
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
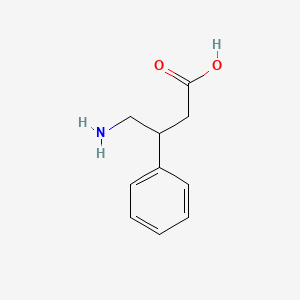

1. 4-amino-3-phenylbutyric Acid
2. 4-amino-3-phenylbutyric Acid Hydrochloride
3. 4-amino-3-phenylbutyric Acid, (dl)-(+-)-mixture
4. 4-amino-3-phenylbutyric Acid, (l)-(-)-isomer
5. Beta-(aminomethyl)benzenepropanoic Acid
6. Beta-(aminomethyl)hydrocinnamic Acid
7. Beta-phenyl-gamma-aminobutyric Acid
8. Citrocard
9. Fenibut
10. Fenibut Citrate
11. Noophen
12. Phenibut
13. Phenybut
14. Phenyl-gaba
1. Phenibut
2. 1078-21-3
3. Fenibut
4. 4-amino-3-phenylbutyric Acid
5. Phgaba
6. Phenylgamma
7. Phenigamma
8. Fenigama
9. Phenigam
10. Phenylgam
11. Pgaba
12. Fenigam
13. Phenygam
14. Beta-phenyl-gamma-aminobutyric Acid
15. Beta-(aminomethyl)hydrocinnamic Acid
16. Beta-phenyl-gamma-aminobutyrate
17. 4-amino-3-phenyl-butyric Acid
18. Beta-(aminomethyl)benzenepropanoic Acid
19. Benzenepropanoic Acid, .beta.-(aminomethyl)-
20. 4-amino-3-pheny-butyric Acid
21. T2m58d6la8
22. Dl-4-amino-3-phenylbutanoic Acid
23. Phenyl-gaba
24. Phenigama
25. Einecs 214-079-6
26. Brn 2804903
27. Unii-t2m58d6la8
28. Benzenepropanoic Acid, Beta-(aminomethyl)-
29. Phenibut; Fenibut
30. Benzenepropanoic Acid, Beta-(aminomethyl)-, Hydrochloride
31. Butyric Acid, 4-amino-3-phenyl-
32. Phenibut [who-dd]
33. Oprea1_183000
34. Oprea1_381119
35. 4-14-00-01723 (beilstein Handbook Reference)
36. (+/-)-fenibut
37. Schembl340838
38. 3-phenyl-4-aminobutanoic Acid
39. Chembl315818
40. Dtxsid70870838
41. Ex-a734
42. Chebi:136039
43. Hms3886k05
44. (s)-4-amino-3-phenylbutanoicacid
45. Albb-022462
46. Bcp10710
47. Bbl000576
48. Mfcd00456233
49. S4892
50. Stk182861
51. (+/-)-.beta.-phenyl-gaba
52. 3-amino-2-phenylpropanecarboxylic Acid
53. Akos000555914
54. Akos016347357
55. Ccg-266416
56. Db13455
57. Gs-3145
58. Benzenepropanoic Acid, B-(aminomethyl)-
59. 4-amino-3-phenylbutyric Acid [mi]
60. Ft-0600356
61. .beta.-(aminomethyl)hydrocinnamic Acid
62. D10509
63. D96082
64. .beta.-(aminomethyl)benzenepropanoic Acid
65. .beta.-phenyl-.gamma.-aminobutyric Acid
66. 078a213
67. Q419559
68. Z1436181457
| Molecular Weight | 179.22 g/mol |
|---|---|
| Molecular Formula | C10H13NO2 |
| XLogP3 | -1.6 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 4 |
| Exact Mass | 179.094628657 g/mol |
| Monoisotopic Mass | 179.094628657 g/mol |
| Topological Polar Surface Area | 63.3 Ų |
| Heavy Atom Count | 13 |
| Formal Charge | 0 |
| Complexity | 164 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Anticonvulsants
Drugs used to prevent SEIZURES or reduce their severity. (See all compounds classified as Anticonvulsants.)
Hypnotics and Sedatives
Drugs used to induce drowsiness or sleep or to reduce psychological excitement or anxiety. (See all compounds classified as Hypnotics and Sedatives.)
Convulsants
Substances that act in the brain stem or spinal cord to produce tonic or clonic convulsions, often by removing normal inhibitory tone. They were formerly used to stimulate respiration or as antidotes to barbiturate overdose. They are now most commonly used as experimental tools. (See all compounds classified as Convulsants.)
GABA Agonists
Endogenous compounds and drugs that bind to and activate GAMMA-AMINOBUTYRIC ACID receptors (RECEPTORS, GABA). (See all compounds classified as GABA Agonists.)
Tranquilizing Agents
A traditional grouping of drugs said to have a soothing or calming effect on mood, thought, or behavior. Included here are the ANTI-ANXIETY AGENTS (minor tranquilizers), ANTIMANIC AGENTS, and the ANTIPSYCHOTIC AGENTS (major tranquilizers). These drugs act by different mechanisms and are used for different therapeutic purposes. (See all compounds classified as Tranquilizing Agents.)
N - Nervous system
N06 - Psychoanaleptics
N06B - Psychostimulants, agents used for adhd and nootropics
N06BX - Other psychostimulants and nootropics
N06BX22 - Phenibut
Market Place

ABOUT THIS PAGE
14
PharmaCompass offers a list of 4-Amino-3-Phenylbutanoic Acid API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right 4-Amino-3-Phenylbutanoic Acid manufacturer or 4-Amino-3-Phenylbutanoic Acid supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred 4-Amino-3-Phenylbutanoic Acid manufacturer or 4-Amino-3-Phenylbutanoic Acid supplier.
A 4-Amino-3-Phenylbutanoic Acid manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of 4-Amino-3-Phenylbutanoic Acid, including repackagers and relabelers. The FDA regulates 4-Amino-3-Phenylbutanoic Acid manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. 4-Amino-3-Phenylbutanoic Acid API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of 4-Amino-3-Phenylbutanoic Acid manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A 4-Amino-3-Phenylbutanoic Acid supplier is an individual or a company that provides 4-Amino-3-Phenylbutanoic Acid active pharmaceutical ingredient (API) or 4-Amino-3-Phenylbutanoic Acid finished formulations upon request. The 4-Amino-3-Phenylbutanoic Acid suppliers may include 4-Amino-3-Phenylbutanoic Acid API manufacturers, exporters, distributors and traders.
click here to find a list of 4-Amino-3-Phenylbutanoic Acid suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
4-Amino-3-Phenylbutanoic Acid Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of 4-Amino-3-Phenylbutanoic Acid GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right 4-Amino-3-Phenylbutanoic Acid GMP manufacturer or 4-Amino-3-Phenylbutanoic Acid GMP API supplier for your needs.
A 4-Amino-3-Phenylbutanoic Acid CoA (Certificate of Analysis) is a formal document that attests to 4-Amino-3-Phenylbutanoic Acid's compliance with 4-Amino-3-Phenylbutanoic Acid specifications and serves as a tool for batch-level quality control.
4-Amino-3-Phenylbutanoic Acid CoA mostly includes findings from lab analyses of a specific batch. For each 4-Amino-3-Phenylbutanoic Acid CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
4-Amino-3-Phenylbutanoic Acid may be tested according to a variety of international standards, such as European Pharmacopoeia (4-Amino-3-Phenylbutanoic Acid EP), 4-Amino-3-Phenylbutanoic Acid JP (Japanese Pharmacopeia) and the US Pharmacopoeia (4-Amino-3-Phenylbutanoic Acid USP).

