
Synopsis
Synopsis
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
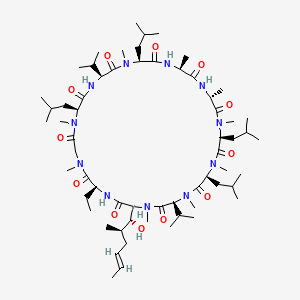

1. Ciclosporin
2. Csa Neoral
3. Csa-neoral
4. Csaneoral
5. Cya Nof
6. Cya-nof
7. Cyclosporin
8. Cyclosporine
9. Cyclosporine A
10. Neoral
11. Ol 27 400
12. Ol 27-400
13. Ol 27400
14. Sandimmun
15. Sandimmun Neoral
16. Sandimmune
1. Ciclosporin
2. Cyclosporine
3. 59865-13-3
4. Cyclosporine A
5. Sandimmune
6. Neoral
7. Sandimmun
8. Cyclosporin
9. Ramihyphin A
10. Ciclosporine
11. Equoral
12. Gengraf
13. Neoplanta
14. Sang-35
15. Ciclosporinum
16. Sandimmun Neoral
17. Zinograf Me
18. Ciclosporina
19. Consupren
20. Mitogard
21. Optimmune
22. Ciclosporin A
23. Neurostat
24. Atopica
25. Antibiotic S 7481f1
26. Ciclosporin [inn]
27. Sangcya
28. Cyclosporine, Modified
29. Cyclosporine Microemulsion
30. Csa
31. 83hn0gtj6d
32. Sdz-oxl-400
33. Mls001333756
34. Chebi:4031
35. Antibiotic S-7481f1
36. S-neoral
37. Cipol N
38. Sigmasporin Microoral
39. Nsc-290193
40. Cyclosporin A Solution
41. Ol-27-400
42. Dsstox_cid_365
43. Mfcd00274558
44. Ciclosporin (ciclosporin A)
45. Dsstox_rid_75541
46. Dsstox_gsid_20365
47. (3s,6s,9s,12r,15s,18s,21s,24s,30s,33s)-30-ethyl-33-[(e,1r,2r)-1-hydroxy-2-methylhex-4-enyl]-1,4,7,10,12,15,19,25,28-nonamethyl-6,9,18,24-tetrakis(2-methylpropyl)-3,21-di(propan-2-yl)-1,4,7,10,13,16,19,22,25,28,31-undecazacyclotritriacontane-2,5,8,11,14,17,20,23,26,29,32-undecone
48. Sdz-oxl 400
49. Ol 27-400
50. Abrammune
51. Imusporin
52. Seciera
53. Vekacia
54. Papilock Mini
55. Arpimune Me
56. Sandimmune Neoral
57. Ciclosporina Germed
58. (r-(r*,r*-(e)))-cyclic(l-alanyl-d-alanyl-n-methyl-l-leucyl-n-methyl-l-leucyl-n-methyl-l-valyl-3-hydroxy-n,4-dimethyl-l-2-amino-6-octenoyl-l-alpha-aminobutyryl-n-methylglycyl-n-methyl-l-leucyl-l-valyl-n-methyl-l-leucyl)
59. Cya
60. Cyclo(((e)-(2s,3r,4r)-3-hydroxy-4-methyl-2-(methylamino)-6-octenoyl)-l-2-aminobutyryl-n-methylglycyl-n-methyl-l-leucyl-l-valyl-n-methyl-l-leucyl-l-alanyl-d-alanyl-n-methyl-l-leucyl-n-methyl-l-leucyl-n-methyl-l-valyl)
61. Smr000058578
62. Cicloral (antibiotic)
63. Cyclosporine [usan]
64. Debio088
65. Sang 35
66. Cyclosporine [usan:usp]
67. Unii-83hn0gtj6d
68. Ciclosporine [inn-french]
69. Ciclosporinum [inn-latin]
70. Ciclosporina [inn-spanish]
71. Cyclospori
72. Sigmasporin
73. Cyclokat
74. Ikervis
75. Papilock
76. Pulminiq
77. Zyclorin
78. Ciclomulsion
79. Ciclosporin;
80. Ccris 1590
81. Ciclosporin Dt
82. Consupren S
83. Nsc290193
84. Modusik-a
85. Hsdb 6881
86. 1cyn
87. 2wfj
88. 4jjm
89. Cipol-n
90. Ncgc00016890-01
91. (3s,6s,9s,12r,15s,18s,21s,24s,30s,33s)-30-ethyl-33-[(1r,2r,4e)-1-hydroxy-2-methylhex-4-en-1-yl]-6,9,18,24-tetraisobutyl-3,21-diisopropyl-1,4,7,10,12,15,19,25,28-nonamethyl-1,4,7,10,13,16,19,22,25,28,31-undecaazacyclotritriacontane-2,5,8,11,14,17,20,23,2
92. (3s,6s,9s,12r,15s,18s,21s,24s,30s,33s)-30-ethyl-33-[(1r,2r,4e)-1-hydroxy-2-methylhex-4-en-1-yl]-6,9,18,24-tetraisobutyl-3,21-diisopropyl-1,4,7,10,12,15,19,25,28-nonamethyl-1,4,7,10,13,16,19,22,25,28,31-undecaazacyclotritriacontane-2,5,8,11,14,17,20,23,26,29,32-undecone
93. (3s,6s,9s,12r,15s,18s,21s,24s,30s,33s)-30-ethyl-33-[(e,1r,2r)-1-hydroxy-2-methyl-hex-4-enyl]-6,9,18,24-tetraisobutyl-3,21-diisopropyl-1,4,7,10,12,15,19,25,28-nonamethyl-1,4,7,10,13,16,19,22,25,28,31-undecazacyclotritriacontane-2,5,8,11,14,17,20,23,26,29,32-undecone
94. (3s,6s,9s,12r,15s,18s,21s,24s,30s,33s)-30-ethyl-33-[(e,1r,2r)-1-hydroxy-2-methylhex-4-enyl]-1,4,7,10,12,15,19,25,28-nonamethyl-6,9,18,24-tetrakis(2-methylpropyl)-3,21-di(propan-2-yl)-1,4,7,10,13,16,19
95. (r-[r*,r*-(e)])-cyclic(l-alanyl-d-alanyl-n-methyl-l-leucyl-n-methyl-l-leucyl-n-methyl-l-valyl-3-hydroxy-n,4-dimethyl-l-2-amino-6-octenoyl-l-alpha-aminobutyryl-n-methylglycyl-n-methyl-l-leucyl-l-valyl-n-methyl-l-leucyl)
96. Cyclo[[(e)-(2s,3r,4r)-3-hydroxy-4-methyl-2-(methylamino)-6-octenoyl]-l-2-aminobutyryl-n-methylglycyl-n-methyl-l-leucyl-l-valyl-n-methyl-l-leucyl-l-alanyl-d-alanyl-n-methyl-l-leucyl-n-methyl-l-leucyl-n-methyl-l-valyl]
97. Drg-0275
98. Prestwick_731
99. Cas-59865-13-3
100. Cequa
101. Verkazia
102. Nsc 290193
103. Csa & Ifn.alpha.
104. 1c5f
105. 2z6w
106. S 7481f1
107. Cyclosporine Manufacturer
108. Prestwick2_000435
109. Prestwick3_000435
110. Ciclosporin [jan]
111. Chembl160
112. Sang-2000
113. Cyclosporine [hsdb]
114. Cyclosporine [iarc]
115. Cyclosporin A [mi]
116. Schembl3491
117. Schembl4442
118. Ciclosporin [mart.]
119. Cyclosporine [vandf]
120. Nova-22007
121. Ikervis (opthalmic Solution)
122. Bspbio_000450
123. Ciclosporin [who-dd]
124. Ciclosporin [who-ip]
125. Cyclosporin A & Ifn.alpha.
126. Mls000028376
127. Mls002153454
128. Mls002207033
129. Verkazia (opthalmic Solution)
130. Cyclosporine [usp-rs]
131. Bpbio1_000496
132. Gtpl1024
133. Dtxsid0020365
134. Chebi:92233
135. Ath-002
136. Cyclosporine [green Book]
137. Olo-400
138. Cb-01-09 Mmx
139. Ciclosporin [ep Monograph]
140. Cyclosporine [orange Book]
141. Hms1569g12
142. Hms2089a09
143. Hms2096g12
144. Hms2230m14
145. Hms3713g12
146. Cyclosporine [usp Impurity]
147. 1,11-cyclo[l-alanyl-d-alanyl-n-methyl-l-leucyl-n-methyl-l-leucyl-n-methyl-l-valyl-(e)-(2s,3r,4r)-2-amino-3-hydroxy-n,4-dimethyloct-6-enoyl-l-2-aminobutanoyl-n-methylglycyl-n-methyl-l-leucyl-l-valyl-n-methyl-l-leucine]
148. 30-ethyl-33-(1-hydroxy-2-methyl-hex-4-enyl)-6,9,18,24-tetraisobutyl-3,21-diisopropyl-1,4,7,10,12,15,19,25,28-nonamethyl-1,4,7,10,13,16,19,22,25,28,31undecaaza-cyclotritriacontan-2,5,8,11,14,17,20,23,26,29,32-undecaone
149. 30-ethyl-33-[(4e)-1-hydroxy-2-methylhex-4-en-1-yl]-1,4,7,10,12,15,19,25,28-nonamethyl-6,9,18,24-tetrakis(2-methylpropyl)-3,21-bis(propan-2-yl)-1,4,7,10,13,16,19,22,25,28,31-undecaazacyclotritriacontane-2,5,8,11,14,17,20,23,26,29,32-undecone
150. 30-ethyl-33-[(4e)-1-hydroxy-2-methylhex-4-en-1-yl]-6,9,18,24-tetraisobutyl-3,21-diisopropyl-1,4,7,10,12,15,19,25,28-nonamethyl-1,4,7,10,13,16,19,22,25,28,31-undecaazacyclotritriacontane-2,5,8,11,14,17,20,23,26,29,32-undecone
151. Cyclo(l-alanyl-d-alanyl-n-methyl-l-leucyl-n-methyl-l-leucyl-n-methyl-l-valyl-((3r,4r,6e)-6,7-didehydro-3-hydroxy-n,4-dimethyl-l-2-aminooctanoyl)-l-2-aminobutanoyl-n-methylglycyl-n-methyl-l-leucyl-l-valyl-n-methylleucyl)
152. Cyclosporin A, Ready Made Solution
153. Ex-a4110
154. Hy-b0579
155. Cyclosporine [usp Monograph]
156. Tox21_110667
157. Tox21_301849
158. Bdbm50022815
159. Ciclosporinum [who-ip Latin]
160. De-076
161. St-603
162. Cyclosporin A, >=98.5% (tlc)
163. Akos015969287
164. Tox21_110667_1
165. Ccg-208184
166. Db00091
167. Ks-1257
168. Sdccgsbi-0050230.p004
169. Ncgc00093704-12
170. Ncgc00164258-01
171. Ncgc00164258-02
172. Ncgc00164258-03
173. Ncgc00255232-01
174. Bc164336
175. Cyclo(l-alanyl-d-alanyl-n-methyl-l-leucyl-n-methyl-l-leucyl-n-methyl-l-valyl-((3r,4r,6e)-6,7-didehydro-3-hydroxy-n,4-dimethyl-l-2-aminooctanoyl-l-2-aminobutanoyl-n-methylglycyl-n-methyl-l-leucyl-l-valyl-n-methylleucyl)
176. Ol-27400
177. Sbi-0050230.p003
178. M01532
179. 865c133
180. A832514
181. Cyclosporin A, Vetranal(tm), Analytical Standard
182. Q367700
183. Sr-01000780563
184. Q-200913
185. Sr-01000780563-3
186. Brd-k03222093-001-01-8
187. Brd-k13533483-001-03-0
188. Ciclosporin, European Pharmacopoeia (ep) Reference Standard
189. Cyclosporin A, From Tolypocladium Inflatum, >=95% (hplc), Solid
190. Cyclosporine, United States Pharmacopeia (usp) Reference Standard
191. Ciclosporin For System Suitability, European Pharmacopoeia (ep) Reference Standard
192. Cyclosporin A, From Tolypocladium Inflatum, Bioreagent, For Molecular Biology, >=95%
193. Cyclosporine, Pharmaceutical Secondary Standard; Certified Reference Material
194. (3s,6s,9s,12r,15s,18s,21s,24s,30s,33s)-30-ethyl-33-((1r,2r,e)-1-hydroxy-2-methylhex-4-en-1-yl)-6,9,18,24-tetraisobutyl-3,21-diisopropyl-1,4,7,10,12,15,19,25,28-nonamethyl-1,4,7,10,13,16,19,22,25,28,31-undecaazacyclotritriacontan-2,5,8,11,14,17,20,23,26,29,32-undecaone
195. (3s,6s,9s,12r,15s,18s,21s,24s,30s,33s)-30-ethyl-33-[(1r,2r,4e)-1-hydroxy-2-methylhex-4-en-1-yl]-1,4,7,10,12,15,19,25,28-nonamethyl-6,9,18,24-tetrakis(2-methylpropyl)-3,21-bis(propan-2-yl)-1,4,7,10,13,16,19,22,25,28,31-undecaazacyclotritriacontan-2,5,8,11,14,17,20,23,26,29,32-undecone
196. (3s,6s,9s,12r,15s,18s,21s,24s,30s,33s)-30-ethyl-33-[(e,1r,2r)-1-hydroxy-2-methyl-pent-3-enyl]-3,6,9,18,24-pentaisobutyl-21-isopropyl-1,4,7,10,12,15,19,25,28-nonamethyl-1,4,7,10,13,16,19,22,25,28,31-undecazacyclotritriacontane-2,5,8,11,14,17,20,23,26,29,32
197. (r-(r*,r*-(e)))-cyclic(l-alanyl-d-alanyl-n-methyl-l-leucyl-n-methyl-l-leucyl-n-methyl-l-valyl-3-hydroxy-n,4-dimethyl-l-2-amino-6-octenoyl-l-.alpha.-aminobutyryl-n-methylglycyl-n-methyl-l-leucyl-l-valyl-n-methyl-l-leucyl)
198. 104250-72-8
199. 30-ethyl-33-((e)-1-hydroxy-2-methyl-hex-4-enyl)-6,9,18,24-tetraisobutyl-3,21-diisopropyl-1,4,7,10,12,15,19,28-octamethyl-1,4,7,10,13,16,19,22,25,28,31undecaaza-cyclotritriacontan-2,5,8,11,14,17,20,23,26,29,32-undecaone
| Molecular Weight | 1202.6 g/mol |
|---|---|
| Molecular Formula | C62H111N11O12 |
| XLogP3 | 7.5 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 15 |
| Exact Mass | 1201.84136802 g/mol |
| Monoisotopic Mass | 1201.84136802 g/mol |
| Topological Polar Surface Area | 279 Ų |
| Heavy Atom Count | 85 |
| Formal Charge | 0 |
| Complexity | 2330 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 12 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 8 | |
|---|---|
| Drug Name | Cyclosporine |
| PubMed Health | Cyclosporine |
| Drug Classes | Anti-Inflammatory, Immune Suppressant |
| Drug Label | Cyclosporine, the active principle in Sandimmune (cyclosporine) is a cyclic polypeptide immunosuppressant agent consisting of 11 amino acids. It is produced as a metabolite by the fungus species Beauveria nivea.Chemically, cyclosporine is designate... |
| Active Ingredient | Cyclosporine |
| Dosage Form | Injectable; Capsule; Solution |
| Route | Injection; Oral |
| Strength | 50mg/ml; 25mg; 100mg; 100mg/ml; 50mg |
| Market Status | Prescription |
| Company | Watson Labs; Wockhardt; Ivax Sub Teva Pharms; Apotex; Luitpold; Abbvie; Eurohlth Intl; Sandoz |
| 2 of 8 | |
|---|---|
| Drug Name | Gengraf |
| PubMed Health | Cyclosporine |
| Drug Classes | Anti-Inflammatory, Immune Suppressant |
| Drug Label | Gengraf Oral Solution (cyclosporine oral solution, USP [MODIFIED]) is a modified oral formulation of cyclosporine that forms an aqueous dispersion in an aqueous environment.Cyclosporine, the active principle in Gengraf Oral Solution, is a cyclic... |
| Active Ingredient | Cyclosporine |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 25mg; 100mg; 50mg |
| Market Status | Prescription |
| Company | Abbvie |
| 3 of 8 | |
|---|---|
| Drug Name | Neoral |
| PubMed Health | Cyclosporine |
| Drug Classes | Anti-Inflammatory, Immune Suppressant |
| Drug Label | Cyclosporine, the active principle in Sandimmune (cyclosporine) is a cyclic polypeptide immunosuppressant agent consisting of 11 amino acids. It is produced as a metabolite by the fungus species Beauveria nivea.Chemically, cyclosporine is designate... |
| Active Ingredient | Cyclosporine |
| Dosage Form | Capsule; Solution |
| Route | Oral |
| Strength | 25mg; 100mg; 100mg/ml |
| Market Status | Prescription |
| Company | Novartis |
| 4 of 8 | |
|---|---|
| Drug Name | Sandimmune |
| PubMed Health | Cyclosporine |
| Drug Classes | Anti-Inflammatory, Immune Suppressant |
| Drug Label | Cyclosporine, the active principle in Sandimmune (cyclosporine) is a cyclic polypeptide immunosuppressant agent consisting of 11 amino acids. It is produced as a metabolite by the fungus species Beauveria nivea.Chemically, cyclosporine is designate... |
| Active Ingredient | Cyclosporine |
| Dosage Form | Capsule; Injectable; Solution |
| Route | Injection; Oral |
| Strength | 25mg; 50mg/ml; 100mg; 100mg/ml; 50mg |
| Market Status | Prescription |
| Company | Novartis |
| 5 of 8 | |
|---|---|
| Drug Name | Cyclosporine |
| PubMed Health | Cyclosporine |
| Drug Classes | Anti-Inflammatory, Immune Suppressant |
| Drug Label | Cyclosporine, the active principle in Sandimmune (cyclosporine) is a cyclic polypeptide immunosuppressant agent consisting of 11 amino acids. It is produced as a metabolite by the fungus species Beauveria nivea.Chemically, cyclosporine is designate... |
| Active Ingredient | Cyclosporine |
| Dosage Form | Injectable; Capsule; Solution |
| Route | Injection; Oral |
| Strength | 50mg/ml; 25mg; 100mg; 100mg/ml; 50mg |
| Market Status | Prescription |
| Company | Watson Labs; Wockhardt; Ivax Sub Teva Pharms; Apotex; Luitpold; Abbvie; Eurohlth Intl; Sandoz |
| 6 of 8 | |
|---|---|
| Drug Name | Gengraf |
| PubMed Health | Cyclosporine |
| Drug Classes | Anti-Inflammatory, Immune Suppressant |
| Drug Label | Gengraf Oral Solution (cyclosporine oral solution, USP [MODIFIED]) is a modified oral formulation of cyclosporine that forms an aqueous dispersion in an aqueous environment.Cyclosporine, the active principle in Gengraf Oral Solution, is a cyclic... |
| Active Ingredient | Cyclosporine |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 25mg; 100mg; 50mg |
| Market Status | Prescription |
| Company | Abbvie |
| 7 of 8 | |
|---|---|
| Drug Name | Neoral |
| PubMed Health | Cyclosporine |
| Drug Classes | Anti-Inflammatory, Immune Suppressant |
| Drug Label | Cyclosporine, the active principle in Sandimmune (cyclosporine) is a cyclic polypeptide immunosuppressant agent consisting of 11 amino acids. It is produced as a metabolite by the fungus species Beauveria nivea.Chemically, cyclosporine is designate... |
| Active Ingredient | Cyclosporine |
| Dosage Form | Capsule; Solution |
| Route | Oral |
| Strength | 25mg; 100mg; 100mg/ml |
| Market Status | Prescription |
| Company | Novartis |
| 8 of 8 | |
|---|---|
| Drug Name | Sandimmune |
| PubMed Health | Cyclosporine |
| Drug Classes | Anti-Inflammatory, Immune Suppressant |
| Drug Label | Cyclosporine, the active principle in Sandimmune (cyclosporine) is a cyclic polypeptide immunosuppressant agent consisting of 11 amino acids. It is produced as a metabolite by the fungus species Beauveria nivea.Chemically, cyclosporine is designate... |
| Active Ingredient | Cyclosporine |
| Dosage Form | Capsule; Injectable; Solution |
| Route | Injection; Oral |
| Strength | 25mg; 50mg/ml; 100mg; 100mg/ml; 50mg |
| Market Status | Prescription |
| Company | Novartis |
Clinical indications for cyclosporine are kidney, liver, heart, & other organ transplantation; rheumatoid arthritis; & psoriasis. ... Cyclosporine usually is used in combination with other agents, especially glucocorticoids & either azathioprine or mycophenolate mofetil &, most recently, sirolimus. ... In rheumatoid arthritis, cyclosporine is used in cases of severe disease that have not responded to methotrexate. Cyclosporine can be used in combination with methotrexate, but the levels of both drugs must be monitored closely. In psoriasis, cyclosporine is indicated for treatment of adult nonimmunocompromised patients with severe & disabling disease who have failed other systemic therapies. Because of its mechanism of action, there is a theoretical bases for the use of cyclosporine in a variety of other T-cell-mediated diseases. Cyclosporine has been reported to be effective in Behcet's acute ocular syndrome, endogenous uveitis, atopic dermatitis, inflammatory bowel disease, & nephrotic syndrome when standard therapies have failed.
Hardman, J.G., L.E. Limbird, P.B. Molinoff, R.W. Ruddon, A.G. Goodman (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 9th ed. New York, NY: McGraw-Hill, 1996., p. 1468
For prevention of allograft rejection in adults and children ... .
Ellenhorn, M.J., S. Schonwald, G. Ordog, J. Wasserberger. Ellenhorn's Medical Toxicology: Diagnosis and Treatment of Human Poisoning. 2nd ed. Baltimore, MD: Williams and Wilkins, 1997., p. 780
Cyclosporine is indicated, usually in combination with corticosteroids, for prevention of rejection of renal, hepatic, and cardiac transplants (allografts). /Included in US product labeling/
MICROMEDEX Thomson Health Care. USPDI - Drug Information for the Health Care Professional 21 st ed. Volume 1. MICROMEDEX Thomson Health Care, Englewood, CO. 2001. Content Reviewed and Approved by the U.S. Pharmacopeial Convention, Inc., p. 1136
Cyclosporine is also indicated for prevention of rejection of heart-lung and pancreatic transplants. /NOT included in US product labeling/
MICROMEDEX Thomson Health Care. USPDI - Drug Information for the Health Care Professional 21 st ed. Volume 1. MICROMEDEX Thomson Health Care, Englewood, CO. 2001. Content Reviewed and Approved by the U.S. Pharmacopeial Convention, Inc., p. 1136
For more Therapeutic Uses (Complete) data for CYCLOSPORIN A (13 total), please visit the HSDB record page.
Non-PVC containers & administration sets should be used to administer cyclosporine solns. ... Use of glass containers & tubing that does not contain DEHP to administer cyclosporine was recommended.
Trissel, L.A. Handbook on Injectable Drugs. 9th ed. Bethesda, MD. American Society of Health-System Pharmacists' Product Development. 1996., p. 308
Cyclosporine is distributed into breast milk. Mothers taking cyclosporine should not breast-feed their babies, because of the potential risk of serious adverse effects (e.g., hypertension, nephrotoxicity, malignancy) in the infant.
MICROMEDEX Thomson Health Care. USPDI - Drug Information for the Health Care Professional 21 st ed. Volume 1. MICROMEDEX Thomson Health Care, Englewood, CO. 2001. Content Reviewed and Approved by the U.S. Pharmacopeial Convention, Inc., p. 1136
Appropriate studies performed to date in pediatric patients receiving cyclosporine for organ transplantation have not demonstrated pediatrics-specific problems that would limit the usefulness of cyclosporine in children. Cyclosporine has been used in pediatric patients 1 year of age and older receiving organ transplantations. Pediatric patients have increased clearance of cyclosporine as compared with adult patients. The safety and efficacy of cyclosporine to treat psoriasis and rheumatoid arthritis in pediatric patients have not been established.
MICROMEDEX Thomson Health Care. USPDI - Drug Information for the Health Care Professional 21 st ed. Volume 1. MICROMEDEX Thomson Health Care, Englewood, CO. 2001. Content Reviewed and Approved by the U.S. Pharmacopeial Convention, Inc., p. 1136
Geriatric patients were included in the clinical trials of cyclosporine to treat rheumatoid arthritis. Geriatric patients were more likely to experience hypertension and increases in serum creatinine concentrations than were younger adult patients.
MICROMEDEX Thomson Health Care. USPDI - Drug Information for the Health Care Professional 21 st ed. Volume 1. MICROMEDEX Thomson Health Care, Englewood, CO. 2001. Content Reviewed and Approved by the U.S. Pharmacopeial Convention, Inc., p. 1136
For more Drug Warnings (Complete) data for CYCLOSPORIN A (30 total), please visit the HSDB record page.
Cyclosporine is approved for a variety of conditions. Firstly, it is approved for the prophylaxis of organ rejection in allogeneic kidney, liver, and heart transplants. It is also used to prevent bone marrow transplant rejection. For the above indications, cyclosporine can be used in conjunction with azathioprine and corticosteroids. Finally, cyclosporine can be used in patients who have chronic transplant rejection and have received previous immunosuppressive therapy and to prevent or treat graft-versus-host disease (GVHD). Secondly, cyclosporine is used for the treatment of patients with severe active rheumatoid arthritis (RA) when they no longer respond to methotrexate alone. It can be used for the treatment of adult non-immunocompromised patients with severe, recalcitrant, plaque psoriasis that have failed to respond to at least one systemic therapy or when systemic therapies are not tolerated or contraindicated. The ophthalmic solution of cyclosporine is indicated to increase tear production in patients suffering from keratoconjunctivitis sicca. In addition, cyclosporine is approved for the treatment of steroid dependent and steroid-resistant nephrotic syndrome due to glomerular diseases which may include minimal change nephropathy, focal and segmental glomerulosclerosis or membranous glomerulonephritis. A cyclosporine ophthalmic emulsion is indicated in the treatment of vernal keratoconjunctivitis in adults and children. Off-label, cyclosporine is commonly used for the treatment of various autoimmune and inflammatory conditions such as atopic dermatitis, blistering disorders, ulcerative colitis, juvenile rheumatoid arthritis, uveitis, connective tissue diseases, as well as idiopathic thrombocytopenic purpura.
FDA Label
Treatment of severe keratitis in adult patients with dry eye disease, which has not improved despite treatment with tear substitutes.
Treatment of severe vernal keratoconjunctivitis (VKC) in children from 4 years of age and adolescents.
Treatment of dry eye disease
Treatment of dry eye disease
Treatment of keratoconjunctivitis sicca
Treatment of dry eye disease
Treatment of keratoconjunctivitis sicca, Treatment of vernal keratoconjunctivitis
Treatment of atopic keratoconjunctivitis
Prevention of rejection following lung transplantation
Cyclosporine exerts potent immunosuppressive actions on T cells, thereby prolonging survival following organ and bone marrow transplants. This drug prevents and controls serious immune-mediated reactions including allograft rejection, graft versus host disease, and inflammatory autoimmune disease. Some notable effects of cyclosporine are hypertrichosis, gingival hyperplasia, and hyperlipidemia. There is also some debate about this drug causing nephrotoxicity.
Enzyme Inhibitors
Compounds or agents that combine with an enzyme in such a manner as to prevent the normal substrate-enzyme combination and the catalytic reaction. (See all compounds classified as Enzyme Inhibitors.)
Antirheumatic Agents
Drugs that are used to treat RHEUMATOID ARTHRITIS. (See all compounds classified as Antirheumatic Agents.)
Calcineurin Inhibitors
Compounds that inhibit or block the PHOSPHATASE activity of CALCINEURIN. (See all compounds classified as Calcineurin Inhibitors.)
Dermatologic Agents
Drugs used to treat or prevent skin disorders or for the routine care of skin. (See all compounds classified as Dermatologic Agents.)
Immunosuppressive Agents
Agents that suppress immune function by one of several mechanisms of action. Classical cytotoxic immunosuppressants act by inhibiting DNA synthesis. Others may act through activation of T-CELLS or by inhibiting the activation of HELPER CELLS. While immunosuppression has been brought about in the past primarily to prevent rejection of transplanted organs, new applications involving mediation of the effects of INTERLEUKINS and other CYTOKINES are emerging. (See all compounds classified as Immunosuppressive Agents.)
Antifungal Agents
Substances that destroy fungi by suppressing their ability to grow or reproduce. They differ from FUNGICIDES, INDUSTRIAL because they defend against fungi present in human or animal tissues. (See all compounds classified as Antifungal Agents.)
S01XA18
S01XA18
L04AD01
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
L - Antineoplastic and immunomodulating agents
L04 - Immunosuppressants
L04A - Immunosuppressants
L04AD - Calcineurin inhibitors
L04AD01 - Ciclosporin
S - Sensory organs
S01 - Ophthalmologicals
S01X - Other ophthalmologicals
S01XA - Other ophthalmologicals
S01XA18 - Ciclosporin
Absorption
The absorption of cyclosporine occurs mainly in the intestine. Absorption of cyclosporine is highly variable with a peak bioavailability of 30% sometimes occurring 1-8 hours after administration with a second peak observed in certain patients. The absorption of cyclosporine from the GI tract has been found to be incomplete, likely due to first pass effects. Cmax in both the blood and plasma occurs at approximately 3.5 hours post-dose. The Cmax of a 0.1% cyclosporine ophthalmic emulsion is 0.67 ng/mL after instilling one drop four times daily. A note on erratic absorption During chronic administration, the absorption of Sandimmune Soft Gelatin Capsules and Oral Solution have been observed to be erratic, according to Novartis prescribing information. Those being administered the soft gelatin capsules or oral solution over the long term should be regularly monitored by testing cyclosporine blood concentrations and adjusting the dose accordingly. When compared with the other oral forms of Sandimmune, Neoral capsules and solution have a higher rate of absorption that results in a higher Tmax and a 59% higher Cmax with a 29 % higher bioavailability.
Route of Elimination
After sulfate conjugation, cyclosporine remains in the bile where it is broken down to the original compound and then re-absorbed into the circulation. Cyclosporine excretion is primarily biliary with only 3-6% of the dose (including the parent drug and metabolites) excreted in the urine while 90% of the administered dose is eliminated in the bile. From the excreted proportion, under 1% of the dose is excreted as unchanged cyclosporine.
Volume of Distribution
The distribution of cyclosporine in the blood consists of 33%-47% in plasma, 4%-9% in the lymphocytes, 5%-12% in the granulocytes, and 41%-58% in the erythrocytes. The reported volume of distribution of cyclosporine ranges from 4-8 L/kg. It concentrates mainly in leucocyte-rich tissues as well as tissues that contain high amounts of fat because it is highly lipophilic. Cyclosporine, in the eye drop formulation, crosses the blood-retinal barrier.
Clearance
Cyclosporin shows a linear clearance profile that ranges from 0.38 to 3 Lxh/kg, however, there is substantial inter- patient variability. A 250 mg dose of cyclosporine in the oral soft gelatin capsule of a lipid micro-emulsion formulation shows an approximate clearance of 22.5 L/h.
Following oral admin of cyclosporine, the time to peak blood concns is 1.5-2.0 hr. Admin with food both delays & decreases absorption. High & low fat meals consumed within 30 min of admin decr the AUC by approx 13% & the max concn by 33%. This makes it imperative to individualize dosage regimens for outpatients. Cyclosporine is distributed extensively outside the vascular compartment. After iv dosing, the steady-state volume of distribution has been reported to be as high as 3-5 liters/kg in solid-organ transplant recipients. Only 0.1% of cyclosporine is excreted unchanged in urine. ... Cyclosporine & its metabolites are excreted principally through the bile into the feces, with only approx 6% being excreted in the urine. Cyclosporine also is excreted in human milk.
Hardman, J.G., L.E. Limbird, P.B. Molinoff, R.W. Ruddon, A.G. Goodman (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 9th ed. New York, NY: McGraw-Hill, 1996., p. 1468
... Absorption of cyclosporine is incomplete following oral admin. The extent of absorption depends upon several variables, including the individual patient & formulation used. The elimination of cyclosporine form the blood is generally biphasic, with a terminal half-life of 5-18 hr. After iv infusion, clearance is approx 5-7 ml/min/kg in adult recipients of renal transplants, but results differ by age & patient populations. For example, clearance is slower in cardiac transplant patients & more rapid in children. The relationship between admin dose & the area under the plasma concn-vs-time curve is linear within the therapeutic range, but the intersubject variability is so large that individual monitoring is required.
Hardman, J.G., L.E. Limbird, P.B. Molinoff, R.W. Ruddon, A.G. Goodman (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 9th ed. New York, NY: McGraw-Hill, 1996., p. 1468
Clinicians can administer cyclosporine by continuous iv infusion during the first few days after transplantation, then orally by twice-daily doses, to achieve plasma cyclosporine concns (measured by HPLC) of 75-150 ng/ml (equivalent to whole blood cyclosporine concns of 300-600 ng/ml measured by radioimmunoassay). It appears safe to maintain a trough plasma cyclosporine concn of about 75-150 ng/ml; however, this does not necessarily guarantee safety from nephrotoxicity. Because of preferential distribution of cyclosporine & its metabolites into red blood cells, blood levels are generally higher than plasma levels. When blood cyclosporine levels are 300-600 ng/ml by radioimmunoassay, cerebrospinal fluid levels range from 10-50 ng/ml. The apparent volume of distribution in children under 10 yr of age is about 35 l/kg, & in adults, 4.7 l/kg.
Ellenhorn, M.J., S. Schonwald, G. Ordog, J. Wasserberger. Ellenhorn's Medical Toxicology: Diagnosis and Treatment of Human Poisoning. 2nd ed. Baltimore, MD: Williams and Wilkins, 1997., p. 780
The elimination half-life of an oral cyclosporine dose of 350 mg is 8.9 hr; after a 1400 mg dose, the half-life is 11.9 hr. Elimination occurs predominantly by metab in the liver to form 18-25 metabolites. Metabolites of cyclosporine possess little immunosuppressive activity. Cyclosporine is extensively metabolized in the liver by cytochrome P450IIIA oxidase; however, neurotoxicity & possibly nephrotoxoicity usually correlate with raised blood levels of cyclosporine metabolites. Only 0.1% of a dose ix s excreted unchanged.
Ellenhorn, M.J., S. Schonwald, G. Ordog, J. Wasserberger. Ellenhorn's Medical Toxicology: Diagnosis and Treatment of Human Poisoning. 2nd ed. Baltimore, MD: Williams and Wilkins, 1997., p. 780
For more Absorption, Distribution and Excretion (Complete) data for CYCLOSPORIN A (7 total), please visit the HSDB record page.
Cyclosporine is metabolized in the intestine and the liver by CYP450 enzymes, predominantly CYP3A4 with contributions from CYP3A5. The involvement of CYP3A7 is not clearly established. Cyclosporine undergoes several metabolic pathways and about 25 different metabolites have been identified. One of its main active metabolites, AM1, demonstrates only 10-20% activity when compared to the parent drug, according to some studies. The 3 primary metabolites are M1, M9, and M4N, which are produced from oxidation at the 1-beta, 9-gamma, and 4-N-demethylated positions, respectively.
Cyclosporine is extensively metabolized in the liver by the cytochrome-P450 3A (CYP3A) enzyme system & to a lesser degree by the GI tract & kidneys. At least 25 metabolites have been identified in human bile, feces, blood, & urine. Although the cyclic peptide structure of cyclosporine is relatively resistant to metab, the side chains are extensively metabolized. All of the metabolites have both reduced biological activity & toxicity compared to the parent drug.
Hardman, J.G., L.E. Limbird, P.B. Molinoff, R.W. Ruddon, A.G. Goodman (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 9th ed. New York, NY: McGraw-Hill, 1996., p. 1468
The half-life of cyclosporine is biphasic and very variable on different conditions but it is reported in general to last 19 hours. Prescribing information also states a terminal half-life of approximately 19 hours, but with a range between 10 to 27 hours.
Cyclosporine is a calcineurin inhibitor that inhibits T cell activation. Its binding to the receptor cyclophilin-1 inside cells produces a complex known as cyclosporine-cyclophilin. This complex subsequently inhibits calcineurin, which in turn stops the dephosphorylation as well as the activation of the nuclear factor of activated T cells (NF-AT) that normally cause inflammatory reactions. NF-AT is a transcription factor that promotes the production of cytokines such as IL-2, IL-4, interferon-gamma and TNF-alpha, all of which are involved in the inflammatory process. Specifically, the inhibition of IL-2, which is necessary for T cell activation or proliferation, is believed to be responsible for cyclosporine's immunosuppressive actions. In addition to the above, the inhibition of NF-AT leads to lower levels of other factors associated with T helper cell function and thymocyte development.
Cyclosporine suppresses some humoral immunity but is more effective against T cell-dependent immune mechanisms such as those underlying transplant rejection & some forms of autoimmunity. It preferentially inhibits antigen-triggered signal transduction in T lymphocytes, blunting expression of many lymphokines, including /(interleukin-2)/ IL-2, as well as expression of antiapoptotic proteins. Cyclosporine forms a complex with cyclophilin, a cytoplasmic receptor protein present in target cells. This complex binds to calcineurin, inhibiting Ca2+ stimulated dephosphorylation of the cytosolic component of NFAT. When the cytoplasmic component of NFAT is dephosphorylated, it translocates to the nucleus, where it complexes with nuclear components required for complete T-cell activation, including transactivation of IL-2 & other lymphokine genes. Calcineurin enzymatic activity is inhibited following physical interaction with the cyclosporine/cyclophilin complex. This results in the blockade of NFAT dephosphorylation; thus, the cytoplasmic component of NFAT does not enter the nucleus, gene transcription is not activated, & the T lymphocyte fails to respond to specific antigenic stimulation Cyclosporine also increases expression of transforming growth fact /beta/ (TGF-B), a potent inhibitor of IL-2-stimulated T-cell proliferation & generation of cytotoxic T lymphocytes (CTL).
Hardman, J.G., L.E. Limbird, P.B. Molinoff, R.W. Ruddon, A.G. Goodman (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 9th ed. New York, NY: McGraw-Hill, 1996., p. 1466
The exact mechanism of action is unknown but seems to be related to the inhibition of production and release of interleukin-2, which is a proliferative factor necessary for the induction of cytotoxic T lymphocytes in response to alloantigenic challenge, and which plays a major role in both cellular and humoral immune responses. Cyclosporine does not affect the nonspecific defense system of the most and does not cause significant myelosuppression.
MICROMEDEX Thomson Health Care. USPDI - Drug Information for the Health Care Professional 21 st ed. Volume 1. MICROMEDEX Thomson Health Care, Englewood, CO. 2001. Content Reviewed and Approved by the U.S. Pharmacopeial Convention, Inc., p. 1136
The major pharmacodynamic action of cyclosporin within T cells is calcineurin inhibition. The complex cyclophilin-cyclosporin competitively binds to the Ca(2+)- & calmodulin-dependent phosphatase calcineurin which then inhibits downstream dephosphorylation & activation of NFAT(transcription factor). The greatest calcineurin inhibition is seen 1-2 hr after admin of Neoral in parallel to the highest blood concn.
PMID:11530680 Nishi Y; Nippon Yakurigaku Zasshi 118 (2): 107-115 (2001)
Treatment of patients after organ transplantation with the immunosuppressive drug cyclosporin A (CsA) is often accompanied by impaired glucose tolerance, thus promoting the development of diabetes mellitus. ... /The authors/ show that 2-5 microM CsA diminishes glucose-induced insulin secretion of isolated mouse pancreatic islets in vitro by inhibiting glucose-stimulated oscillations of the cytoplasmic free-Ca(2+) concn [Ca(2+)](c). This effect is not due to an inhibition of calcineurin, which mediates the immunosuppressive effect of CsA, because other calcineurin inhibitors, deltamethrin & tacrolimus, did not affect the oscillations in [Ca(2+)](c) of the B-cells. The CsA-induced decr in [Ca(2+)](c) to basal values was not caused by a direct inhibition of L-type Ca(2+) channels. CsA is known to be a potent inhibitor of the mitochondrial permeability transition pore (PTP), which ... /the authors/ recently suggested to be involved in the regulation of oscillations. Consequently, CsA also inhibited the oscillations of the cell membrane potential, & it is shown that these effects could not be ascribed to cellular ATP depletion. However, the mitochondrial membrane potential Delta Psi was affected by CsA by inhibiting the oscillations in Delta Psi. ... The observed reduction in [Ca(2+)](c) could be counteracted by the K(+)(ATP) channel blocker tolbutamide, indicating that the stimulus-secretion coupling was interrupted before the closure of K(+)(ATP) channels. It is concluded that CsA alters B-cell function by inhibiting the mitochondrial PTP. This terminates the oscillatory activity that is indispensable for adequate insulin secretion. Thus, CsA acts on different targets to induce the immunosuppressive & the diabetogenic effect.
PMID:11562451 Dufer M et al; Mol Pharmacol 60 (4): 873-879 (2001)
CsA increases CTGF, collagen I, & collagen III mRNA expressions in the heart. The induction of CTGF gene is mediated, at least in part, by angiotensin II.
PMID:11349731 Finckenberg P et al; Transplantation 71 (7): 951-958 (2001)
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
38
PharmaCompass offers a list of Cyclosporine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Cyclosporine manufacturer or Cyclosporine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Cyclosporine manufacturer or Cyclosporine supplier.
A Restasis manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Restasis, including repackagers and relabelers. The FDA regulates Restasis manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Restasis API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Restasis manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Restasis supplier is an individual or a company that provides Restasis active pharmaceutical ingredient (API) or Restasis finished formulations upon request. The Restasis suppliers may include Restasis API manufacturers, exporters, distributors and traders.
click here to find a list of Restasis suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Restasis DMF (Drug Master File) is a document detailing the whole manufacturing process of Restasis active pharmaceutical ingredient (API) in detail. Different forms of Restasis DMFs exist exist since differing nations have different regulations, such as Restasis USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Restasis DMF submitted to regulatory agencies in the US is known as a USDMF. Restasis USDMF includes data on Restasis's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Restasis USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Restasis suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Restasis Drug Master File in Japan (Restasis JDMF) empowers Restasis API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Restasis JDMF during the approval evaluation for pharmaceutical products. At the time of Restasis JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Restasis suppliers with JDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Restasis Drug Master File in Korea (Restasis KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Restasis. The MFDS reviews the Restasis KDMF as part of the drug registration process and uses the information provided in the Restasis KDMF to evaluate the safety and efficacy of the drug.
After submitting a Restasis KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Restasis API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Restasis suppliers with KDMF on PharmaCompass.
A Restasis CEP of the European Pharmacopoeia monograph is often referred to as a Restasis Certificate of Suitability (COS). The purpose of a Restasis CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Restasis EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Restasis to their clients by showing that a Restasis CEP has been issued for it. The manufacturer submits a Restasis CEP (COS) as part of the market authorization procedure, and it takes on the role of a Restasis CEP holder for the record. Additionally, the data presented in the Restasis CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Restasis DMF.
A Restasis CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Restasis CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Restasis suppliers with CEP (COS) on PharmaCompass.
A Restasis written confirmation (Restasis WC) is an official document issued by a regulatory agency to a Restasis manufacturer, verifying that the manufacturing facility of a Restasis active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Restasis APIs or Restasis finished pharmaceutical products to another nation, regulatory agencies frequently require a Restasis WC (written confirmation) as part of the regulatory process.
click here to find a list of Restasis suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Restasis as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Restasis API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Restasis as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Restasis and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Restasis NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Restasis suppliers with NDC on PharmaCompass.
Restasis Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Restasis GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Restasis GMP manufacturer or Restasis GMP API supplier for your needs.
A Restasis CoA (Certificate of Analysis) is a formal document that attests to Restasis's compliance with Restasis specifications and serves as a tool for batch-level quality control.
Restasis CoA mostly includes findings from lab analyses of a specific batch. For each Restasis CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Restasis may be tested according to a variety of international standards, such as European Pharmacopoeia (Restasis EP), Restasis JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Restasis USP).

