
Synopsis
Synopsis
0
EU WC
0
NDC API
0
VMF
0
Australia
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
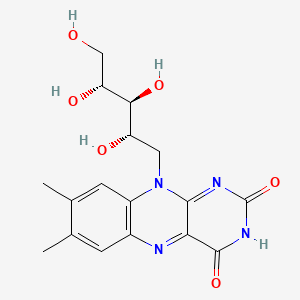

1. Vitamin B 2
2. Vitamin B2
3. Vitamin G
1. Vitamin B2
2. Lactoflavin
3. Riboflavine
4. 83-88-5
5. Vitamin G
6. (-)-riboflavin
7. Lactoflavine
8. Flavaxin
9. Beflavin
10. Riboflavinum
11. Riboflavina
12. Russupteridine Yellow Iii
13. Beflavine
14. Bisulase
15. 7,8-dimethyl-10-ribitylisoalloxazine
16. 6,7-dimethyl-9-d-ribitylisoalloxazine
17. Ribocrisina
18. Vitaflavine
19. Flaxain
20. Lactobene
21. Ribipca
22. Riboderm
23. Ribosyn
24. Ribotone
25. Ribovel
26. E101
27. Vitamin Bi
28. Flavin Bb
29. Vitasan B2
30. Hyre
31. Lacto-flavin
32. Riboflavin (vitamin B2)
33. C.i. Food Yellow 15
34. Vitamin-b2
35. Vitamin B-2
36. Riboflavinequinone
37. Chebi:17015
38. 7,8-dimethyl-10-(d-ribo-2,3,4,5-tetrahydroxypentyl)isoalloxazine
39. Ins No.101(iii)
40. Ins-101(iii)
41. 7,8-dimethyl-10-[(2s,3s,4r)-2,3,4,5-tetrahydroxypentyl]benzo[g]pteridine-2,4-dione
42. Vitamin B2 (as Riboflavin)
43. E-101(iii)
44. Tlm2976ofr
45. C.i. 50900
46. Ins No. 101(i)
47. Nsc-33298
48. 1-deoxy-1-(7,8-dimethyl-2,4-dioxo-3,4-dihydrobenzo[g]pteridin-10(2h)-yl)-d-ribitol
49. 7,8-dimethyl-10-((2s,3s,4r)-2,3,4,5-tetrahydroxypentyl)benzo[g]pteridine-2,4(3h,10h)-dione
50. D-ribitol, 1-deoxy-1-(3,4-dihydro-7,8-dimethyl-2,4-dioxobenzo(g)pteridin-10(2h)-yl)-
51. Nci-0033298
52. Mfcd00005022
53. Nsc 33298
54. Dsstox_cid_1777
55. Dsstox_rid_76321
56. Dsstox_gsid_21777
57. Riboflavine [inn-french]
58. Riboflavinum [inn-latin]
59. Riboflavina [inn-spanish]
60. Isoalloxazine, 7,8-dimethyl-10-d-ribityl-
61. Hsdb 817
62. 1-deoxy-1-(7,8-dimethyl-2,4-dioxo-3,4-dihydrobenzo[g]pteridin-10(2h)-yl)pentitol
63. 7,8-dimethyl-10-(d-ribo-2,3,4,5-tetrahydroxypentyl)benzo[g]pteridine-2,4(3h,10h)-dione
64. Ccris 1904
65. Isoalloxazine, 7,8-dimethyl-10-(d-ribo-2,3,4,5-tetrahydroxypentyl)-
66. Riboflavin (vit B2)
67. Einecs 201-507-1
68. Unii-tlm2976ofr
69. Riboflavin [usp:inn:ban]
70. Ai3-14697
71. 1kyv
72. 2ccb
73. 2vxa
74. ()-riboflavin
75. San Yellow B
76. Cas-83-88-5
77. Ncgc00017291-05
78. Bisulase (tn)
79. Food Yellow 15
80. Prestwick_442
81. 2fl5
82. 2vx9
83. 4d1y
84. Riboflavin [ii]
85. Riboflavin [mi]
86. Riboflavin [fcc]
87. Riboflavin [inn]
88. Riboflavin [jan]
89. Vitamin B2; E101
90. Prestwick3_000634
91. Riboflavin [hsdb]
92. Riboflavin [inci]
93. Lactoflavin [inci]
94. Riboflavin [vandf]
95. Epitope Id:161730
96. Riboflavinum [hpus]
97. Riboflavin [mart.]
98. Schembl7706
99. Chembl1534
100. Riboflavin [usp-rs]
101. Riboflavin [who-dd]
102. Riboflavin [who-ip]
103. Vitamin B2 [vandf]
104. Bspbio_000628
105. Mls001066391
106. Bpbio1_000692
107. Gtpl6578
108. Riboflavin (jp17/usp/inn)
109. Riboflavine; Vitamin B2
110. Dtxsid8021777
111. Riboflavin [orange Book]
112. Riboflavin [ep Monograph]
113. Vitamin B2 [green Book]
114. (-)-riboflavin, 97-103%
115. Hms2096p10
116. Riboflavin [usp Monograph]
117. Benzo[g]pteridine Riboflavin Deriv.
118. Hy-b0456
119. Riboflavinum [who-ip Latin]
120. Zinc2036848
121. Tox21_110813
122. Tox21_111714
123. Tox21_201633
124. Tox21_302980
125. 6,7-dimethyl-9-ribitylisoalloxazine
126. Bdbm50362895
127. S2540
128. Riboflavin (b2), Analytical Standard
129. Tox21_111714_1
130. Db00140
131. Ncgc00091288-01
132. Ncgc00091288-02
133. Ncgc00091288-03
134. Ncgc00091288-04
135. Ncgc00091288-05
136. Ncgc00179498-01
137. Ncgc00256408-01
138. Ncgc00259182-01
139. As-15936
140. Smr000112236
141. R0020
142. (-)-riboflavin, Tested According To Ph.eur.
143. C00255
144. D00050
145. (-)-riboflavin, Meets Usp Testing Specifications
146. A840676
147. A934900
148. Q130365
149. (-)-riboflavin, From Eremothecium Ashbyii, >=98%
150. W-104132
151. (-)-riboflavin, Acrylamide Photopolymerization Tested
152. 7,8-dimethyl-10-(1'-d-ribityl)isoalloxazine
153. Riboflavin Sodium Phosphate Impurity D [ep Impurity]
154. Riboflavin, European Pharmacopoeia (ep) Reference Standard
155. Riboflavin, United States Pharmacopeia (usp) Reference Standard
156. Riboflavin, Pharmaceutical Secondary Standard; Certified Reference Material
157. 5-deoxy-5-(7,8-dimethyl-2,4-dioxo-3,4-dihydrobenzo[g]pteridin-10(2h)-yl)-d-ribitol
158. 7,8-dimethyl-10-(d-ribo-2,3,4,5-tetrahydroxypentyl)-benzo[g]pteridine-2,4(3h,10h)-dione
159. Riboflavin For Peak Identification, European Pharmacopoeia (ep) Reference Standard
160. (-)-riboflavin (vitamin B2) Solution, 100 Mug/ml (methanol:0.1% Ammonium Acetate In Water (1:1)), Ampule Of 1 Ml, Analytical Standard
161. (-)-riboflavin, 100 Mug/ml (1% Ammonium Acetate In 50:50 Methanol:water), Certified Reference Material, Ampule Of 1 Ml
162. (-)-riboflavin, Bioreagent, Suitable For Cell Culture, Suitable For Insect Cell Culture, >=98%
163. (-)-riboflavin-13c4,15n2 (vitamin B2-13c4,15n2) Solution, 100 Mug/ml (1% Ammonium Acetate In 50:50 Methanol:water), Certified Reference Material, Ampule Of 1 Ml
164. 7,8-dimethyl-10-[(2s,3s,4r)-2,3,4,5-tetrahydroxypentyl]-2h,3h,4h,10h-benzo[g]pteridine-2,4-dione
165. 7,8-dimethyl-10-[(2s,3s,4r)-2,3,4,5-tetrakis(oxidanyl)pentyl]benzo[g]pteridine-2,4-dione
| Molecular Weight | 376.4 g/mol |
|---|---|
| Molecular Formula | C17H20N4O6 |
| XLogP3 | -1.5 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 5 |
| Exact Mass | 376.13828437 g/mol |
| Monoisotopic Mass | 376.13828437 g/mol |
| Topological Polar Surface Area | 155 Ų |
| Heavy Atom Count | 27 |
| Formal Charge | 0 |
| Complexity | 680 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 3 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Riboflavin is used to prevent riboflavin deficiency and to treat ariboflavinosis. Whenever possible, poor dietary habits should be corrected, and many clinicians recommend administration of multivitamin preparations containing riboflavin in patients with vitamin deficiencies since poor dietary habits often result in concurrent deficiencies.
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009)
Riboflavin may be useful in treating microcytic anemia that occurs in patients with a familial metabolic disease associated with splenomegaly and glutathione reductase deficiency.
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009)
Although riboflavin has not been shown by well-controlled trials to have any therapeutic value, the drug also has been used for the management of acne, congenital methemoglobinemia, muscle cramps, and burning feet syndrome.
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009)
People undergoing hemodialysis or peritioneal dialysis and those with severe malabsorption are likely to require extra riboflavin. Women who are carrying more than one fetus or breastfeeding more than one infant are also likely to require more riboflavin. It is possible that individuals who are ordinarily extremely physically active may also have increased needs for riboflavin.
Otten JJ, Hellwig JP, Meyers LD, eds; Dietary Reference Intakes: The Essential Guide to Nutrient Requirements, Washington, DC: The National Academies Press, 2006, p. 276
For more Therapeutic Uses (Complete) data for Riboflavin (11 total), please visit the HSDB record page.
Riboflavin may cause urine to have a more yellow color than normal, especially if large doses are taken. This is to be expected and is no cause for alarm. Usually, however, riboflavin does not cause any side effects.
Novak, K.M. (ed.). Drug Facts and Comparisons2008 Edition. Wolters Kluwer Health. St. Louis, Missouri 2008., p. 18
No short-term side effects /were observed/ in 49 patients treated with 400 mg/day of riboflavin taken with meals for at least 3 months. One patient receiving riboflavin and aspirin withdrew from the study because of gastric upset. This isolated finding may be an anomaly because no side effects were reported in other patients.
NAS, Food and Nutrition Board, Institute of Medicine; Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. National Academy Press, Washington, D.C., pg. 115, 1998. Available from, as of March 2, 2010: https://www.nap.edu/catalog/6015.html
Maternal Medication usually Compatible with Breast-Feeding: Riboflavin: Reported Sign or Symptom in Infant or Effect on Lactation: None. /from Table 6/
Report of the American Academy of Pediatrics Committee on Drugs in Pediatrics 93 (1): 141 (1994)
Infants treated for hyperbilirubinemia may also be sensitive to excess riboflavin.
NAS, Food and Nutrition Board, Institute of Medicine; Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. National Academy Press, Washington, D.C., pg. 116, 1998. Available from, as of March 2, 2010: https://www.nap.edu/catalog/6015.html
For more Drug Warnings (Complete) data for Riboflavin (6 total), please visit the HSDB record page.
For the treatment of ariboflavinosis (vitamin B2 deficiency).
Riboflavin or vitamin B2 is an easily absorbed, water-soluble micronutrient with a key role in maintaining human health. Like the other B vitamins, it supports energy production by aiding in the metabolising of fats, carbohydrates, and proteins. Vitamin B2 is also required for red blood cell formation and respiration, antibody production, and for regulating human growth and reproduction. It is essential for healthy skin, nails, hair growth and general good health, including regulating thyroid activity. Riboflavin also helps in the prevention or treatment of many types of eye disorders, including some cases of cataracts.
Photosensitizing Agents
Drugs that are pharmacologically inactive but when exposed to ultraviolet radiation or sunlight are converted to their active metabolite to produce a beneficial reaction affecting the diseased tissue. These compounds can be administered topically or systemically and have been used therapeutically to treat psoriasis and various types of neoplasms. (See all compounds classified as Photosensitizing Agents.)
Vitamin B Complex
A group of water-soluble vitamins, some of which are COENZYMES. (See all compounds classified as Vitamin B Complex.)
A - Alimentary tract and metabolism
A11 - Vitamins
A11H - Other plain vitamin preparations
A11HA - Other plain vitamin preparations
A11HA04 - Riboflavin (vit B2)
S - Sensory organs
S01 - Ophthalmologicals
S01X - Other ophthalmologicals
S01XA - Other ophthalmologicals
S01XA26 - Riboflavin
Absorption
Vitamin B2 is readily absorbed from the upper gastrointestinal tract.
Riboflavin is readily absorbed from the upper GI tract; however, absorption of the drug involves active transport mechanisms and the extent of GI absorption is limited by the duration of contact of the drug with the specialized segment of mucosa where absorption occurs. Riboflavin 5-phosphate is rapidly and almost completely dephosphorylated in the GI lumen before absorption occurs. The extent of GI absorption of riboflavin is increased when the drug is administered with food and is decreased in patients with hepatitis, cirrhosis, biliary obstruction, or in those receiving probenecid.
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009)
Primary absorption of riboflavin occurs in the small intestine via a rapid, saturable transport system. A small amount is absorbed in the large intestine. The rate of absorption is proportional to intake, and it increases when riboflavin is ingested along with other foods and in the presence of bile salts. At low intake levels, most absorption of riboflavin occurs via an active or facilitated transport system. At higher levels of intake, riboflavin can be absorbed by passive diffusion.
Otten JJ, Hellwig JP, Meyers LD, eds; Dietary Reference Intakes: The Essential Guide to Nutrient Requirements, Washington, DC: The National Academies Press, 2006, pp. 275-6
In the plasma, a large portion of riboflavin associates with other proteins, mainly immunoglobulins, for transport. Pregnancy increases the level of carrier proteins available for riboflavin, which results in a higher rate of riboflavin uptake at the maternal surface of the placenta.
Otten JJ, Hellwig JP, Meyers LD, eds; Dietary Reference Intakes: The Essential Guide to Nutrient Requirements, Washington, DC: The National Academies Press, 2006, p. 276
In the stomach, gastric acidification releases most of the coenzyme forms of riboflavin (flavin-adenine dinucleotide (FAD) and flavin mononucleotide (FMN)) from the protein. The noncovalently bound coenzymes are then hydrolyzed to riboflavin by nonspecific pyrophosphatases and phosphatases in the upper gut. Primary absorption of riboflavin occurs in the proximal small intestine via a rapid, saturable transport system. The rate of absorption is proportional to intake, and it increases when riboflavin is ingested along with other foods and in the presence of bile salts. A small amount of riboflavin circulates via the enterohepatic system. At low intake levels most absorption of riboflavin is via an active or facilitated transport system.
NAS, Food and Nutrition Board, Institute of Medicine; Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. National Academy Press, Washington, D.C., pg. 88, 1998. Available from, as of March 2, 2010: https://www.nap.edu/catalog/6015.html
For more Absorption, Distribution and Excretion (Complete) data for Riboflavin (16 total), please visit the HSDB record page.
Hepatic.
Free riboflavin is converted in the intestinal mucosa into flavin mononucleotide which is transformed into flavin adenine dinucleotide in the liver.
Evaluations of Drug Interactions. 2nd ed. and supplements. Washington, DC: American Pharmaceutical Assn., 1976, 1978., p. 453
The metabolism of riboflavin is a tightly controlled process that depends on the riboflavin status of the individual. Riboflavin is converted to coenzymes within the cellular cytoplasm of most tissues but mainly in the small intestine, liver, heart, and kidney. The metabolism of riboflavin begins with the adenosine triphosphate (ATP)-dependent phosphorylation of the vitamin to flavin mononucleotide (FMN). Flavokinase, the catalyst for this conversion, is under hormonal control. FMN can then be complexed with specific apoenzymes to form a variety of flavoproteins; however, most is converted to flavin-adenine dinucleotide (FAD) by FAD synthetase. As a result, FAD is the predominant flavocoenzyme in body tissues. Production of FAD is controlled by product inhibition such that an excess of FAD inhibits its further production.
NAS, Food and Nutrition Board, Institute of Medicine; Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. National Academy Press, Washington, D.C., pg. 89, 1998. Available from, as of March 2, 2010: https://www.nap.edu/catalog/6015.html
The biosynthesis of one riboflavin molecule requires one molecule of GTP and two molecules of ribulose 5-phosphate as substrates. GTP is hydrolytically opened, converted into 5-amino-6-ribitylamino-2,4(1H,3H)-pyrimidinedione by a sequence of deamination, side chain reduction and dephosphorylation. Condensation with 3,4-dihydroxy-2-butanone 4-phosphate obtained from ribulose 5-phosphate leads to 6,7-dimethyl-8-ribityllumazine. The final step in the biosynthesis of the vitamin involves the dismutation of 6,7-dimethyl-8-ribityllumazine catalyzed by riboflavin synthase. The mechanistically unusual reaction involves the transfer of a four-carbon fragment between two identical substrate molecules. The second product, 5-amino-6-ribitylamino-2,4(1H,3H)-pyrimidinedione, is recycled in the biosynthetic pathway by 6,7-dimethyl-8-ribityllumazine synthase. This article will review structures and reaction mechanisms of riboflavin synthases and related proteins up to 2007 and 122 references are cited.
PMID:18298940 Fischer M, Bacher A; Arch Biochem Biophys 474 (2): 252-65 (2008).
66-84 minutes
The biologic half-life of riboflavin is about 66-84 minutes following oral or IM administration of a single large dose in healthy individuals.
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009)
Binds to riboflavin hydrogenase, riboflavin kinase, and riboflavin synthase. Riboflavin is the precursor of flavin mononucleotide (FMN, riboflavin monophosphate) and flavin adenine dinucleotide (FAD). The antioxidant activity of riboflavin is principally derived from its role as a precursor of FAD and the role of this cofactor in the production of the antioxidant reduced glutathione. Reduced glutathione is the cofactor of the selenium-containing glutathione peroxidases among other things. The glutathione peroxidases are major antioxidant enzymes. Reduced glutathione is generated by the FAD-containing enzyme glutathione reductase.
Riboflavin is converted to 2 coenzymes, flavin mononucleotide (FMN) and flavin adenine dinucleotide (FAD), which are necessary for normal tissue respiration. Riboflavin is also required for activation of pyridoxine, conversion of tryptophan to niacin, and may be involved in maintaining erythrocyte integrity.
USP Convention. USPDI-Drug Information for the Health Care Professional. 14th ed. Volume I. Rockville, MD: United States Pharmacopeial Convention, Inc., 1994. (Plus Updates)., p. 2400
Riboflavin functions as the coenzyme for flavin adenine dinucleotide (FAD) and flavin mononucleotide (FMN), which primarily influence hydrogen transport in oxidative enzyme systems (eg, cytochrome C reductase, succinic dehydrogenase, xanthine oxidase).
American Medical Association, Council on Drugs. AMA Drug Evaluations Annual 1994. Chicago, IL: American Medical Association, 1994., p. 2171
Two active forms of riboflavin exist ... coenzyme flavin mononucleotide (FMN) and coenzyme flavin adenine dinucleotide (FAD). They are formed by reaction of riboflavin with 1 and 2 molecules of ATP as follow: riboflavin + ATP = riboflavin-P (FMN) + ADP; FMN + ATP = riboflavin-ADP (FAD) + PP.
Di Palma, J. (ed.). Drill's Pharmacology in Medicine. 4th ed. New York: McGraw Hill Book Co., 1971., p. 1285
Riboflavin is a water-soluble, yellow, fluorescent compound. The primary form of the vitamin is as an integral component of the coenzymes flavin mononucleotide (FMN) and flavin-adenine dinucleotide (FAD). It is in these bound coenzyme forms that riboflavin functions as a catalyst for redox reactions in numerous metabolic pathways and in energy production. ... The redox reactions in which flavocoenzymes participate include flavoprotein-catalyzed dehydrogenations that are both pyridine nucleotide (niacin) dependent and independent, reactions with sulfur-containing compounds, hydroxylations, oxidative decarboxylations (involving thiamin as its pyrophosphate), dioxygenations, and reduction of oxygen to hydrogen peroxide. There are obligatory roles of flavocoenzymes in the formation of some vitamins and their coenzymes. For example, the biosynthesis of two niacin-containing coenzymes from tryptophan occurs via FAD-dependent kynurenine hydroxylase, an FMN-dependent oxidase catalyzes the conversion of the 5'-phosphates of vitamin B6 to coenzymic pyridoxal 5'-phosphate, and an FAD-dependent dehydrogenase reduces 5,10-methylene-tetrahydrofolate to the 5'-methyl product that interfaces with the B12-dependent formation of methionine from homocysteine and thus with sulfur amino acid metabolism.
NAS, Food and Nutrition Board, Institute of Medicine; Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. National Academy Press, Washington, D.C., pg. 87-88, 1998. Available from, as of March 2, 2010: https://www.nap.edu/catalog/6015.html
For more Mechanism of Action (Complete) data for Riboflavin (7 total), please visit the HSDB record page.
 Rochem, your partner in developing, sourcing, and supplying pharmaceutical & animal health ingredients of Chinese origin.
Rochem, your partner in developing, sourcing, and supplying pharmaceutical & animal health ingredients of Chinese origin.
 Cerata Pharmaceuticals LLP: WHO-GMP Certified Leading Manufacturer & Exporter of Steroid-Hormone & Peptide APIs From India.
Cerata Pharmaceuticals LLP: WHO-GMP Certified Leading Manufacturer & Exporter of Steroid-Hormone & Peptide APIs From India.
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 3541
Submission : 1979-05-31
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 4407
Submission : 1981-11-25
Status : Inactive
Type : II

Certificate Number : R1-CEP 2011-069 - Rev 00
Issue Date : 2017-11-07
Type : Chemical
Substance Number : 292
Status : Valid

Registration Number : 228MF10235
Registrant's Address : 14-16F KCCI Bldg. , 39, Sejongdaero, Jung-gu, Seoul, Republic of Korea
Initial Date of Registration : 2016-12-26
Latest Date of Registration :


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 15576
Submission : 2001-08-06
Status : Inactive
Type : II

Registration Number : 217MF10986
Registrant's Address : 14-16F KCCI Bldg. , 39, Sejongdaero, Jung-gu, Seoul, Republic of Korea
Initial Date of Registration : 2005-11-21
Latest Date of Registration :


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 18846
Submission : 2005-10-10
Status : Inactive
Type : II




Certificate Number : CEP 2000-088 - Rev 03
Issue Date : 2026-01-16
Type : Chemical
Substance Number : 292
Status : Valid

Registration Number : 217MF11283
Registrant's Address : Wurmisweg 576, CH-4303, Kaiseraugst, Switzerland
Initial Date of Registration : 2005-12-22
Latest Date of Registration :



Certificate Number : R1-CEP 2013-010 - Rev 01
Issue Date : 2022-10-06
Type : Chemical
Substance Number : 292
Status : Valid

Registrant Name : Daeshin Muyak Co., Ltd.
Registration Date : 2025-05-20
Registration Number : 20250305-211-J-1762(1)
Manufacturer Name : Hubei Guangji Pharmaceutical Co., Ltd.
Manufacturer Address : B area of Dajin Guangji pharmaceutical Bio-Industry Park, Wuxue City, Hubei Province, China.


 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
93
PharmaCompass offers a list of Riboflavin API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Riboflavin manufacturer or Riboflavin supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Riboflavin manufacturer or Riboflavin supplier.
A Riboflavin manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Riboflavin, including repackagers and relabelers. The FDA regulates Riboflavin manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Riboflavin API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Riboflavin manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Riboflavin supplier is an individual or a company that provides Riboflavin active pharmaceutical ingredient (API) or Riboflavin finished formulations upon request. The Riboflavin suppliers may include Riboflavin API manufacturers, exporters, distributors and traders.
click here to find a list of Riboflavin suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Riboflavin DMF (Drug Master File) is a document detailing the whole manufacturing process of Riboflavin active pharmaceutical ingredient (API) in detail. Different forms of Riboflavin DMFs exist exist since differing nations have different regulations, such as Riboflavin USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Riboflavin DMF submitted to regulatory agencies in the US is known as a USDMF. Riboflavin USDMF includes data on Riboflavin's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Riboflavin USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Riboflavin suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Riboflavin Drug Master File in Japan (Riboflavin JDMF) empowers Riboflavin API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Riboflavin JDMF during the approval evaluation for pharmaceutical products. At the time of Riboflavin JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Riboflavin suppliers with JDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Riboflavin Drug Master File in Korea (Riboflavin KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Riboflavin. The MFDS reviews the Riboflavin KDMF as part of the drug registration process and uses the information provided in the Riboflavin KDMF to evaluate the safety and efficacy of the drug.
After submitting a Riboflavin KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Riboflavin API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Riboflavin suppliers with KDMF on PharmaCompass.
A Riboflavin CEP of the European Pharmacopoeia monograph is often referred to as a Riboflavin Certificate of Suitability (COS). The purpose of a Riboflavin CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Riboflavin EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Riboflavin to their clients by showing that a Riboflavin CEP has been issued for it. The manufacturer submits a Riboflavin CEP (COS) as part of the market authorization procedure, and it takes on the role of a Riboflavin CEP holder for the record. Additionally, the data presented in the Riboflavin CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Riboflavin DMF.
A Riboflavin CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Riboflavin CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Riboflavin suppliers with CEP (COS) on PharmaCompass.
Riboflavin Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Riboflavin GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Riboflavin GMP manufacturer or Riboflavin GMP API supplier for your needs.
A Riboflavin CoA (Certificate of Analysis) is a formal document that attests to Riboflavin's compliance with Riboflavin specifications and serves as a tool for batch-level quality control.
Riboflavin CoA mostly includes findings from lab analyses of a specific batch. For each Riboflavin CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Riboflavin may be tested according to a variety of international standards, such as European Pharmacopoeia (Riboflavin EP), Riboflavin JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Riboflavin USP).

