
Synopsis
Synopsis
0
CEP/COS
0
KDMF
0
VMF
0
Australia

DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
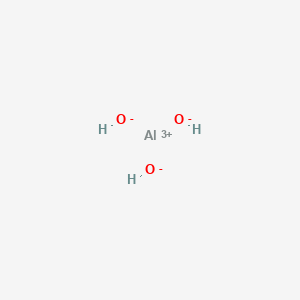

1. Dried Aluminum Hydroxide Gel
2. Aluminum;trihydroxide
3. Aluminium Hydroxide Gel, Dried
4. Dried Aluminium Hydroxide
5. Aluminum Hydroxide, Dried
6. Aluminium Hydroxide, Dried
7. Aluminum Hydroxide Gel, Dried
8. Nsc-664400
9. Aluminum Hydrate
10. Aluminum Hyroxide
11. Aluminium Trihydroxide
12. Hydroxyde D' Aluminium
13. Algeldrate Anhydrous
14. Aluminum (as Hydroxide)
15. Aluminum Hydroxide [ii]
16. Aluminum Hydroxide [mi]
17. Chembl1200706
18. Dtxsid2036405
19. Niosh/bd0708000
20. Aluminum Hydroxide [inci]
21. Di-mu-hydroxytetrahydroxydialuminum
22. Aluminum Hydroxide [vandf]
23. Aluminium Hydroxide[who-ip]
24. Aluminum Hydroxide [mart.]
25. Aluminum Hydroxide Gel,dried
26. Aluminium Hydroxide Dried Gel
27. Af-260
28. Aluminium Hydroxide [who-dd]
29. Akos015904617
30. Aluminum, Di-mu-hydroxytetrahydroxydi-
31. Db06723
32. Aluminum Hydroxide [orange Book]
33. Aluminii Hydroxidum[who-ip Latin]
34. Aluminum (as Hydroxide) [vandf]
35. Aluminum Hydroxide [usp Impurity]
36. Aluminum Hydroxide, Dried [hsdb]
37. As04 Component Aluminum Hydroxide
38. Aluminum Hydroxide Gel, Dried [ii]
39. Foamcoat Component Aluminum Hydroxide
40. Foamicon Component Aluminum Hydroxide
41. Gaviscon Component Aluminum Hydroxide
42. Aluminum Hydroxide Gel,dried [vandf]
43. Bd07080000
44. Dried Aluminum Hydroxide Gel [usp-rs]
45. Aluminum Hydroxide Component Of Foamcoat
46. Aluminum Hydroxide Component Of Foamicon
47. Aluminum Hydroxide Component Of Gaviscon
48. Aluminium Hydroxide Gel, Dried [who-dd]
49. Aluminum Hydroxide, Dried [usp Impurity]
50. Q407125
51. J-014205
52. 8012-63-3
| Molecular Weight | 78.004 g/mol |
|---|---|
| Molecular Formula | AlH3O3 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 0 |
| Exact Mass | 77.9897574 g/mol |
| Monoisotopic Mass | 77.9897574 g/mol |
| Topological Polar Surface Area | 3 Ų |
| Heavy Atom Count | 4 |
| Formal Charge | 0 |
| Complexity | 0 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 4 |
For relief of heartburn and acid indigestion.
Gastric-peptic disease occurs as a result of an imbalance between protective factors, such as mucus, bicarbonate, and prostaglandin secretion, and aggressive factors, such as hydrochloric acid, pepsin, and Helicobacter pylori (H. pylori). Antacids work by restoring acid-base balance, attenuating the pepsin activity and increasing bicarbonate and prostaglandin secretion.
Absorption
Approximately 17-30% of the aluminum chloride formed is absorbed.
Route of Elimination
Absorbed aluminum chloride is rapidly eliminated by the kidneys in patients with normal renal function.
Not metabolized.
Aluminum hydroxide is a basic inorganic salt that acts by neutralizing hydrochloric acid in gastric secretions. Aluminum hydroxide is slowly solubilized in the stomach and reacts with hydrochloric acid to form aluminum chloride and water. It also inhibits the action of pepsin by increasing the pH and via adsorption. Cytoprotective effects may occur through increases in bicarbonate ion (HCO3-) and prostaglandins.
 Rochem, your partner in developing, sourcing, and supplying pharmaceutical & animal health ingredients of Chinese origin.
Rochem, your partner in developing, sourcing, and supplying pharmaceutical & animal health ingredients of Chinese origin.

NDC Package Code : 12784-442
Start Marketing Date : 2019-12-09
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (61.5kg/100kg 5kg/100kg)
Marketing Category : DRUG FOR FURTHER PROCESSING
 Octavius Pharma has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.
Octavius Pharma has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.
 Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others
USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39719
Submission : 2024-04-22
Status : Active
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 38885
Submission : 2023-11-18
Status : Active
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 38895
Submission : 2023-11-18
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 8222
Submission : 1989-09-20
Status : Inactive
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
AD04 is a new class of therapeutic for mild Alzheimer’s disease that exhibits durable multifaceted effects, including both immediate symptomatic relief and long-term disease-modification.
Lead Product(s): Aluminium Hydroxide
Therapeutic Area: Neurology Brand Name: AD04
Study Phase: Phase IIProduct Type: Other Small Molecule
Sponsor: Inapplicable
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable July 30, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Aluminium Hydroxide
Therapeutic Area : Neurology
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : Inapplicable
Deal Size : Inapplicable
Deal Type : Inapplicable
ADvantage Therapeutics Announces Breakthrough Findings for AD04TM in Alzheimer's Disease
Details : AD04 is a new class of therapeutic for mild Alzheimer’s disease that exhibits durable multifaceted effects, including both immediate symptomatic relief and long-term disease-modification.
Product Name : AD04
Product Type : Other Small Molecule
Upfront Cash : Inapplicable
July 30, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
ANK-101 is an anchored drug complex composed of interleukin-12 (IL-12) linked to aluminum hydroxide. It is being evaluated for the treatment of solid tumors.
Lead Product(s): ANK-101,Aluminium Hydroxide
Therapeutic Area: Oncology Brand Name: ANK-101
Study Phase: Phase IProduct Type: Protein
Sponsor: Inapplicable
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable May 11, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : ANK-101,Aluminium Hydroxide
Therapeutic Area : Oncology
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Inapplicable
Deal Size : Inapplicable
Deal Type : Inapplicable
Ankyra Therapeutics Doses First Visceral Tumor Patient in ANCHOR Trial
Details : ANK-101 is an anchored drug complex composed of interleukin-12 (IL-12) linked to aluminum hydroxide. It is being evaluated for the treatment of solid tumors.
Product Name : ANK-101
Product Type : Protein
Upfront Cash : Inapplicable
May 11, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
ANK-101 is an anchored drug complex composed of interleukin-12 (IL-12) linked to aluminum hydroxide. It is being evaluated for the treatment of solid tumors.
Lead Product(s): ANK-101,Aluminium Hydroxide
Therapeutic Area: Oncology Brand Name: ANK-101
Study Phase: Phase IProduct Type: Protein
Sponsor: Inapplicable
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable May 03, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : ANK-101,Aluminium Hydroxide
Therapeutic Area : Oncology
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Inapplicable
Deal Size : Inapplicable
Deal Type : Inapplicable
Ankyra Doses First Patient in Phase 1 Trial of ANK-101 for Solid Tumors
Details : ANK-101 is an anchored drug complex composed of interleukin-12 (IL-12) linked to aluminum hydroxide. It is being evaluated for the treatment of solid tumors.
Product Name : ANK-101
Product Type : Protein
Upfront Cash : Inapplicable
May 03, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
AD04 is a novel immunotherapy for mild alzheimer’s disease. The compound has been used as an adjuvant in human and animal vaccination programs. It has demonstrated significantly slower decline in hippocampal volume as a biomarker.
Lead Product(s): Aluminium Hydroxide
Therapeutic Area: Neurology Brand Name: AD04
Study Phase: Phase IIProduct Type: Other Small Molecule
Sponsor: Inapplicable
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable November 27, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Aluminium Hydroxide
Therapeutic Area : Neurology
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : Inapplicable
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : AD04 is a novel immunotherapy for mild alzheimer’s disease. The compound has been used as an adjuvant in human and animal vaccination programs. It has demonstrated significantly slower decline in hippocampal volume as a biomarker.
Product Name : AD04
Product Type : Other Small Molecule
Upfront Cash : Inapplicable
November 27, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
AD04 (alhydrogel) is a novel immunotherapy for early alzheimer’s disease. The compound has been used as an adjuvant in human and animal vaccination programs. It has demonstrated significantly slower decline in hippocampal volume as a biomarker.
Lead Product(s): Aluminium Hydroxide
Therapeutic Area: Neurology Brand Name: AD04
Study Phase: Phase IIProduct Type: Other Small Molecule
Sponsor: Inapplicable
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable June 20, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Aluminium Hydroxide
Therapeutic Area : Neurology
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : Inapplicable
Deal Size : Inapplicable
Deal Type : Inapplicable
ADvantage Therapeutics Secures Approvals for Clinical Trial of AD04 in Alzheimer’s Disease Treat...
Details : AD04 (alhydrogel) is a novel immunotherapy for early alzheimer’s disease. The compound has been used as an adjuvant in human and animal vaccination programs. It has demonstrated significantly slower decline in hippocampal volume as a biomarker.
Product Name : AD04
Product Type : Other Small Molecule
Upfront Cash : Inapplicable
June 20, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
AD04™ (alhydrogel) is a novel immunotherapy for mild Alzheimer’s Disease under the Innovative Licensing and Access Pathway (ILAP). AD04™ also showed slower decline in hippocampal volume as a biomarker.
Lead Product(s): Aluminium Hydroxide
Therapeutic Area: Neurology Brand Name: AD04
Study Phase: Phase IIProduct Type: Other Small Molecule
Sponsor: Inapplicable
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable May 04, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Aluminium Hydroxide
Therapeutic Area : Neurology
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : Inapplicable
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : AD04™ (alhydrogel) is a novel immunotherapy for mild Alzheimer’s Disease under the Innovative Licensing and Access Pathway (ILAP). AD04™ also showed slower decline in hippocampal volume as a biomarker.
Product Name : AD04
Product Type : Other Small Molecule
Upfront Cash : Inapplicable
May 04, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
AD04™ (alhydrogel) is a novel immunotherapy for early alzheimer’s disease. The compound has been used as an adjuvant in human and animal vaccination programs. It has demonstrated significantly slower decline in hippocampal volume as a biomarker.
Lead Product(s): Aluminium Hydroxide
Therapeutic Area: Neurology Brand Name: AD04
Study Phase: Phase IIProduct Type: Other Small Molecule
Sponsor: Inapplicable
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable March 15, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Aluminium Hydroxide
Therapeutic Area : Neurology
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : Inapplicable
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : AD04™ (alhydrogel) is a novel immunotherapy for early alzheimer’s disease. The compound has been used as an adjuvant in human and animal vaccination programs. It has demonstrated significantly slower decline in hippocampal volume as a biomarker.
Product Name : AD04
Product Type : Other Small Molecule
Upfront Cash : Inapplicable
March 15, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
SCB-2019 antigen, a stabilized trimeric form of the S-protein based on the original strain of the SARS-CoV-2 virus, and combined it with Dynavax’s CpG 1018 advanced adjuvant and aluminum hydroxide (alum). Clover is developing it as a universal booster for COVID-19.
Lead Product(s): SCB-2019,Aluminium Hydroxide
Therapeutic Area: Infections and Infectious Diseases Brand Name: SCB-2019
Study Phase: Approved FDFProduct Type: Vaccine
Sponsor: Inapplicable
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable February 14, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : SCB-2019,Aluminium Hydroxide
Therapeutic Area : Infections and Infectious Diseases
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Inapplicable
Deal Size : Inapplicable
Deal Type : Inapplicable
Clover Launches COVID-19 Booster Vaccine in the People’s Republic of China
Details : SCB-2019 antigen, a stabilized trimeric form of the S-protein based on the original strain of the SARS-CoV-2 virus, and combined it with Dynavax’s CpG 1018 advanced adjuvant and aluminum hydroxide (alum). Clover is developing it as a universal booster ...
Product Name : SCB-2019
Product Type : Vaccine
Upfront Cash : Inapplicable
February 14, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
SCB-2019 antigen, a stabilized trimeric form of the S-protein based on the original strain of the SARS-CoV-2 virus, and combined it with Dynavax’s CpG 1018 advanced adjuvant and aluminum hydroxide (alum). Clover is developing it as a universal booster for COVID-19.
Lead Product(s): SCB-2019,Aluminium Hydroxide
Therapeutic Area: Infections and Infectious Diseases Brand Name: SCB-2019
Study Phase: Approved FDFProduct Type: Vaccine
Sponsor: Inapplicable
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable January 15, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : SCB-2019,Aluminium Hydroxide
Therapeutic Area : Infections and Infectious Diseases
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Inapplicable
Deal Size : Inapplicable
Deal Type : Inapplicable
Clover Provides Updates on COVID-19 Vaccine Commercial Launch and Strategic Priorities in 2023
Details : SCB-2019 antigen, a stabilized trimeric form of the S-protein based on the original strain of the SARS-CoV-2 virus, and combined it with Dynavax’s CpG 1018 advanced adjuvant and aluminum hydroxide (alum). Clover is developing it as a universal booster ...
Product Name : SCB-2019
Product Type : Vaccine
Upfront Cash : Inapplicable
January 15, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
VLA2001 consists of inactivated whole virus particles of SARS-CoV-2 with high S-protein density, in combination with two adjuvants, alum and CpG 1018. VLA2001’s shelf life is expected to be extended to up to 24 months, compared to 18 months currently.
Lead Product(s): VLA2001,Aluminium Hydroxide
Therapeutic Area: Infections and Infectious Diseases Brand Name: VLA2001
Study Phase: Phase IIIProduct Type: Vaccine
Sponsor: Inapplicable
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable December 30, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : VLA2001,Aluminium Hydroxide
Therapeutic Area : Infections and Infectious Diseases
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Inapplicable
Deal Size : Inapplicable
Deal Type : Inapplicable
Valneva Reports Further Heterologous Booster Data For Its Inactivated COVID-19 Vaccine
Details : VLA2001 consists of inactivated whole virus particles of SARS-CoV-2 with high S-protein density, in combination with two adjuvants, alum and CpG 1018. VLA2001’s shelf life is expected to be extended to up to 24 months, compared to 18 months currently.
Product Name : VLA2001
Product Type : Vaccine
Upfront Cash : Inapplicable
December 30, 2022

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Originator
Registration Country : South Africa
Brand Name : Mucaine Suspension
Dosage Form : SUS
Dosage Strength : 291mg/5ml
Packaging : 100X1mg/5ml
Approval Date :
Application Number :
Regulatory Info : Originator
Registration Country : South Africa
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Originator
Registration Country : South Africa
Brand Name : Mucaine Suspension
Dosage Form : SUS
Dosage Strength : 291mg/5ml
Packaging : 200X1mg/5ml
Approval Date :
Application Number :
Regulatory Info : Originator
Registration Country : South Africa
 Octavius Pharma has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.
Octavius Pharma has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.
Regulatory Info :
Registration Country : India
Magnesium Trisilicate; Aluminium Hydroxide
Brand Name : Magnesium Trisilicate; Aluminium Hydroxide
Dosage Form : DC Granules
Dosage Strength : 250MG; 200MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : Microgel Suspension
Dosage Form : SUS
Dosage Strength : 200mg
Packaging : 350X1mg
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : Dicyclomine Co Gel
Dosage Form : SUS
Dosage Strength : 400mg/10ml
Packaging : 100X1mg/10ml
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Magnesium Hydroxide; aluminium(III) hydroxide; Dianeusine; Dimethicone
Brand Name :
Dosage Form : Tablet
Dosage Strength : 250MG; 250MG; 50MG; 50MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Aluminium oxide hydrochloride (Algeldratum); Magnesium hydroxide
Brand Name : Alucol Gel
Dosage Form : Susp Peppermint
Dosage Strength :
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Zimbabwe
Brand Name : Novacid
Dosage Form : Chewable Tablet
Dosage Strength : 216MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Zimbabwe

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Gel
Dosage Strength : 600MG/5ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

Regulatory Info :
Registration Country : Sweden
Several substances refer to content
Brand Name : Gaviscon
Dosage Form : ORAL SUSPENSION
Dosage Strength : -
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : Yes
TE Code :
ALUMINUM HYDROXIDE; MAGNESIUM TRISILICATE
Brand Name : GAVISCON
Dosage Form : TABLET, CHEWABLE;ORAL
Dosage Strength : 80MG;20MG **Federal Register determination that product was not discontinued or withdrawn for safety or effectiveness reasons**
Approval Date : 1983-12-09
Application Number : 18685
RX/OTC/DISCN : DISCN
RLD : Yes
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : Yes
TE Code :
ALUMINUM HYDROXIDE; MAGNESIUM TRISILICATE
Brand Name : GAVISCON
Dosage Form : TABLET, CHEWABLE;ORAL
Dosage Strength : 160MG;40MG **Federal Register determination that product was not discontinued or withdrawn for safety or effectiveness reasons**
Approval Date : 1983-12-09
Application Number : 18685
RX/OTC/DISCN : DISCN
RLD : Yes
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code :
ALUMINUM HYDROXIDE; MAGNESIUM TRISILICATE
Brand Name : FOAMCOAT
Dosage Form : TABLET, CHEWABLE;ORAL
Dosage Strength : 80MG;20MG
Approval Date : 1987-09-04
Application Number : 71793
RX/OTC/DISCN : DISCN
RLD : No
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code :
ALUMINUM HYDROXIDE; MAGNESIUM TRISILICATE
Brand Name : FOAMICON
Dosage Form : TABLET, CHEWABLE;ORAL
Dosage Strength : 80MG;20MG
Approval Date : 1989-06-28
Application Number : 72687
RX/OTC/DISCN : DISCN
RLD : No
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code :
ALUMINUM HYDROXIDE; MAGNESIUM TRISILICATE
Brand Name : ALUMINUM HYDROXIDE AND MAGNESIUM TRISILICATE
Dosage Form : TABLET, CHEWABLE;ORAL
Dosage Strength : 80MG;20MG
Approval Date : 1987-11-27
Application Number : 89449
RX/OTC/DISCN : DISCN
RLD : No
TE Code :

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Aluminium oxide hydrochloride (Algeldratum); Magnesium hydroxide
Brand Name : Alucol Gel
Dosage Form : Susp Fruit
Dosage Strength :
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Aluminium oxide hydrochloride (Algeldratum); Magnesium hydroxide
Brand Name : Alucol Gel
Dosage Form : Susp Fruit
Dosage Strength :
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Aluminium oxide hydrochloride (Algeldratum); Magnesium hydroxide
Brand Name : Alucol Gel
Dosage Form : Susp Peppermint
Dosage Strength :
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Aluminium oxide hydrochloride (Algeldratum); Magnesium hydroxide
Brand Name : Alucol Gel
Dosage Form : Susp Peppermint
Dosage Strength :
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

Regulatory Info :
Registration Country : Sweden
Several substances refer to content
Brand Name : Gaviscon
Dosage Form : ORAL SUSPENSION
Dosage Strength : -
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info :
Registration Country : Sweden
Several substances refer to content
Brand Name : Gaviscon
Dosage Form : CHEWABLE TABLET
Dosage Strength : -
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Canada
Brand Name : STOMAAX PLUS
Dosage Form : SUSPENSION
Dosage Strength : 200MG/5ML
Packaging : 350 ML
Approval Date :
Application Number : 623709
Regulatory Info :
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Canada
Brand Name : ALMAGEL PLUS SUS
Dosage Form : SUSPENSION
Dosage Strength : 200MG/5ML
Packaging : 100/350ML
Approval Date :
Application Number : 815527
Regulatory Info :
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : OTC
Registration Country : Canada
Brand Name : PMS-ALUMINA MAG AND SIMETHICONE SUS
Dosage Form : SUSPENSION
Dosage Strength : 2005MG/5ML
Packaging : 350ML
Approval Date :
Application Number :
Regulatory Info : OTC
Registration Country : Canada

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Originator
Registration Country : South Africa
Brand Name : Mucaine Suspension
Dosage Form : SUS
Dosage Strength : 291mg/5ml
Packaging : 100X1mg/5ml
Approval Date :
Application Number :
Regulatory Info : Originator
Registration Country : South Africa
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Originator
Registration Country : South Africa
Brand Name : Mucaine Suspension
Dosage Form : SUS
Dosage Strength : 291mg/5ml
Packaging : 200X1mg/5ml
Approval Date :
Application Number :
Regulatory Info : Originator
Registration Country : South Africa
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : Microgel Suspension
Dosage Form : SUS
Dosage Strength : 200mg
Packaging : 350X1mg
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : Betaclomin Gel
Dosage Form : SUS
Dosage Strength : 400mg/10ml
Packaging : 100X1mg/10ml
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : Dicyclomine Co Gel
Dosage Form : SUS
Dosage Strength : 400mg/10ml
Packaging : 100X1mg/10ml
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : Dicyclomine Co Gel
Dosage Form : SUS
Dosage Strength : 400mg/10ml
Packaging : 200X1mg/10ml
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : Dicyclomine Co Gel
Dosage Form : SUS
Dosage Strength : 400mg/10ml
Packaging : 200X1mg/10ml
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa

Regulatory Info : Generic
Registration Country : South Africa
Brand Name : Amgel
Dosage Form : LIQ
Dosage Strength : 400mg
Packaging : 100X1mg
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : MEDDEV SUSPENSION
Dosage Form : SUS
Dosage Strength : 400mg
Packaging : 200X1mg
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info : Originator
Registration Country : South Africa
Brand Name : Bisma-Rex Powder
Dosage Form : POW
Dosage Strength : 19mg
Packaging : 450X1mg
Approval Date :
Application Number :
Regulatory Info : Originator
Registration Country : South Africa

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Octavius Pharma has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.
Octavius Pharma has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.
Regulatory Info :
Registration Country : India
Magnesium Trisilicate; Aluminium Hydroxide
Brand Name : Magnesium Trisilicate;...
Dosage Form : DC Granules
Dosage Strength : 250MG; 200MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
 Octavius Pharma has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.
Octavius Pharma has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.
Packaging :
Regulatory Info :
Magnesium Trisilicate; Aluminium Hydroxide
Dosage : DC Granules
Dosage Strength : 250MG; 200MG
Brand Name : Magnesium Trisilicate;...
Approval Date :
Application Number :
Registration Country : India
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Magnesium trisilicate (USP); Aluminum hyroxide
Brand Name : Appyacid
Dosage Form : Chewable Tablet
Dosage Strength : 250MG; 120MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Magnesium trisilicate (USP); Aluminum hyroxide
Dosage : Chewable Tablet
Dosage Strength : 250MG; 120MG
Brand Name : Appyacid
Approval Date :
Application Number :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : India
Brand Name :
Dosage Form : Tablet
Dosage Strength : 500MG
Packaging :
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info : Generic
Dosage : Tablet
Dosage Strength : 500MG
Brand Name :
Approval Date :
Application Number :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Tablet
Dosage Strength : 500MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Dosage : Tablet
Dosage Strength : 500MG
Brand Name :
Approval Date :
Application Number :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Venezuela
Aluminium Hydroxide; Magnesium Hydroxide; Simethicone
Brand Name : Acifar
Dosage Form : Oral suspension
Dosage Strength :
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Venezuela

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Aluminium Hydroxide; Magnesium Hydroxide; Simethicone
Dosage : Oral suspension
Dosage Strength :
Brand Name : Acifar
Approval Date :
Application Number :
Registration Country : Venezuela

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Magnesium Trisilicate; Aluminium Hydroxide
Brand Name :
Dosage Form : DC Granules
Dosage Strength : 250MG; 200MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Magnesium Trisilicate; Aluminium Hydroxide
Dosage : DC Granules
Dosage Strength : 250MG; 200MG
Brand Name :
Approval Date :
Application Number :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Magnesium Hydroxide; aluminium(III) hydroxide; Dianeusine; Dimethicone
Brand Name :
Dosage Form : Tablet
Dosage Strength : 250MG; 250MG; 50MG; 50...
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Magnesium Hydroxide; aluminium(III) hydroxide; Dianeusine; Dimethicone
Dosage : Tablet
Dosage Strength : 250MG; 250MG; 50MG; 50...
Brand Name :
Approval Date :
Application Number :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Aluminium Hydroxide; Magnesium Hydroxide; Dimethicone
Brand Name :
Dosage Form : Tablet
Dosage Strength : 250MG; 250MG; 50MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Aluminium Hydroxide; Magnesium Hydroxide; Dimethicone
Dosage : Tablet
Dosage Strength : 250MG; 250MG; 50MG
Brand Name :
Approval Date :
Application Number :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Gel
Dosage Strength : 600MG/5ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Dosage : Gel
Dosage Strength : 600MG/5ML
Brand Name :
Approval Date :
Application Number :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Turkey
Aluminum Hydroxide; Magnesium Hydroxide; Simethicone
Brand Name : SIMALGEL
Dosage Form : SUSPENSION
Dosage Strength : 405MG/5ML; 100MG/5ML; ...
Packaging : 250 ML/BOTTLE
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Turkey

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging : 250 ML/BOTTLE
Regulatory Info :
Aluminum Hydroxide; Magnesium Hydroxide; Simethicone
Dosage : SUSPENSION
Dosage Strength : 405MG/5ML; 100MG/5ML; ...
Brand Name : SIMALGEL
Approval Date :
Application Number :
Registration Country : Turkey

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Related Excipient Companies

Dosage Form : Tablet
Grade : Oral
Dosage Form : Cream / Lotion / Ointment
Grade : Topical, Parenteral
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Dosage Form : Suspension
Grade : Oral
Dosage Form : Tablet
Grade : Oral
Dosage Form : Capsule
Grade : Topical, Oral
Dosage Form : Tablet
Grade : Oral
Application : Taste Masking
Excipient Details : Mannitol is used as a filler, bulking agent and taste masking agent in ODT formulations such as tablets.
Dosage Form : Suspension
Grade : Oral
Dosage Form : Tablet
Grade : Oral
Dosage Form : Suspension
Grade : Oral
Dosage Form : Cream / Lotion / Ointment
Grade : Topical
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Dosage Form : Tablet
Grade : Oral
Dosage Form : Tablet
Grade : Oral
Dosage Form : Tablet
Grade : Oral
Application : Taste Masking
Excipient Details : CM90 is a directly compressible, granulated calcium carbonate with maltodextrin used for swallow tablets due to its high density and compressibility.
Pharmacopoeia Ref : NA
Technical Specs : PSD D50: 490- 500 µm; Tapped Density: 1.50
Ingredient(s) : Calcium Carbonate Excipient
Dosage Form : Tablet
Grade : Oral
Dosage Form : Tablet
Grade : Oral
Application : Taste Masking
Excipient Details : CS90 is a directly compressible calcium carbonate with starch used for chewable tablets due to its smooth mouthfeel and creamy texture.
Pharmacopoeia Ref : NA
Technical Specs : PSD D50: 150-175 µm, Tapped Density: 0.85
Ingredient(s) : Calcium Carbonate Excipient
Dosage Form : Orodispersible Tablet
Grade : Oral
Dosage Form : Tablet
Grade : Oral
Application : Direct Compression
Excipient Details : DC SIM 100 is a directly compressible simethicone powder used for antacid and anti-gas tablets.
Dosage Form : Tablet
Grade : Oral
Dosage Form : Orodispersible Tablet
Grade : Oral
Dosage Form : Tablet
Grade : Oral
Application : Taste Masking
Excipient Details : MS90 is a directly compressible magnesium hydroxide with starch used for chewable tablets due to its smooth mouthfeel and creamy texture.
Pharmacopoeia Ref : NA
Technical Specs : PSD D50: 150-170 µm, Tapped Density: 0.80
Ingredient(s) : Magnesium Hydroxide Excipient
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
24
PharmaCompass offers a list of Aluminium Hydroxide API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Aluminium Hydroxide manufacturer or Aluminium Hydroxide supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Aluminium Hydroxide manufacturer or Aluminium Hydroxide supplier.
PharmaCompass also assists you with knowing the Aluminium Hydroxide API Price utilized in the formulation of products. Aluminium Hydroxide API Price is not always fixed or binding as the Aluminium Hydroxide Price is obtained through a variety of data sources. The Aluminium Hydroxide Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Algeldrato manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Algeldrato, including repackagers and relabelers. The FDA regulates Algeldrato manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Algeldrato API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Algeldrato manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Algeldrato supplier is an individual or a company that provides Algeldrato active pharmaceutical ingredient (API) or Algeldrato finished formulations upon request. The Algeldrato suppliers may include Algeldrato API manufacturers, exporters, distributors and traders.
click here to find a list of Algeldrato suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Algeldrato DMF (Drug Master File) is a document detailing the whole manufacturing process of Algeldrato active pharmaceutical ingredient (API) in detail. Different forms of Algeldrato DMFs exist exist since differing nations have different regulations, such as Algeldrato USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Algeldrato DMF submitted to regulatory agencies in the US is known as a USDMF. Algeldrato USDMF includes data on Algeldrato's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Algeldrato USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Algeldrato suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Algeldrato Drug Master File in Japan (Algeldrato JDMF) empowers Algeldrato API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Algeldrato JDMF during the approval evaluation for pharmaceutical products. At the time of Algeldrato JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Algeldrato suppliers with JDMF on PharmaCompass.
A Algeldrato written confirmation (Algeldrato WC) is an official document issued by a regulatory agency to a Algeldrato manufacturer, verifying that the manufacturing facility of a Algeldrato active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Algeldrato APIs or Algeldrato finished pharmaceutical products to another nation, regulatory agencies frequently require a Algeldrato WC (written confirmation) as part of the regulatory process.
click here to find a list of Algeldrato suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Algeldrato as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Algeldrato API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Algeldrato as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Algeldrato and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Algeldrato NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Algeldrato suppliers with NDC on PharmaCompass.
Algeldrato Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Algeldrato GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Algeldrato GMP manufacturer or Algeldrato GMP API supplier for your needs.
A Algeldrato CoA (Certificate of Analysis) is a formal document that attests to Algeldrato's compliance with Algeldrato specifications and serves as a tool for batch-level quality control.
Algeldrato CoA mostly includes findings from lab analyses of a specific batch. For each Algeldrato CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Algeldrato may be tested according to a variety of international standards, such as European Pharmacopoeia (Algeldrato EP), Algeldrato JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Algeldrato USP).
