
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
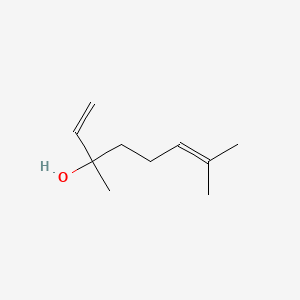

1. 3,7-dimethyl-1,6-octadien-3-ol
2. 7-methyl-3-methyleneocta-4,6-dien-2-ol
3. Allo-ocimenol
4. Linalool, (+-)-isomer
5. Linalool, (r)-isomer
6. Linalool, (s)-isomer
7. Linalool, Titanium (4+) Salt
8. Muguol
1. 78-70-6
2. 3,7-dimethylocta-1,6-dien-3-ol
3. Linalol
4. Linalyl Alcohol
5. 3,7-dimethyl-1,6-octadien-3-ol
6. Allo-ocimenol
7. Beta-linalool
8. 1,6-octadien-3-ol, 3,7-dimethyl-
9. (+-)-linalool
10. P-linalool
11. Linanool
12. Linolool
13. Phantol
14. 2,6-dimethyl-2,7-octadien-6-ol
15. 2,6-dimethylocta-2,7-dien-6-ol
16. .beta.-linalool
17. (+/-)-linalool
18. 2,6-dimethyl-2,7-octadiene-6-ol
19. Fema No. 2635
20. Nsc 3789
21. (rs)-linalool
22. (1)-3,7-dimethyl-1,6-octadien-3-ol
23. Chebi:17580
24. 3,7-dimethyl-octa-1,6-dien-3-ol
25. D81qy6i88e
26. Nsc3789
27. Nsc-3789
28. Dsstox_cid_5502
29. Coriandrol
30. Dsstox_rid_77812
31. Dsstox_gsid_25502
32. Licareol
33. Linalool (natural)
34. Caswell No. 526a
35. (s)-linalol
36. Dl-linalool
37. Fema Number 2635
38. Cas-78-70-6
39. Ccris 6557
40. Hsdb 645
41. Linalool (+)
42. Einecs 201-134-4
43. Einecs 245-083-6
44. Epa Pesticide Chemical Code 128838
45. Brn 1721488
46. Unii-d81qy6i88e
47. Ai3-00942
48. Linalool B
49. Howood Oil
50. Natural Linalool
51. Linalool, .beta.
52. Linalool,(s)
53. ( )-linalool
54. Linalool Ex Ho Oil
55. Mfcd00008906
56. Diabexall
57. Linalool, 97%
58. 2,7-octadien-6-ol, 2,6-dimethyl-
59. Linalool Ex Orange Oil
60. 3,6-octadien-3-ol
61. 2,7-octadiene-6-ol
62. Linalool [fhfi]
63. Linalool [hsdb]
64. Linalool [inci]
65. Linalool [fcc]
66. 2,7-dien-6-ol
67. 3,6-dien-3-ol
68. Dl-3,7-dimethyl-3-hydroxy-1,6-octadiene
69. Linalool [mi]
70. Linalool, Dl-
71. Linolool (d)
72. (.+/-.)-linalool
73. Linalool [usp-rs]
74. Linalool [who-dd]
75. Ec 201-134-4
76. Linalool Oil [ndi]
77. L 260-2
78. Schembl20316
79. Linalool Ex Bois De Rose Oil
80. Linalool, Analytical Standard
81. 0-01-00-00462 (beilstein Handbook Reference)
82. 22564-99-4
83. Mls002152908
84. Chembl25306
85. Pentaerythritoltetramethacrylate
86. Gtpl2469
87. Ndi 595 [fdms]
88. Dtxsid7025502
89. Linalool Ex Bois De Rose
90. Linalool, (+/-)-
91. Fema 2635
92. ((c)i)-linalool-d3(vinyl-d3)
93. Hms2268e18
94. Hms3886g07
95. Linalool, >=97%, Fcc, Fg
96. 1, 3,7-dimethyl-, (-)-
97. Hy-n0368
98. Wln: 1u1xq1&3uy1&1
99. Tox21_201658
100. Tox21_303037
101. Ac-551
102. Bbl027734
103. Bdbm50459894
104. Mfcd09025547
105. S4957
106. Stl373777
107. 3,7-dimethyl-1, 6-octadien-3-ol
108. Akos015901617
109. ( Inverted Exclamation Marka)-linalool
110. Ccg-266253
111. Linalool 1000 Microg/ml In Isopropanol
112. Ncgc00091688-01
113. Ncgc00091688-02
114. Ncgc00091688-03
115. Ncgc00091688-04
116. Ncgc00257060-01
117. Ncgc00259207-01
118. (?)-3,7-dimethyl-1,6-octadien-3-ol
119. As-56047
120. Smr000112394
121. Sy264412
122. Wln: 1y1&u3xq1&1u1 -,-
123. Db-062552
124. Cs-0008916
125. Ft-0614785
126. Ft-0772303
127. L0048
128. C03985
129. F17676
130. Linalool, Primary Pharmaceutical Reference Standard
131. Q410932
132. Cu-01000013132-2
133. Q-201306
134. Linalool, Certified Reference Material, Tracecert(r)
| Molecular Weight | 154.25 g/mol |
|---|---|
| Molecular Formula | C10H18O |
| XLogP3 | 2.7 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 4 |
| Exact Mass | 154.135765193 g/mol |
| Monoisotopic Mass | 154.135765193 g/mol |
| Topological Polar Surface Area | 20.2 Ų |
| Heavy Atom Count | 11 |
| Formal Charge | 0 |
| Complexity | 154 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Insecticides
Pesticides designed to control insects that are harmful to man. The insects may be directly harmful, as those acting as disease vectors, or indirectly harmful, as destroyers of crops, food products, or textile fabrics. (See all compounds classified as Insecticides.)
Linalool may permeate porcine (and by extension also human) buccal mucosa in function of its concentration (14.46% w/w) and of formulation /as in the essential oil of Salvia desoleana Atzei & Picci/
Organization for Economic Cooperation and Development; Screening Information Data Set for LINALOOL (78-70-6) p.95 (March 2002). Available from, as of July 15, 2008: https://www.chem.unep.ch/irptc/sids/OECDSIDS/sidspub.html
Based on experiments with rats using (14)C-labelled substance, linalool is rapidly absorbed from the intestinal tract following oral uptake ... judging from the delay in fecal excretion, intestinal absorption is complete. Subsequent to absorption, linalool is metabolized rapidly, with urinary excretion of (14)C activity starting without delay. Several hours after gavage, substantial amounts of radioactivity were detected in the expired air as (14)CO2, evidencing complete intermediary metabolism. Fecal excretion of radioactivity was delayed and found mostly between 36 and 48 hours after dosing, suggesting entero-hepato-biliary re-circulation; this re-circulation was confirmed in a second experiment involving cross-linking a treated and an untreated rat with a biliary-to-intestinal cannula and subsequent radio-analysis. Overall, approximately 60% of the total excreted dose was found in urine over 72 hours after administration; approximately 23% of activity was detected in exhaled air and approximately 15% was found in the feces; there is no indication of tissue accumulation of linalool whatsoever. The study suggests that large doses of oral linalool will be metabolized in the rat by conjugation and excretion in urine and bile, while a substantial proportion will enter intermediary metabolisms up to the formation of carbon dioxide and pulmonary excretion. Entero-hepato-biliary re-circulation may have the effect of enhancing the metabolic load on the liver over a certain period.
Organization for Economic Cooperation and Development; Screening Information Data Set for LINALOOL (78-70-6) p.12 (March 2002). Available from, as of July 15, 2008: https://www.chem.unep.ch/irptc/sids/OECDSIDS/sidspub.html
After a 1 hour inhalation of 27 mg linalool ... /mouse/ plasma levels of linalool 30, 60 and 90 min after exposure: about 1, 2.7 and 2.9 ng/mL.
European Chemicals Bureau; IUCLID Dataset, Linalool (78-70-6) p.46 (2000 CD-ROM edition).
Linalool applied to mouse skin was not absorbed within two hours.
Organization for Economic Cooperation and Development; Screening Information Data Set for LINALOOL (78-70-6) p.100 (March 2002). Available from, as of July 15, 2008: https://www.chem.unep.ch/irptc/sids/OECDSIDS/sidspub.html
... Metabolites isolated from urine of rats after oral administration of linalool (VII) were 8-hydroxy-linalool (VIII) and 8-carboxy-linalool (IX). After three days of feeding rats with either geraniol or linalool, liver-microsomal cytochrome P-450 was increased. Both NADH- and NADPH-cytochrome c reductase activities were not significantly changed during the six days of treatment. Oral administration of these two terpenoids did not affect any of the lung-microsomal parameters measured.
PMID:6475100 Chadha A, Madyastha KM; Xenobiotica 14 (5): 365-74 (1984)
Based on experiments with rats using (14)C-labelled substance ... subsequent to absorption, linalool is metabolized rapidly, with urinary excretion of (14)C activity starting without delay. Several hours after gavage, substantial amounts of radioactivity were detected in the expired air as (14)CO2, evidencing complete intermediary metabolism. Fecal excretion of radioactivity was delayed and found mostly between 36 and 48 hours after dosing, suggesting entero-hepato-biliary re-circulation ... Entero-hepato-biliary re-circulation may have the effect of enhancing the metabolic load on the liver over a certain period.
Organization for Economic Cooperation and Development; Screening Information Data Set for LINALOOL (78-70-6) p.12 (March 2002). Available from, as of July 15, 2008: https://www.chem.unep.ch/irptc/sids/OECDSIDS/sidspub.html
For the induction study 600 mg linalool/kg bw was administered /to male IISc strain rats/ once daily for 6 days by gastric tube as a suspension in 1% methyl cellulose solution. Control rats were only given the vehicle. For the identification of metabolites, 800 mg linalool/kg bw was administered once daily for 20 days ... . 8-Hydroxy-linalool and 8-carboxy-linalool were identified in the urine, showing selective oxidation of the C8-methyl in linalool. The 8-hydroxylase present in both lung and liver microsomes was shown to be mediated by a cytochrome P-450 (CYP450) system. After 3 days of dosing, liver and lung microsomal CYP450 was increased; on the other hand, both NADH- and NADPH-cytochrome c reductase activities were not significantly changed during the 6 days of treatment. /Purity > 99.5%/
Organization for Economic Cooperation and Development; Screening Information Data Set for LINALOOL (78-70-6) p.97 (March 2002). Available from, as of July 15, 2008: https://www.chem.unep.ch/irptc/sids/OECDSIDS/sidspub.html
... Hydrolysis occurs more rapidly at the low pH of gastric fluids. The reaction products are linalool and acetic acid (ester hydrolysis). This is supported by the findings of the hydrolysis study ... at pH 4, 7 and 9. Therefore it is expected that linalool is the substance that will enter the systemic circulation after oral uptake of linalyl acetate. Linalool is probably converted to geraniol and its metabolites, 1,5-dimethyl-hexadiene-1,6-dicarboxylic acid and 7-carboxy-5-methylocto-6-enoic acid ... /linalool acetate/
Organization for Economic Cooperation and Development; Screening Information Data Set for LINALYL ACETATE (115-95-7) p.10 (March 2002). Available from, as of July 14, 2008: https://www.chem.unep.ch/irptc/sids/OECDSIDS/sidspub.html
For more Metabolism/Metabolites (Complete) data for LINALOOL (7 total), please visit the HSDB record page.
... The specific toxic effect of linalool on animals is therefore likely to be caused by its neurotoxic respectively neuropharmacological mode of action. In turn, this may explain the use of linalool-containing natural products (aromatic herbs and spices or their essential oils respectively extracts) in traditional medicinal systems, specifically for their sleep-inducing and anticonvulsant purposes. Moreover, it also accounts for the widespread traditional use of herbs containing linalool for stored-food pest control for the use of linalool-containing extracts as a pet flea insecticide.
Organization for Economic Cooperation and Development; Screening Information Data Set for LINALOOL (78-70-6) p.20 (March 2002). Available from, as of July 15, 2008: https://www.chem.unep.ch/irptc/sids/OECDSIDS/sidspub.html
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
84
PharmaCompass offers a list of Linalool API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Linalool manufacturer or Linalool supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Linalool manufacturer or Linalool supplier.
A Linalool manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Linalool, including repackagers and relabelers. The FDA regulates Linalool manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Linalool API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Linalool supplier is an individual or a company that provides Linalool active pharmaceutical ingredient (API) or Linalool finished formulations upon request. The Linalool suppliers may include Linalool API manufacturers, exporters, distributors and traders.
Linalool Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Linalool GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Linalool GMP manufacturer or Linalool GMP API supplier for your needs.
A Linalool CoA (Certificate of Analysis) is a formal document that attests to Linalool's compliance with Linalool specifications and serves as a tool for batch-level quality control.
Linalool CoA mostly includes findings from lab analyses of a specific batch. For each Linalool CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Linalool may be tested according to a variety of international standards, such as European Pharmacopoeia (Linalool EP), Linalool JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Linalool USP).

