
Synopsis
0
JDMF
0
KDMF
0
NDC API
0
VMF
0
Australia
DRUG PRODUCT COMPOSITIONS
Annual Reports
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
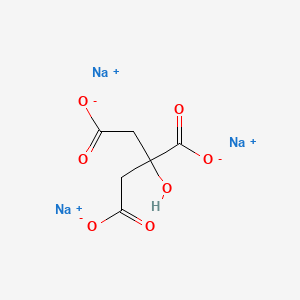

1. Anhydrous Sodium Citrate
2. Citra Ph
3. Monosodium Citrate
4. Sodium Citrate Dihydrate
5. Sodium Citrate Monobasic
6. Sodium Citrate, Anhydrous
7. Trisodium Citrate Dihydrate
1. Trisodium Citrate
2. 68-04-2
3. Citrosodine
4. Natrocitral
5. Sodium Citrate Anhydrous
6. Sodium Citrate, Anhydrous
7. Citric Acid, Trisodium Salt
8. Trisodium Citrate, Anhydrous
9. 1,2,3-propanetricarboxylic Acid, 2-hydroxy-, Trisodium Salt
10. Anhydrous Sodium Citrate
11. Citric Acid Trisodium Salt
12. Sodium 2-hydroxypropane-1,2,3-tricarboxylate
13. Trisodium Citrate Anhydrous
14. Fema No. 3026
15. Trisodium-citrate
16. Anhydrous Trisodium Citrate
17. 994-36-5
18. Sodium Citrate,anhydrous
19. Rs7a450lga
20. Ins No.331(iii)
21. Ins-331(iii)
22. Chebi:53258
23. E-331(iii)
24. Trisodium 2-hydroxypropane-1,2,3-tricarboxylate
25. Trisodium;2-hydroxypropane-1,2,3-tricarboxylate
26. Mfcd00012462
27. Fema No. 3026, Anhydrous-
28. Citric Acid, Sodium Salt
29. Citrosodina
30. Citnatin
31. Citreme
32. Citrosodna
33. Sodium Citrate Hydrous
34. 1,2,3-propanetricarboxylic Acid, 2-hydroxy-, Sodium Salt
35. Ccris 3293
36. Sodium Citrate (na3c6h5o7)
37. Hsdb 5201
38. Einecs 200-675-3
39. Unii-rs7a450lga
40. N-1560
41. Natrii Citras
42. Tri-sodium Citrate
43. Trisodium 2-hydroxy-1,2,3-propanetricarboxylate
44. Sodium Citrate Salt
45. 1,2,3-propanetricarboxylic Acid, 2-hydroxy-, Sodium Salt (1:3)
46. Sodium (iii) Citrate
47. Sodium Citrate (usp)
48. Natrii Citras, Dehydrate
49. Ec 200-675-3
50. Anticoagulant Sodium Citrate
51. Chembl1355
52. Sodium Citrate [mi]
53. Citrate Concentrated Solution
54. Dtxsid2026363
55. Sodium Citrate [who-ip]
56. 2-hydroxy-1,2,3-propanetricarboxylic Acid, Trisodium Salt
57. Citric Acid Trisodium Salt, 99%
58. Natrii Citras [who-ip Latin]
59. Akos015915009
60. Citrate Solution, Ph ~3.0, 30 Mm
61. Db09154
62. Sodium Citrate Anhydrous [hsdb]
63. Anhydrous Trisodium Citrate [ii]
64. Sodium Citrate,anhydrous [vandf]
65. Ac-15008
66. E331
67. Sodium Citrate Dihydrate Usp Fine Granular
68. Sodium Citrate, Anhydrous [who-ip]
69. B7298
70. Ft-0623960
71. D05855
72. D77308
73. Sodium Citrate, 0.5m Buffer Solution, Ph 5.0
74. Sodium Citrate, 0.5m Buffer Solution, Ph 5.5
75. Sodium Citrate, 0.5m Buffer Solution, Ph 6.0
76. Sodium Citrate, 0.5m Buffer Solution, Ph 6.5
77. Anhydrous Trisodium Citrate [usp Monograph]
78. Q409728
79. J-520101
80. Citric Acid Trisodium Salt, Anhydrous, >=98% (gc)
81. Citrate Solution, Ph 3.6+/-0.1 (25 C), 27 Mm
82. Citric Acid Trisodium Salt, Vetec(tm) Reagent Grade, 98%
83. 2-hydroxy-1,2,3-propanenetricarboxylic Acid Trisodium Salt Dihydrate
84. Citrate Concentrated Solution, Bioultra, For Molecular Biology, 1 M In H2o
85. Buffer Solution Ph 5.0 (20 C), Citric Acid ~0.096 M, Sodium Hydroxide ~0.20 M
86. Citrate Concentrated Solution, Bioreagent, Suitable For Coagulation Assays, 4 % (w/v)
87. 8055-55-8
| Molecular Weight | 258.07 g/mol |
|---|---|
| Molecular Formula | C6H5Na3O7 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 2 |
| Exact Mass | 257.97283534 g/mol |
| Monoisotopic Mass | 257.97283534 g/mol |
| Topological Polar Surface Area | 141 Ų |
| Heavy Atom Count | 16 |
| Formal Charge | 0 |
| Complexity | 211 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 4 |
...USED AS AN EXPECTORANT...& SYSTEMIC ALKALIZER. SALINE EXPECTORANTS ARE ESPECIALLY USEFUL WHEN IT IS DESIRED TO LIQUEFY THICK, TENACIOUS SPUTUM. IN THE BODY, SODIUM CITRATE IS OXIDIZED TO BICARBONATE & EXCRETED IN THE URINE; THUS, WHEN GIVEN ORALLY IT IS USEFUL IN ACIDOSIS & TO OVERCOME EXCESSIVE URINARY ACIDITY.
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 802
SODIUM CITRATE ALSO HAS A DIURETIC...ACTION.
American Hospital Formulary Service. Volumes I and II. Washington, DC: American Society of Hospital Pharmacists, to 1984., p. 40:08
SODIUM CITRATE ALSO INCREASES THE URINARY EXCRETION OF CALCIUM. THEREFORE, IT HAS BEEN EMPLOYED IN HYPERCALCEMIA & TO FACILITATE ELIMINATION OF LEAD IN POISONING DUE TO THE LATTER AGENT. /SRP: FORMER USE/
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 802
MEDICATION (VET): ANTICOAGULANT FOR COLLECTION OF BLOOD /SRP: FORMER USE/
The Merck Index. 9th ed. Rahway, New Jersey: Merck & Co., Inc., 1976., p. 1112
WHEN GIVEN IN EXCESS AMT SODIUM CITRATE MAY PRODUCE ALKALOSIS & MAY CAUSE TETANY OR DEPRESS THE HEART BY DECREASING THE IONIZED CALCIUM LEVEL OF THE BLOOD.
American Hospital Formulary Service. Volumes I and II. Washington, DC: American Society of Hospital Pharmacists, to 1984., p. 40:08
Used as an anticoagulant during plasmophoresis as well as a neutralizing agent in the treatment of upset stomach and acidic urine.
FDA Label
Citrate prevents activation of the clotting cascade by chelating calcium ions. Citrate neutralizes acid in the stomach and urine, raising the pH.
Buffers
A chemical system that functions to control the levels of specific ions in solution. When the level of hydrogen ion in solution is controlled the system is called a pH buffer. (See all compounds classified as Buffers.)
Food Preservatives
Substances capable of inhibiting, retarding or arresting the process of fermentation, acidification or other deterioration of foods. (See all compounds classified as Food Preservatives.)
Anticoagulants
Agents that prevent BLOOD CLOTTING. (See all compounds classified as Anticoagulants.)
B - Blood and blood forming organs
B05 - Blood substitutes and perfusion solutions
B05C - Irrigating solutions
B05CB - Salt solutions
B05CB02 - Sodium citrate
Absorption
Tmax of 98-130min.
Route of Elimination
Largely eliminated through hepatic metabolism with very little cleared by the kidneys.
Volume of Distribution
19-39L.
Clearance
Total clearance of 313-1107mL/min.
IN THE BODY, SODIUM CITRATE IS OXIDIZED TO BICARBONATE & EXCRETED IN THE URINE...
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 802
Citrate is metabolized to bicarbonate in the liver and plays a role as an intermediate in the citric acid cycle.
IN THE BODY, SODIUM CITRATE IS OXIDIZED TO BICARBONATE...
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 802
18-54 min
Citrate chelates free calcium ions preventing them from forming a complex with tissue factor and coagulation factor VIIa to promote the activation of coagulation factor X. This inhibits the extrinsic initiation of the coagulation cascade. Citrate may also exert an anticoagulant effect via a so far unknown mechanism as restoration of calcium concentration does not fully reverse the effect of citrate. Citrate is a weak base and so reacts with hydrochloric acid in the stomach to raise the pH. It it further metabolized to bicarbonate which then acts as a systemic alkalizing agent, raising the pH of the blood and urine. It also acts as a diuretic and increases the urinary excretion of calcium.
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : ETHICAL
Registration Country : Canada
Brand Name : AMO ENDOSOL SOLUTION
Dosage Form : SOLUTION
Dosage Strength : 0.17%
Packaging : 500ML
Approval Date :
Application Number : 2007967
Regulatory Info : ETHICAL
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : ETHICAL
Registration Country : Canada
Brand Name : ANTICOAGULANT SOD CITRATE SOLTN 4GM/100ML
Dosage Form : SOLUTION
Dosage Strength : 4G/100ML
Packaging : 250/500/1000ML
Approval Date :
Application Number : 60313
Regulatory Info : ETHICAL
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : ETHICAL
Registration Country : Canada
Brand Name : REGIOCIT
Dosage Form : SOLUTION
Dosage Strength : 5.29G/L
Packaging :
Approval Date :
Application Number : 2496755
Regulatory Info : ETHICAL
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : ETHICAL
Registration Country : Canada
Brand Name : ANTICOAGULANT CITRATE DEXTROSE SOLUTION USP (ACD) FORMULA A
Dosage Form : SOLUTION
Dosage Strength : 2.2G/100ML
Packaging : 500ML/1000ML
Approval Date :
Application Number : 788139
Regulatory Info : ETHICAL
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : ETHICAL
Registration Country : Canada
Brand Name : ANTICOAGULANT SODIUM CITRATE SOLUTION, USP
Dosage Form : SOLUTION
Dosage Strength : 4G/100ML
Packaging :
Approval Date :
Application Number : 2529637
Regulatory Info : ETHICAL
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada
Brand Name : DALMACOL
Dosage Form : SYRUP
Dosage Strength : 200MG/5ML
Packaging : 500ML/2L
Approval Date :
Application Number : 2230769
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : ETHICAL
Registration Country : Canada
Brand Name : SSP+
Dosage Form : SOLUTION
Dosage Strength : 3.18G/L
Packaging :
Approval Date :
Application Number : 2492067
Regulatory Info : ETHICAL
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : ETHICAL
Registration Country : Canada
Brand Name : SSP+
Dosage Form : SOLUTION
Dosage Strength : 3.18G/L
Packaging :
Approval Date :
Application Number : 2492075
Regulatory Info : ETHICAL
Registration Country : Canada

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
59
PharmaCompass offers a list of Sodium Citrate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Sodium Citrate manufacturer or Sodium Citrate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Sodium Citrate manufacturer or Sodium Citrate supplier.
A Sodium Citrate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Sodium Citrate, including repackagers and relabelers. The FDA regulates Sodium Citrate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Sodium Citrate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Sodium Citrate manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Sodium Citrate supplier is an individual or a company that provides Sodium Citrate active pharmaceutical ingredient (API) or Sodium Citrate finished formulations upon request. The Sodium Citrate suppliers may include Sodium Citrate API manufacturers, exporters, distributors and traders.
click here to find a list of Sodium Citrate suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Sodium Citrate DMF (Drug Master File) is a document detailing the whole manufacturing process of Sodium Citrate active pharmaceutical ingredient (API) in detail. Different forms of Sodium Citrate DMFs exist exist since differing nations have different regulations, such as Sodium Citrate USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Sodium Citrate DMF submitted to regulatory agencies in the US is known as a USDMF. Sodium Citrate USDMF includes data on Sodium Citrate's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Sodium Citrate USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Sodium Citrate suppliers with USDMF on PharmaCompass.
A Sodium Citrate CEP of the European Pharmacopoeia monograph is often referred to as a Sodium Citrate Certificate of Suitability (COS). The purpose of a Sodium Citrate CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Sodium Citrate EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Sodium Citrate to their clients by showing that a Sodium Citrate CEP has been issued for it. The manufacturer submits a Sodium Citrate CEP (COS) as part of the market authorization procedure, and it takes on the role of a Sodium Citrate CEP holder for the record. Additionally, the data presented in the Sodium Citrate CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Sodium Citrate DMF.
A Sodium Citrate CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Sodium Citrate CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Sodium Citrate suppliers with CEP (COS) on PharmaCompass.
A Sodium Citrate written confirmation (Sodium Citrate WC) is an official document issued by a regulatory agency to a Sodium Citrate manufacturer, verifying that the manufacturing facility of a Sodium Citrate active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Sodium Citrate APIs or Sodium Citrate finished pharmaceutical products to another nation, regulatory agencies frequently require a Sodium Citrate WC (written confirmation) as part of the regulatory process.
click here to find a list of Sodium Citrate suppliers with Written Confirmation (WC) on PharmaCompass.
Sodium Citrate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Sodium Citrate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Sodium Citrate GMP manufacturer or Sodium Citrate GMP API supplier for your needs.
A Sodium Citrate CoA (Certificate of Analysis) is a formal document that attests to Sodium Citrate's compliance with Sodium Citrate specifications and serves as a tool for batch-level quality control.
Sodium Citrate CoA mostly includes findings from lab analyses of a specific batch. For each Sodium Citrate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Sodium Citrate may be tested according to a variety of international standards, such as European Pharmacopoeia (Sodium Citrate EP), Sodium Citrate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Sodium Citrate USP).

