The year
2020 was an eventful year for the pharmaceutical industry, with several
companies across the world working at a feverish pace to find a treatment or a
vaccine for the raging Covid-19, which has so far taken over 1.79 million lives
worldwide.
With
countries imposing lockdowns and regulators putting on-site inspections on
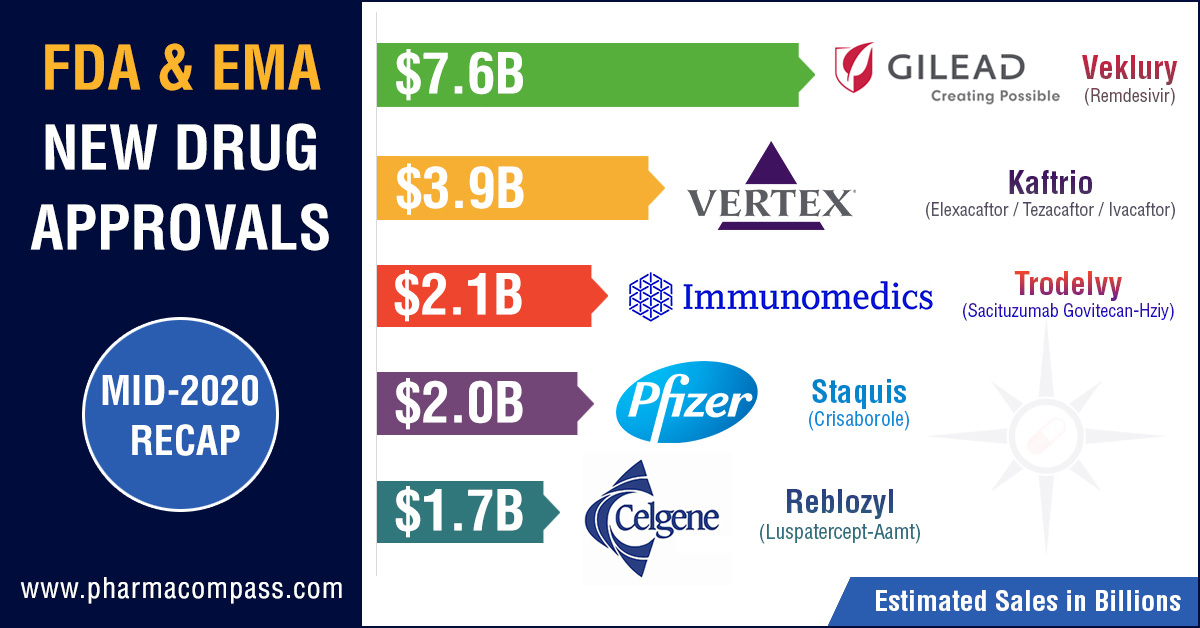
hold, we were expecting far lower new drug approvals in mid-2020. But our mid-2020 recap published in July, which
looked at new drug approvals by the US Food and Drug Administration (FDA) and European Medical Agency (EMA),
found that the FDA had approved 33 new drugs by the end of June. This put the
approvals within the ballpark of the past two years.
This week, we bring you a roundup of 2020, a tumultuous year when 58 drugs (53 approvals by the Center for Drug Evaluation and Research and 5 by the Center for Biologics Evaluation and Research) bagged FDA’s new drug approvals. While this number is lower than the number of drugs approved in 2018 (62), it is higher than the number for 2019 (54). Out of this, while 23 approvals were in the field of oncology, 9 were for infectious diseases and infections, 8 for genetic diseases, 7 for neurology, 3 for immunology and 2 for gastroenterology.
View New Drug Approvals in 2020 with Estimated Sales (Free Excel Available)
A year marked by EUA
With the pandemic raging across the world, emergency use authorizations (EUAs) dominated news headlines in 2020 — the FDA issued 10 EUAs, with the most
prominent being those issued to Pfizer-BioNTech and Moderna for their Covid-19 vaccines.
EMA was busy as well since they issued 75 positive opinions
with Novartis leading
the pack with 8, followed by Pfizer and Sanofi which received 4 each.
The EUAs came with their own set of controversies. In March,
the FDA had issued an EUA “for oral formulations of chloroquine phosphate and hydroxychloroquine sulfate
for the treatment of” Covid-19. However, by June, FDA had revoked the EUA,
as the agency determined that chloroquine and
hydroxychloroquine were not likely to be effective in treating Covid-19 for the
authorized uses in the EUA.
Amongst treatments for Covid-19, in May the FDA
authorized the emergency use of Gilead’s antiviral
drug remdesivir. In our mid-2020 recap, Gilead’s remdesivir was on top of
our list of top-selling drugs after it received an EUA from the FDA.
In October, remdesivir became the first drug to
be approved by the FDA for treatment of Covid-19 patients requiring
hospitalization. While analysts predicted US$ 3.5 billion in revenue in early
October, the future of this drug as a treatment for Covid-19 in hospitalized
patients remains uncertain, especially in wake of results from the World Health
Organization (WHO)-led Solidarity Trial that said Gilead’s remdesivir
had little or no effect on the 28-day mortality or length of hospital
stays for Covid-19 patients. The FDA approved remdesivir for
hospitalized patients a week after the WHO results.
View New Drug Approvals in 2020 with Estimated Sales (Free Excel Available)
Gilead US$ 21 billion Immunomedics acquisition
Immunomedics' antibody-drug conjugate (ADC) — Trodelvy (sacituzumab govitecan-hziy) — was approved by the FDA in April this year for the treatment of adult patients with metastatic triple-negative breast cancer who have received at least two prior therapies for the disease. Such tumor types account for 15 to 20 percent of breast cancers. Trodelvy follows remdesivir in our list of FDA approved drugs in 2020 with the highest sales potential. The current forecast for Trodelvy sales is US$ 2.151 billion by 2026.
In
September, Gilead made a big move and acquired biotech company
Immunomedics Inc for US$ 21 billion. The
transaction, which was completed in October, will strengthen Gilead’s cancer portfolio and add another potential blockbuster to it.
Immunomedics
plans to submit a supplemental Biologics License Application (BLA) to support
full approval of Trodelvy in the US over the next quarter. According to a statement, Immunomedics
is also on track to file for regulatory approval of the drug in Europe in the
first half of 2021. Moreover, ongoing studies are also evaluating the potential
of Trodelvy as a treatment for non-small cell lung cancer and other types of
solid tumors.
View New Drug Approvals in 2020 with Estimated Sales (Free Excel Available)
Roche-PTC Therapeutics’ risdiplam bags approval
Following
Troveldy in sales potential for drugs approved by the FDA is Roche and PTC Therapeutics’ drug Evrysdi (risdiplam), the first oral medicine approved for the
rare genetic disease, spinal muscular atrophy, which until four years ago had
no available treatments.
The approval of Evrysdi presents patients and their families with a unique choice between a one-time gene therapy, an RNA-based drug infused three times a year at the doctor’s office and a daily medicine taken at home.
Roche priced the drug by patient weight, with a maximum cost of US$ 340,000 per year — substantially lesser than the competing (and approved) therapies from Biogen and Novartis.
View New Drug Approvals in 2020 with Estimated Sales (Free Excel Available)
Vertex’s Kaftrio bags EMA approval
Earlier
this year, PharmaCompass had published its compilation of sales
forecasts for the new drugs approved by the FDA in 2019. The list was led by Vertex’s cystic
fibrosis treatment — Trikafta — which was expected to have sales of US$ 3.935 billion by 2024.
Trikafta
is a combination of ivacaftor, tezacaftor and elexacaftor and its
stellar clinical data made the FDA approve the drug within three months of
Vertex’s application
filing and five months before FDA’s action date.
In
June 2020, EMA’s CHMP adopted a positive opinion, recommending the granting of a marketing authorization for
Vertex’s
combination, which will be marketed as Kaftrio.
View New Drug Approvals in 2020 with Estimated Sales (Free Excel Available)
A year
of multiple setbacks
Not everything went smoothly in 2020. In fact, the year saw several setbacks — almost 44 drugs were not granted approval by the FDA. Bristol Myers Squibb was one such company that
received setbacks. As part of Bristol’s US$ 74 billion acquisition of Celgene, the New
York-headquartered drug company offered Celgene shareholders Contingent Value Rights or
CVRs. But to realize the US$ 9-a piece payment, approvals for three
ex-Celgene drugs must meet their pre-specified deadlines.
While in March, the FDA approved Bristol’s ozanimod, a sphingosine-1-phosphate receptor agonist for the treatment of relapsing multiple sclerosis, well ahead of the December 31, 2020 deadline, in May, Bristol Myers Squibb and bluebird bio, Inc announced
that they have received a Refusal to File letter from FDA regarding the Biologics License Application (BLA) for their CAR-T therapy — idecabtagene vicleucel
(ide-cel) — for patients with heavily pre-treated relapsed and refractory multiple myeloma, which was submitted in March 2020. Upon preliminary review, the FDA
determined that the Chemistry, Manufacturing and Control (CMC) module of the
BLA requires further details to complete the review.
This was
followed by Bristol Myers announcing that the FDA has extended the action date
by three months for the BLA for lisocabtagene maraleucel (liso-cel), a
CD19-directed CAR-T therapy for the treatment of adults with relapsed or
refractory (R/R) large B-cell lymphoma after at least two prior therapies. The
FDA had then set the new Prescription Drug User Fee Act (PDUFA) action date as
November 16, 2020. However, on that day, FDA informed the company that its
review of the BLA for liso-cel will not be completed by November 16.
The FDA
approval of liso-cel by December 31, 2020 is one of the required remaining
milestones of the CVRs issued upon the close of the Celgene acquisition in the
fourth quarter of 2019. The other is FDA approval of ide-cel by March 31, 2021.
FDA declines approval to Novartis’ inclisiran: Recently, there was news that
the FDA declined to approve Swiss drugmaker Novartis AG’s lipid-lowering therapy, inclisiran (branded as Leqvio). The drug came to Novartis' fold through the
US$ 9.7 billion acquisition of The Medicines Company last year. The drug has been cleared by the European Commission.
In a statement, Novartis said the
FDA has not raised any concerns
related to the efficacy or safety of inclisiran. The complete
response letter (CRL) is due to unresolved facility inspection-related
conditions. No onsite inspection was conducted by the FDA, the company said.
View New Drug Approvals in 2020 with Estimated Sales (Free Excel Available)
Our
view
Like the
drug companies, regulators across the world also worked at a frenetic pace in
order to accelerate EUAs and drug approvals, especially for treatments and
vaccines for Covid-19.
Apart from
Covid-19, the year saw wider adoption and
approvals for cell and gene therapies along with approvals of several innovative medicines like relugolix (the first
oral gonadotropin-releasing hormone receptor antagonist for the treatment of
adult patients with advanced prostate cancer), berotralstat
the first oral once daily plasma kallikrein inhibitor to prevent attacks of
hereditary angioedema in adults and pediatric patients 12 years and older,
lumasiran (an HAO1-directed small interfering
ribonucleic acid indicated for the treatment of primary hyperoxaluria type 1 to lower urinary oxalate levels in pediatric and adult
patients) and osilodrostat for the treatment of adults with Cushing’s disease).
Moreover, there were several interesting ‘non-Covid’ medical breakthroughs that took place during 2020. One such breakthrough is a single pill that combines four medications meant to
lower blood pressure and cholesterol and aspirin that was found to cut the risk
of heart disease.
While the agility shown by pharma companies and regulators was
undoubtedly quite impressive, with many countries granting EUA to vaccines for
Covid-19, the ongoing pandemic did put mankind in a bind for several months.
And if the words of the WHO director general Tedros
Adhanom Ghebreyesus are to be believed, the
coronavirus crisis will not be the last pandemic and attempts to improve human health are “doomed” without tackling climate change and animal welfare.
If that really proves to be the case, the pharma industry has a lot to learn from this pandemic, and science has a lot to demonstrate if mankind is to emerge unscathed from such adversities.
View New Drug Approvals in 2020 with Estimated Sales (Free Excel Available)
Impressions: 6870
In case you thought Covid-19 had slowed down US Food and Drug Administration’s New Drug Approvals, you’re in for a pleasant surprise — the FDA appears to be more active than ever before. By the end of June, the FDA had already approved 33 new drugs which put the approval activities within the ballpark of the past two years — 62 novel drugs were approved in 2018, while 54 were approved in 2019.
FDA’s Center for Drug Evaluation and Research (CDER) approved 25 new molecular entities and new therapeutic biological products, of which almost half — 12 out of 25 — were oncology drugs, while the rest of
the novel therapies were approved by the Center for Biologics Evaluation and Research.
The European Medical Agency (EMA) was also busy as the regulator issued a positive opinion for 41 drugs, of which 27 were classified as novel treatments.
View New Drug Approvals by June 2020 with Estimated Sales (Free Excel Available)
Conditional Approval for Gilead’s Remdesivir
Gilead’s Remdesivir has
certainly been one of the most talked about drugs
this year. While it is still under clinical evaluation, the FDA, EMA and the
Japanese Ministry of Health, Labour and Welfare (MHLW) found ways of providing
market access to this drug as a treatment against Covid-19.
On May 1, 2020, based on the totality of
scientific evidence available to the FDA, the
agency issued an Emergency Use Authorization (EUA), as it believed
that remdesivir may be effective
in treating Covid-19 and that the known and
potential benefits of remdesivir, when used to
treat Covid-19, outweigh the known and potential risks of such products.
On June 25, 2020, EMA’s Committee for Medicinal Products for Human Use (CHMP) adopted a positive opinion, recommending the granting of conditional marketing authorization for Veklury (remdesivir).
The demand for remdesivir is such that the US bought more than 500,000 doses, which is all of Gilead’s production for July and 90 percent of production for August and September, leaving almost
no stock of remdesivir for the UK and Europe.
In 127 poor or middle-income countries, Gilead
is allowing generic drugmakers to supply remdesivir. It has signed non-exclusive voluntary licensing
agreements with generic pharmaceutical manufacturers based in Egypt, India and
Pakistan to further expand the supply of the antiviral drug.
View New Drug Approvals by June 2020 with Estimated Sales (Free Excel Available)
Vertex’s Kaftrio bags EMA approval
Earlier this year, PharmaCompass published its compilation of the sales forecasts for the new drugs approved by the FDA in 2019. The list was led by Vertex’s cystic fibrosis treatment — Trikafta — which is expected to have sales of US$ 3.935 billion by 2024.
Trikafta is a combination of ivacaftor, tezacaftor and elexacaftor and its stellar clinical data made the FDA approve the drug within three months of Vertex’s application filing and five months before FDA’s action date.
In June 2020, EMA’s CHMP adopted a positive opinion, recommending the granting of a marketing authorization for Vertex’s combination which will be marketed as Kaftrio.
EMA also adopted positive opinions on other
drugs which were previously approved by the FDA in
2019, such as Novartis’ Zolgensma and Piqray, Pfizer’s Staquis and Daurismo among many
others.
Immunomedic’s antibody-drug conjugate (ADC) — Trodelvy (sacituzumab govitecan-hziy) — was approved by the FDA for the treatment of adult patients with metastatic triple-negative breast cancer who have received at least two prior therapies for metastatic disease. Trodelvy follows remdesivir in our list of FDA approved drugs in 2020 with the highest sales potential. The current forecast for Trodelvy sales is US$ 2.151 billion by 2026.
FDA’s approval of Lundbeck’s Vyepti (eptinezumab) and Biohaven’s Nurtec ODT (rimegepant) for migraine
headaches brought additional CGRP-targeted products to the market. It will be
interesting to see how Nurtec ODT is accepted given it is a small molecule
drug, which makes administration easier. It was
recently promoted on social media by Khloe Kardashian.
View New Drug Approvals by June 2020 with Estimated Sales (Free Excel Available)
Covid-19 impacts drug launches
The pandemic has, however, started taking a
toll on drug launches. One of the most anticipated drug approvals of the year, Bristol-Myers Squibb’s multiple sclerosis (MS) treatment — Zeposia (ozanimod) — was approved in both the US and Europe. However, the launch of the drug would
be delayed due to the coronavirus outbreak.
The drug was added to BMS’s portfolio through its US$ 74 billion acquisition of Celgene last year. Its
approval was one of the three conditions set for a potentially higher payout
for Celgene investors.
Analysts have high hopes from ozanimod. Its
average peak sales for 2024 have been predicted to be at US$
1.62 billion by Cortellis, though the Covid-19 pandemic may weigh in there
as well.
View New Drug Approvals by June 2020 with Estimated Sales (Free Excel Available)
First non-statin cholesterol drug bags
FDA approval
This year also witnessed the first non-statin
treatment to be cleared for sale in the US in nearly 20 years. The drug, bempedoic acid, is made by Esperion Therapeutics Inc. This cholesterol-lowering drug is aimed at
helping millions of people who can’t tolerate or don’t get enough help from widely used statin pills like Lipitor and Crestor.
This new drug is to be used as an add-on treatment with statins. It lowers bad
cholesterol or low-density lipoprotein (LDL) by inhibiting its synthesis in the
liver. It targets patients with high cardiovascular risk.
Esperion also won approval of bempedoic acid
in combination with ezetimibe, another
cholesterol-lowering drug.
In January last year, Daiichi Sankyo Europe had entered into an exclusive licensing agreement with Esperion Therapeutics for Daiichi Sankyo Europe to market
bempedoic acid and bempedoic acid/ezetimibe combination tablet in the European
Economic Area and Switzerland.
View New Drug Approvals by June 2020 with Estimated Sales (Free Excel Available)
Approvals not granted to almost 20 drugs
There were setbacks too, and not everything
rolled smoothly. This year, almost 20 drug approvals were not granted. Among
the major setbacks were Bristol Myers Squibb and bluebird bio, Inc announcing
that they have received a Refusal to File letter from the FDA regarding the Biologics License
Application (BLA) for their CAR-T therapy, idecabtagene vicleucel (ide-cel), for patients with heavily pre-treated
relapsed and refractory multiple myeloma, which was
submitted in March 2020.
Upon preliminary
review, the FDA determined that the Chemistry, Manufacturing and Control (CMC)
module of the BLA requires further detail to complete the review. No additional
clinical or non-clinical data have been requested or are required.
Two years after Intarcia Therapeutics received
a CRL for its matchstick-sized, long-term drug
implant for type 2 diabetes, the FDA issued a second CRL to the company for its ITCA-650 implant. The
implant is designed to be a small, osmotic pump which can be slipped under the
skin and deliver a continuous, six-month dose of the
GLP-1 agonist exenatide.
The FDA also did not approve Intercept Pharmaceuticals’ obeticholic acid to treat NASH (or
nonalcoholic steatohepatitis, a liver condition in
which the buildup of fat
progressively scars the organ), as it wasn’t convinced that its benefits outweighed the potential risks.
After acquiring Allergan for US$ 63
billion, one of the first drugs which AbbVie was expecting
approval for was Abicipar pegol, their experimental DARPin therapy for patients with neovascular (wet) age-related macular degeneration (nAMD). However, FDA’s review indicated the rate of intraocular inflammation observed following administration of Abicipar pegol 2mg/0.05 mL
results in an unfavorable benefit-risk ratio in the treatment of wet AMD.
View New Drug Approvals by June 2020 with Estimated Sales (Free Excel Available)
Our view
While everyone’s attention is on the Covid-19 pandemic, the industry is certainly busy working towards getting new drugs to market. At the halfway mark, the FDA and EMA seem to be on track to set approval records this year, since the number of drugs approved by June are almost twice the number that were approved at the same time last year.
However, it remains to be seen how companies adapt their sales and marketing strategies in a world where mobility is likely to get restricted and interpersonal contact is set to reduce dramatically.
View New Drug Approvals by June 2020 with Estimated Sales (Free Excel Available)
Impressions: 69883
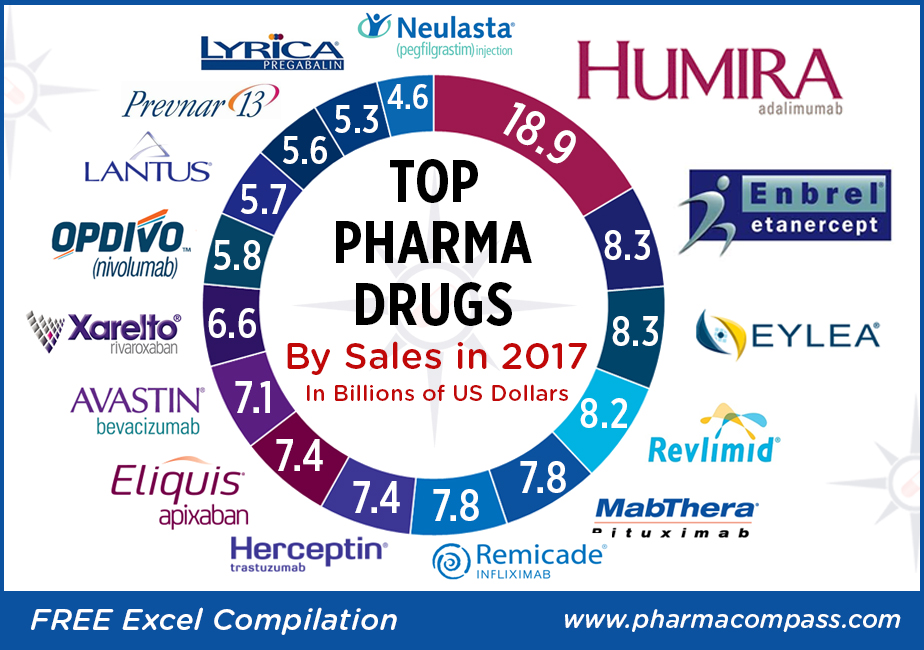
The year 2017 was a landmark year for pharmaceutical
industries in the US and Europe, with a sharp increase in the number of new molecular entities (NMEs) being approved in both geographies.
The US Food
and Drug Administration (USFDA) approved 46 NMEs in 2017, the second highest
since 1996 when 53 NMEs were approved. In Europe, the European Medicines Agency
(EMA) approved 35 drugs with a new active substance, up from 27 in 2016.
Sales for most major pharmaceutical
companies continued to grow in 2017. Earnings forecasts for 2018 have been raised due to the recent US tax reform that has
generated investor hopes for accelerated dividend growth and share buyback
plans.
This week, PharmaCompass brings
you a compilation of the top drugs of 2017 by sales revenue.
Click here to Access All the 2017 Data (Excel
version available) for FREE!
Top-sellers: Humira races ahead, despite launch of biosimilars; Enbrel a distant second
There wasn’t any upheaval
at the top of the pharma drug sales charts. AbbVie’s anti-TNF (tumor necrosis factor) giant
Humira (adalimumab), which is approved to treat
psoriasis and rheumatoid arthritis, added
almost another US $3 billion to its 2016 sales and posted nearly US $19 billion in revenues.
Last year, AbbVie’s raised expectations for Humira’s earnings to reach US $21 billion in global sales by 2020. The
company believes this drug will continue to be a significant cash contributor
until 2025 and the US $21 billion sales forecast
by 2020 is about US $3 billion higher than its expectation two years ago.
In 2016, the US Food and Drug Administration
(FDA) approved Amgen’s Amjevita (adalimumab-atto) — a biosimilar of Humira. And in 2017, another Humira biosimilar — Boehringer Ingelheim’s Cyltezo
(adalimumab-adbm) — received approval from the FDA and European authorities.
Click here to Access All the 2017 Data (Excel
version available) for FREE!
Enbrel (etanercept),
the longest-used biologic medicine for the treatment of rheumatism around the
world, was the second best-selling drug with US $8.262 billion in 2017 sales.
The sales of the drug were down from US $9.366 billion in
2016 owing to lower selling prices and increased
competition, which in turn hurt demand.
Since it was first approved in the United States in 1998,
Enbrel has been approved in over 100 countries and the drug is promoted by Amgen,
Pfizer
and Takeda
in different geographies.
Novartis’ biosimilar copy of Enbrel, which got approved by the FDA in August
2016 for the treatment of patients with
rheumatoid arthritis (RA), plaque psoriasis, ankylosing spondylitis (AS) and
other diseases is still not on the market because of a patent-protection
challenge from Amgen.
Amgen is arguing in the US federal court
that its drug has patent protection until 2029.
Click here to Access All the 2017 Data (Excel
version available) for FREE!
Fast-growing drugs: Eylea and Revlimid bring
fortunes for Regeneron and Celgene
Regeneron’s
flagship eye treatment, Eylea (aflibercept) which is marketed by Bayer outside the United States, added another US $1 billion in
annual sales last year to record US $8.260 billion in total sales. Eylea net
sales grew 11 percent year-on-year in the US and 19 percent year-over-year
outside the US.
The company believes much of the recent
growth in the US was driven by demographic trends with an aging population as
well as an overall increase in the prevalence of diabetes.
These demographic trends are expected to
continue in the coming years, providing an opportunity for continued growth.
Eylea sales alone contribute 63 percent to Regeneron’s total sales.
Click here to Access All the 2017 Data (Excel
version available) for FREE!
Celgene’s
Revlimid
(lenalidomide)
— a thalidomide derivative introduced in 2004 as an immunomodulatory agent for the treatment of various cancers such as multiple myeloma — brought in an additional US $1.2 billion in 2017 sales and had total revenues of US $8.187 billion.
Revlimid continues to contribute more than 60 percent to the company’s total sales of US $13 billion.
Celgene received a setback this month as the
USFDA refused to consider Celgene’s
application for ozanimod, an experimental
treatment for relapsing multiple sclerosis. The treatment was being seen as a
key to the company’s fortunes as Celgene had
said ozanimod is worth US $4 billion to
US $6
billion a year in peak sales.
Click here to Access All the 2017 Data (Excel
version available) for FREE!
Gilead’s Hepatitis C franchise enters free fall
Gilead Sciences’ blockbuster hepatitis C drugs franchise that includes Sovaldi and Harvoni continue to feel the
competitive heat as they registered US $9.137
billion in 2017 sales, down from US $14.834
billion the previous year.
While reporting 2017 results, Gilead provided guidance for
2018 and said its sales of Hepatitis C drugs could fall
further to US $3.5 billion - US $4 billion. At their peak in 2015, Gilead’s Sovaldi and Harvoni had together generated
US $19.1 billion in sales.
One of the major reasons for this drop is AbbVie’s launch of its new treatment Mavyret
at a deep price discount to the competition. AbbVie
also claims to have the shortest treatment course at eight weeks, compared with
12 weeks or longer for other treatments.
AbbVie reported US $1.274 billion in Hepatitis C drug sales
in 2017, down from US $1.522 billion in 2016.
Click here to Access All the 2017 Data (Excel
version available) for FREE!
Novartis’ Gleevec, Merck’s cardiovascular drugs, GSK’s Advair face generic heat
Novartis’ Gleevec (imatinib), which had at one point become the best-selling drug for Novartis and had brought in US $3.323 billion for the company in 2016, started facing generic competition last year and the anti-cancer drug lost US $1.380 billion in sales to bring in ‘only’ US $1.943 billion last year.
The US patents of Merck’s cardiovascular drugs — Zetia (Ezetimibe)
and Vytorin (Ezetimibe
and Simvastatin) — expired in April 2017. In May 2010, Merck and Glenmark
Pharmaceuticals entered into an agreement that allowed Glenmark to launch
a generic version of Zetia in late 2016. The drugs
that had combined sales of US $3.701
billion in 2016 felt the generic heat in 2017 and the sales were US
$1.606 billion lower at US $2.095
billion.
Click here to Access All the 2017 Data (Excel
version available) for FREE!
GSK’s Advair, which was expected
to encounter generic competition in 2017, continued to breathe easy as the FDA
found deficiencies in the applications of Hikma, Mylan and Sandoz.
All three failed to get the FDA nod for their generic versions of Advair, a drug used in the management of asthma and chronic obstructive pulmonary disease that generated sales worth US $4.431 billion (£3.130 billion) in 2017.
Top 15 drugs by sales
Here is PharmaCompass’ compilation
of the best-selling drugs of 2017. This is based on information extracted from
annual reports and US Securities and Exchange Commission (SEC) filings of major
pharmaceutical companies.
If you would like your own copy of all the information we’ve collected, email us at support@pharmacompass.com and we’ll send you an Excel version.
Click here to access all the 2017 data (Excel
version available) for FREE!
S. No.
Company / Companies
Product Name
Active Ingredient
Main Therapeutic Indication
2017 Revenue in Millions (USD)
1
AbbVie Inc., Eisai
Humira®
Adalimumab
Immunology (Organ Transplant, Arthritis etc.)
18,946
2
Amgen, Pfizer Inc., Takeda
Enbrel®
Etanercept
Immunology (Organ Transplant, Arthritis etc.)
8,262
3
Regeneron, Bayer
Eylea
Aflibercept
Ophthalmology
8,260
4
Celgene
Revlimid
Lenalidomide
Oncology
8,187
5
Roche
MabThera®/Rituxan®
Rituximab
Oncology
7,831
6
Johnson & Johnson, Merck, Mitsubishi Tanabe
Remicade®
Infliximab
Autoimmune Disorders
7,784
7
Roche
Herceptin®
Trastuzumab
Oncology
7,435
8
Bristol-Myers Squibb, Pfizer Inc.
Eliquis®
Apixaban
Cardiovascular Diseases
7,395
9
Roche
Avastin®
Bevacizumab
Oncology
7,089
10
Bayer, Johnson & Johnson
XareltoTM
Rivaroxaban
Cardiovascular Diseases
6,590
11
Bristol Myers Squibb, Ono Pharmaceutical
Opdivo
Nivolumab
Oncology
5,815
12
Sanofi
Lantus
Insulin Glargine
Diabetes
5,731
13
Pfizer Inc.
Prevnar 13/Prevenar 13
Pneumococcal 7-Valent Conjugate
Anti-bacterial
5,601
14
Pfizer Inc., Eisai
Lyrica
Pregabalin
Neurological/Mental Disorders
5,318
15
Amgen, Kyowa Hakko Kirin
Neulasta®
Pegfilgrastim
Blood Disorders
4,553
Sign up, stay ahead
In order to stay informed, and receive
industry updates along with our data compilations, do sign up for the PharmaCompass Newsletter and
you will receive updated information as it becomes available along with a lot
more industry analysis.
Click here to Access All
the 2017 Data (Excel version available) for FREE!
Impressions: 58416