission : 2010-08-09
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 250
Submission : 1940-01-01
Status : Inactive
Type : II

USDMF
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 5921
Submission : 1985-07-20
Status : Inactive
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
USDMF
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 4140
Submission : 1981-03-03
Status : Inactive
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Reply
19 Mar 2026
Reply
03 Mar 2026
Reply
25 Sep 2025
Reply
22 Aug 2025
Reply
28 May 2025
Reply
18 Mar 2025
Reply
25 Feb 2025
Reply
09 Nov 2024
Reply
17 Oct 2024
Reply
24 Aug 2024
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
14
PharmaCompass offers a list of Lidocaine Hydrochloride API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Lidocaine Hydrochloride manufacturer or Lidocaine Hydrochloride supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Lidocaine Hydrochloride manufacturer or Lidocaine Hydrochloride supplier.
A Xylocaine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Xylocaine, including repackagers and relabelers. The FDA regulates Xylocaine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Xylocaine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Xylocaine manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Xylocaine supplier is an individual or a company that provides Xylocaine active pharmaceutical ingredient (API) or Xylocaine finished formulations upon request. The Xylocaine suppliers may include Xylocaine API manufacturers, exporters, distributors and traders.
click here to find a list of Xylocaine suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Xylocaine DMF (Drug Master File) is a document detailing the whole manufacturing process of Xylocaine active pharmaceutical ingredient (API) in detail. Different forms of Xylocaine DMFs exist exist since differing nations have different regulations, such as Xylocaine USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Xylocaine DMF submitted to regulatory agencies in the US is known as a USDMF. Xylocaine USDMF includes data on Xylocaine's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Xylocaine USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Xylocaine suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Xylocaine Drug Master File in Japan (Xylocaine JDMF) empowers Xylocaine API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Xylocaine JDMF during the approval evaluation for pharmaceutical products. At the time of Xylocaine JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Xylocaine suppliers with JDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Xylocaine Drug Master File in Korea (Xylocaine KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Xylocaine. The MFDS reviews the Xylocaine KDMF as part of the drug registration process and uses the information provided in the Xylocaine KDMF to evaluate the safety and efficacy of the drug.
After submitting a Xylocaine KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Xylocaine API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Xylocaine suppliers with KDMF on PharmaCompass.
A Xylocaine CEP of the European Pharmacopoeia monograph is often referred to as a Xylocaine Certificate of Suitability (COS). The purpose of a Xylocaine CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Xylocaine EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Xylocaine to their clients by showing that a Xylocaine CEP has been issued for it. The manufacturer submits a Xylocaine CEP (COS) as part of the market authorization procedure, and it takes on the role of a Xylocaine CEP holder for the record. Additionally, the data presented in the Xylocaine CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Xylocaine DMF.
A Xylocaine CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Xylocaine CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Xylocaine suppliers with CEP (COS) on PharmaCompass.
A Xylocaine written confirmation (Xylocaine WC) is an official document issued by a regulatory agency to a Xylocaine manufacturer, verifying that the manufacturing facility of a Xylocaine active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Xylocaine APIs or Xylocaine finished pharmaceutical products to another nation, regulatory agencies frequently require a Xylocaine WC (written confirmation) as part of the regulatory process.
click here to find a list of Xylocaine suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Xylocaine as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Xylocaine API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Xylocaine as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Xylocaine and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Xylocaine NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Xylocaine suppliers with NDC on PharmaCompass.
Xylocaine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Xylocaine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Xylocaine GMP manufacturer or Xylocaine GMP API supplier for your needs.
A Xylocaine CoA (Certificate of Analysis) is a formal document that attests to Xylocaine's compliance with Xylocaine specifications and serves as a tool for batch-level quality control.
Xylocaine CoA mostly includes findings from lab analyses of a specific batch. For each Xylocaine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Xylocaine may be tested according to a variety of international standards, such as European Pharmacopoeia (Xylocaine EP), Xylocaine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Xylocaine USP).

