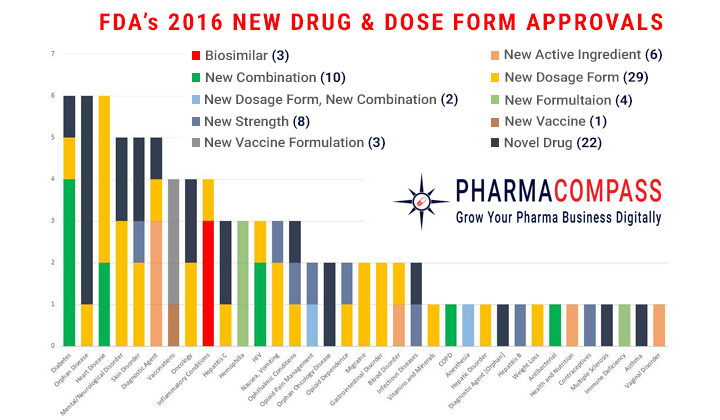
After 2 years of sky-high approval numbers, the US Food and Drug Administration’s (FDA) Center for Drug Evaluation and Research approved 22 novel drugs in 2016, down from the 19-year high of 45 in 2015. The FDA also approved many new dose forms, formulations, combination products and vaccines.
This week, PharmaCompass, shares its analysis of the new drug approvals by the FDA in 2016.
Reasons behind the low approvals in 2016
Of the 22 novel drugs approved by the FDA, the FDA approved 9 products with orphan designation, in line with the industry’s recent focus on rare diseases. However, as the industry shifts its focus towards biotechnology, only 7 of the novel products approved were biologic applications.
The 9 orphan designees approved (41% of all new drug approvals) were significantly lower than the 21 (47%) orphan designees approved in 2015 and 17 (41%) in 2014. FDA’s approval of 4 (18%) oncology drugs in 2016, was also down from the 14 (31%) approvals in 2015, 9 (22%) in 2014 and 9 (33%) in 2013.
The reasons for the low 2016 approval count, have been attributed to the agency approving five drugs in 2015 that actually had approval action dates in 2016 and an increase in the number of drugs that the agency rejected.
A key reason for the rejections was the sponsors’ failure to comply with good manufacturing practice regulations.
Click here to access our list of all New Drug Approvals (Excel version available) for FREE!
Gilead’s Epclusa, the first all-oral, pan-genotypic, single tablet regimen for the treatment of adults with chronic hepatitis C virus (HCV) infection, is expected to become the most profitable approval of 2016 with expected sales of $ 8.4 billion by 2022.
2016 also saw the FDA’s contentious approval of Exondys 51™ (eteplirsen) under its accelerated approval process. Exondys 51™ treats Duchenne muscular dystrophy (DMD), a rare genetic disease which causes progressive muscle wasting that affects around 20,000 boys and young men in the United States.
Approvals of three biosimilars which
target $ 33 billion in brand drug sales
In a giant leap for the generic pharmaceutical industry, three biosimilar applications were approved in 2016 that target brand drugs which generated more than $ 33 billion in sales last year.
All the three biosimilars approved were tumor-necrosis factor alpha (TNF α) inhibitors used to manage inflammatory conditions.
Pfizer and Celltrion’s Inflectra, a biosimilar to Janssen’s Remicade® (2016 sales - $ 8.8 billion), was the first to get approval for all Remicade-approved indications, except pediatric ulcerative colitis. Inflectra was launched at risk in November 2016.
After Inflectra’s approval, the FDA approved Sandoz’s Erelzi, a biosimilar to Enbrel® (etanercept – Amgen, 2016 sales - $ 9.1 billion).
On Sept. 23, 2016, Amgen’s Amjevita™ (adalimumab – atto), a biosimilar of the world’s best-selling drug by revenue, AbbVie’s Humira®(2016 sales - $ 16.4 billion), was also approved for treating adults with a variety of medical conditions ranging from rheumatoid arthritis, plaque psoriasis, to ulcerative colitis.
Click here to access our list of all New Drug Approvals (Excel version available) for FREE!
Kim Kardashian’s pregnancy drug is now available in a new strength
Diclegis, a combination of an antihistamine (doxylamine succinate) and a form of vitamin B6 (pyridoxine HCl), made headlines when the FDA issued a warning letter for the social media promotion of the drug by Kim Kardashain, star of the reality show ‘Keeping Up With The Kardashians’
Duchesnay, the company which produces Diclegis, got approval for Bonjesta which combines twice the amount of doxylamine and pyridoxine when compared with Diclegis which contains 10mg of each ingredient.
Indian generic companies get approvals for
differentiated products
As the generic industry in the U.S. continues to face severe pricing pressure, Indian generic companies are attempting to overcome these challenges by trying to move up the value chain by supplying differentiated generic products.
Consistent with this strategy, the applications of Sun Pharmaceuticals for a new ophthalmic version of Bromfenac (Bromsite) and Dr. Reddy’s Laboratory for an injectable version of Sumatriptan (Zembrace™ SymTouch) to treat migraines were also approved.
Click here to access our list of all New Drug Approvals (Excel version available) for FREE!
Pfizer launches an opioid treatment with
abuse-deterrent properties
The abuse of opioids, including prescription painkillers and drugs like heroin, is something the United States has struggled with since before the 1900s. Last year, the FDA announced that immediate-release opioid painkillers such as oxycodone and fentanyl will have to carry a "black box" warning about the risk of abuse, addiction, overdose and death.
Pfizer got approval last year for Troxyca ER a combination containing oxycodone and naltrexone. Troxyca ER has properties that are expected to reduce abuse when crushed and administered by the oral and intranasal routes.
In it, the oxycodone releases slowly over several hours. If the capsules are crushed, the encased naltrexone mixes with oxycodone, essentially cancelling any euphoric effects.
Pfizer also received approval for an extended release form of Tofacitinib (Xeljanz® XR) to treat adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response or intolerance to methotrexate.
Click here to access our list of all New Drug Approvals (Excel version available) for FREE!
A dietary supplement gets approved along with new forms of commonly used drugs
In 2016, Endoceutics got Intarosa, a once-daily vaginal insert, approved which is the first FDA approved product to contain prasterone, also known as dehydroepiandrosterone (DHEA).
Although DHEA is included in some dietary supplements, the efficacy and safety of those products have not been established for diagnosing, curing, mitigating, treating or preventing any disease.
Lisinopril, the most commonly used drug by U.S. Medicare patients, was approved as an Oral Solution for the first time.
Diabetes treatments, one of the most commonly prescribed drug categories in the U.S., saw the addition of Sanofi and Novo Nordisk’s fixed-dose, long-acting insulin and glucagon-like peptide 1 (GLP-1) agonist combinations to the list of options available to treat adult type-2 diabetes.
Our View
The approval of new drugs ensures that the world has access to improved healthcare solutions and breakthrough medical therapies.
With Donald Trump “focused on accelerating the FDA” and an on-going argument that drugs should not have to be proven effective before getting approved, this year looks like the one where the new drug approval scenario will be shaken up for time to come.
Click here to access our list of all New Drug Approvals (Excel version available) for FREE!
The PharmaCompass Newsletter – Sign Up, Stay Ahead
Feedback, help us to improve. Click here
“ The article is based on the information available in public and which the author believes to be true. The author is not disseminating any information, which the author believes or knows, is confidential or in conflict with the privacy of any person. The views expressed or information supplied through this article is mere opinion and observation of the author. The author does not intend to defame, insult or, cause loss or damage to anyone, in any manner, through this article.”





