Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
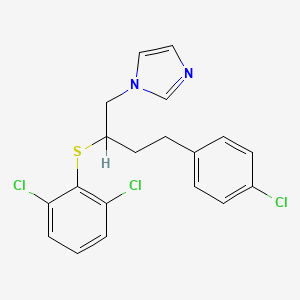

1. 1-(4-(4-chlorophenyl)-2-(2,6-dichlorophenylthio)-n-butyl)-1h-imidazole
2. Butoconazole Nitrate
3. Femstal
4. Femstat
5. Gynazole-1
6. Gynomyk
7. Mycelex-3
8. Rs 35887
1. 64872-76-0
2. Butaconazole
3. Butoconazol
4. Butoconazolum
5. Butoconazol [inn-spanish]
6. Butoconazolum [inn-latin]
7. Gynofort
8. Butoconazole (inn)
9. Gynazole-1
10. Nsc-758658
11. Chebi:3240
12. 0q771797ph
13. Butoconazole [inn]
14. Butoconazole [inn:ban]
15. Mls002153798
16. 1-(4-(4-chlorophenyl)-2-((2,6-dichlorophenyl)thio)butyl)-1h-imidazole
17. 1-[4-(4-chloro-phenyl)-2-(2,6-dichloro-phenylsulfanyl)-butyl]-1h-imidazole
18. 1-[4-(4-chlorophenyl)-2-[(2,6-dichlorophenyl)thio]butyl]-1h-imidazole
19. 1h-imidazole, 1-[4-(4-chlorophenyl)-2-[(2,6-dichlorophenyl)thio]butyl]-
20. Gynofort (tn)
21. Smr001233176
22. Rs 35887-10-3
23. Unii-0q771797ph
24. 67085-13-6
25. Prestwick0_000408
26. Prestwick1_000408
27. Prestwick2_000408
28. Prestwick3_000408
29. Butoconazole [mi]
30. 1-[4-(4-chlorophenyl)-2-(2,6-dichlorophenyl)sulfanyl-butyl]imidazole
31. 1-[4-(4-chlorophenyl)-2-(2,6-dichlorophenyl)sulfanylbutyl]imidazole
32. 1-{4-(4-chlorophenyl)-2-[(2,6-dichlorophenyl)sulfanyl]butyl}-1h-imidazole
33. Butoconazole [vandf]
34. Chembl1295
35. Schembl44237
36. Bspbio_000336
37. Cid_47471
38. Mls006010743
39. Butoconazole [who-dd]
40. Spbio_002275
41. Bpbio1_000370
42. 1-[(2s)-4-(4-chlorophenyl)-2-(2,6-dichlorophenyl)sulfanyl-butyl]imidazole
43. Dtxsid2048537
44. Bdbm79206
45. Hy-b0293a
46. Hms3264e14
47. Pharmakon1600-01504140
48. Albb-028967
49. Amy32540
50. Butoconazole, Butoconazole Nitrate
51. (+-)-1-((4-(p-chlorophenyl)-2-((2,6-dichlorophenyl)thio)imidazole
52. Nsc758658
53. S5359
54. Akos015900067
55. Ccg-213264
56. Db00639
57. Nsc 758658
58. (+-)-1-[4-(4-chlorophenyl)-2-[(2,6-dichlorophenyl)thio]butyl]-1h-imidazole
59. 1h-imidazole, 1-(4-(4-chlorophenyl)-2-((2,6-dichlorophenyl)thio)butyl)-, (+-)-
60. Ncgc00179596-01
61. Ncgc00179596-04
62. Ac-15961
63. Smr001550002
64. Db-054743
65. Ab00513831
66. Cs-0013455
67. Ft-0630751
68. C08065
69. D07598
70. Ab00513831-07
71. Ab00513831_08
72. Ab00513831_09
73. Butoconazole, Vetranal(tm), Analytical Standard
74. 872b760
75. A834909
76. Q2928948
77. Brd-a16665823-001-01-2
78. Brd-a16665823-008-03-3
79. 1-(4-(4-chlorophenyl)-2-(2,6-dichlorophenylthio)butyl)-1h-imidazole
80. 1-[2-[(2,6-dichlorophenyl)thio]-4-(4-chlorophenyl)butyl]-1h-imidazole
81. 1-[4-(4-chlorophenyl)-2-(2,6-dichlorophenyl)sulfanylbutyl]imid-azole
82. 1-[4-(4-chlorophenyl)-2-(2,6-dichlorophenylthio)-n-butyl]-imidazole
83. (+/-)-1-(4-(p-chlorophenyl)-2-((2,6-dichlorophenyl)thio)butyl)imidazole
84. (.+/-.)-1-[4-(p-chlorophenyl)-2-[(2,6-dichlorophenyl)thio]butyl]imidazole
85. 1-(4-(4-chlorophenyl)-2-[(2,6-dichlorophenyl)sulfanyl]butyl)-1h-imidazole #
86. 1h-imidazole, 1-(4-(4-chlorophenyl)-2-((2,6-dichlorophenyl)thio)butyl)-, (+/-)-
87. 1h-imidazole, 1-[4-(4-chlorophenyl)-2-[(2,6-dichlorophenyl)thio]butyl]-, (1)-
88. 1h-imidazole, 1-[4-(4-chlorophenyl)-2-[(2,6-dichlorophenyl)thio]butyl]-, Nitrate (1:1)
| Molecular Weight | 411.8 g/mol |
|---|---|
| Molecular Formula | C19H17Cl3N2S |
| XLogP3 | 6.4 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 7 |
| Exact Mass | 410.017803 g/mol |
| Monoisotopic Mass | 410.017803 g/mol |
| Topological Polar Surface Area | 43.1 Ų |
| Heavy Atom Count | 25 |
| Formal Charge | 0 |
| Complexity | 383 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
For the local treatment of vulvovaginal candidiasis (infections caused by Candida)
FDA Label
Butoconazole is an imidazole derivative that has fungicidal activity in vitro against Candida spp. and has been demonstrated to be clinically effective against vaginal infections due to Candida albicans. Candida albicans has been identified as the predominant species responsible for vulvovaginal candidasis.
Antifungal Agents
Substances that destroy fungi by suppressing their ability to grow or reproduce. They differ from FUNGICIDES, INDUSTRIAL because they defend against fungi present in human or animal tissues. (See all compounds classified as Antifungal Agents.)
G01AF15
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
G - Genito urinary system and sex hormones
G01 - Gynecological antiinfectives and antiseptics
G01A - Antiinfectives and antiseptics, excl. combinations with corticosteroids
G01AF - Imidazole derivatives
G01AF15 - Butoconazole
Absorption
Following vaginal administration of butoconazole nitrate vaginal cream, 2% to 3 women, 1.7% (range 1.3-2.2%) of the dose was absorbed on average.
The exact mechanism of the antifungal action of butoconazole is unknown, however, it is presumed to function as other imidazole derivatives via inhibition of steroid synthesis. Imidazoles generally inhibit the conversion of lanosterol to ergosterol via the inhibition of the enzyme cytochrome P450 14α-demethylase, resulting in a change in fungal cell membrane lipid composition. This structural change alters cell permeability and, ultimately, results in the osmotic disruption or growth inhibition of the fungal cell.
ABOUT THIS PAGE
37
PharmaCompass offers a list of Butoconazole API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Butoconazole manufacturer or Butoconazole supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Butoconazole manufacturer or Butoconazole supplier.
A Butoconazole manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Butoconazole, including repackagers and relabelers. The FDA regulates Butoconazole manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Butoconazole API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Butoconazole supplier is an individual or a company that provides Butoconazole active pharmaceutical ingredient (API) or Butoconazole finished formulations upon request. The Butoconazole suppliers may include Butoconazole API manufacturers, exporters, distributors and traders.
Butoconazole Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Butoconazole GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Butoconazole GMP manufacturer or Butoconazole GMP API supplier for your needs.
A Butoconazole CoA (Certificate of Analysis) is a formal document that attests to Butoconazole's compliance with Butoconazole specifications and serves as a tool for batch-level quality control.
Butoconazole CoA mostly includes findings from lab analyses of a specific batch. For each Butoconazole CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Butoconazole may be tested according to a variety of international standards, such as European Pharmacopoeia (Butoconazole EP), Butoconazole JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Butoconazole USP).

