
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. 7775-09-9
2. Chloric Acid, Sodium Salt
3. Asex
4. Atlacide
5. Desolet
6. Sodakem
7. Oxycil
8. Travex
9. Evau-super
10. Kusa-tohru
11. Polybor-chlorate
12. B-herbatox
13. Granex O
14. Shed-a-leaf
15. Chlorate De Sodium
16. Val-drop
17. Chlorsaure
18. Chlorate Salt Of Sodium
19. Sodium Chlorate [iso]
20. Sodium Chlorate (naclo3)
21. Chloric Acid, Sodium Salt (1:1)
22. T95dr77gmr
23. Chebi:65242
24. 9011-70-5
25. Tumbleleaf
26. Kusatol
27. Rasikal
28. Tumbleaf
29. Defol
30. Dervan
31. Fall
32. Soda Chlorate
33. Drexel Defol
34. Harvest-aid
35. Mfcd00003479
36. Weed Killer
37. Drop-leaf
38. Hibar C
39. Leafex 2
40. Leafex 3
41. Chlorsaure [german]
42. Ortho-c-1-defoliant
43. Caswell No. 753
44. Natriumchloraat [dutch]
45. Natriumchlorat [german]
46. Sodium Chlorate Solution
47. Grain Sorghum Harvest-aid
48. Natrium Chloraat [dutch]
49. Natrium Chlorat [german]
50. Natriumchloraat
51. Natriumchlorat
52. Sodium;chlorate
53. Natrium Chlorat
54. Sodium(chlorate De) [french]
55. Ccris 9185
56. Hsdb 732
57. Natrium Chloraat
58. Sodio (clorato Di) [italian]
59. Sodium (chlorate De) [french]
60. United Chemical Defoliant No. 1
61. Chlorate De Sodium [iso-french]
62. Ortho C-1 Defoliant & Weed Killer
63. Einecs 231-887-4
64. Shed-a-leaf L
65. Sodium(chlorate De)
66. Sodio (clorato Di)
67. Un1495
68. Un2428
69. Epa Pesticide Chemical Code 073301
70. Sodium (chlorate De)
71. Sodium Chlorate Solution (50% Or Less)
72. Naclo3
73. Sodium Chlorate [un1495] [oxidizer]
74. Dsstox_cid_6025
75. Unii-t95dr77gmr
76. Dsstox_rid_77987
77. Dsstox_gsid_26025
78. Sodium Chlorate [ii]
79. Sodium Chlorate [mi]
80. Sodium Chlorate [hsdb]
81. Sodium Chlorate [inci]
82. Chembl1559268
83. Dtxsid7026025
84. Sodium Chlorate [mart.]
85. Sodium Chlorate [who-dd]
86. Sodium Chlorate, Aqueous Solution
87. Nsc41881
88. Tox21_202133
89. Nsc-41881
90. Akos015843818
91. Ncgc00091465-01
92. Ncgc00259682-01
93. Cas-7775-09-9
94. Sodium Chlorate, Acs Reagent, >=99.0%
95. Sodium Chlorate, Reagentplus(r), >=99%
96. Sodium Chlorate, P.a., Acs Reagent, 99%
97. 3-(4-isobutyl-phenyl)-propionicacid
98. Ft-0694099
99. Sodium Chlorate, Saj First Grade, >=98.0%
100. C18765
101. Ec 231-887-4
102. Q407446
103. Sodium Chlorate, Puriss. P.a., >=99.0% (t)
104. Sodium Chlorate, Aqueous Solution [un2428] [oxidizer]
105. Sodium Chlorate - Metastable High Temperature (255c) Phase Iii
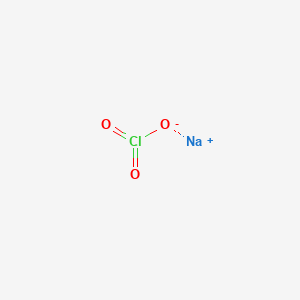
| Molecular Weight | 106.44 g/mol |
|---|---|
| Molecular Formula | ClNaO3 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 0 |
| Exact Mass | 105.9433658 g/mol |
| Monoisotopic Mass | 105.9433658 g/mol |
| Topological Polar Surface Area | 57.2 Ų |
| Heavy Atom Count | 5 |
| Formal Charge | 0 |
| Complexity | 49.8 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
At one time, the chlorate salts, sodium chlorate and potassium chlorate, were used as medicinal agents to treat inflammatory and ulcerative lesions of the oral cavity and could be found in various mouthwash, toothpaste, and gargle preparations. /Former use/
Goldfrank, L.R., Goldfrank's Toxicologic Emergencies 8th Ed. 2006., McGraw-Hill, New York, N.Y., p. 1393
A dose of 5-10 g can prove fatal in adults, as can a dose of 2 g in small children.
Hartley, D. and H. Kidd (eds.). The Agrochemicals Handbook. 2nd ed. Lechworth, Herts, England: The Royal Society of Chemistry, 1987., p. A294/Aug 87
Herbicides
Pesticides used to destroy unwanted vegetation, especially various types of weeds, grasses (POACEAE), and woody plants. Some plants develop HERBICIDE RESISTANCE. (See all compounds classified as Herbicides.)
Oral administration of 3 mL (5mg/L) to rats have shown: absorption T1/2: 1.74 hr, elimination T1/2:36.7 hr. Distribution mainly in blood. Excretion in feces and urine: 43 % of intial dose in 74 hr. Increased formation of chloroform in the liver.
European Chemicals Bureau; IUCLID Dataset, sodium chlorate (7775-09-9) p.38 (2000 CD-ROM edition). Available from, as of July 22, 2008: https://esis.jrc.ec.europa.eu/
Sodium chlorate is unlikely to be absorbed by the skin based on its high water solubility and ionic nature...
USEPA/Office of Pesticide Programs; Reregistration Eligibility Decision Document for Inorganic Chlorates p.8 EPA 738-R-06-014 (July 2006) Available from, as of August 3, 2008: https://www.epa.gov/oppsrrd1/REDs/inorganicchlorates_red.pdf
It is readily absorbed through both roots and leaves and may be translocated throughout plant.
White-Stevens, R. (ed.). Pesticides in the Environment: Volume 1, Part 1, Part 2. New York: Marcel Dekker, Inc., 1971., p. 35
Absorbed through the roots and foliage, with translocation basipetally in the xylem, as the phloem tissue is killed. The rate of respiration is increased, catalase activity decreased, and the plant's food reserves are depleted.
Hartley, D. and H. Kidd (eds.). The Agrochemicals Handbook. 2nd ed. Lechworth, Herts, England: The Royal Society of Chemistry, 1987., p. A294/Aug 87
Oral administration of 3 mL (5mg/L) to rats have shown: absorption half life: 1.74 hr, elimination half life: 36.7 hr.
European Chemicals Bureau; IUCLID Dataset, sodium chlorate (7775-09-9) p.38 (2000 CD-ROM edition). Available from, as of July 22, 2008: https://esis.jrc.ec.europa.eu/
Market Place

ABOUT THIS PAGE
24
PharmaCompass offers a list of Sodium Chlorate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Sodium Chlorate manufacturer or Sodium Chlorate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Sodium Chlorate manufacturer or Sodium Chlorate supplier.
A Sodium Chlorate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Sodium Chlorate, including repackagers and relabelers. The FDA regulates Sodium Chlorate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Sodium Chlorate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Sodium Chlorate supplier is an individual or a company that provides Sodium Chlorate active pharmaceutical ingredient (API) or Sodium Chlorate finished formulations upon request. The Sodium Chlorate suppliers may include Sodium Chlorate API manufacturers, exporters, distributors and traders.
Sodium Chlorate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Sodium Chlorate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Sodium Chlorate GMP manufacturer or Sodium Chlorate GMP API supplier for your needs.
A Sodium Chlorate CoA (Certificate of Analysis) is a formal document that attests to Sodium Chlorate's compliance with Sodium Chlorate specifications and serves as a tool for batch-level quality control.
Sodium Chlorate CoA mostly includes findings from lab analyses of a specific batch. For each Sodium Chlorate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Sodium Chlorate may be tested according to a variety of international standards, such as European Pharmacopoeia (Sodium Chlorate EP), Sodium Chlorate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Sodium Chlorate USP).

