Nearly every
year, drugmakers ring in the new year with drug price increases in the US. This
year too, prices of over 450 prescription
medicines increased by an average of around 5 percent at the start of January.
This, when high drug prices have been one of the biggest political issues in
the US over the last few years.
PharmaCompass decided to usher in 2022 with a review of the US Medicare Part D Prescription Drug data recently released by the Centers for Medicare and Medicaid Services (CMS) for calendar year 2019. Using the available data, we have developed our own dashboard to show recent trends in consumption of prescription drugs. With this analysis, we hope our readers will get a better understanding of the world’s largest market for pharmaceuticals, as also a fix on where it may be headed.
View US Medicare Part D 2019 Drug Spending (Free Excel Available)
Rising healthcare, drug spends in US
Over the
last several years, we have repeatedly heard political leaders in the US
complain about high drug prices. Yet, drug prices and healthcare spends have
risen unabated.
America’s National Health Expenditure Accounts (NHEA) includes annual expenditures on healthcare goods and services, public health activities, the net cost of health insurance, and investment related to healthcare. In 2019, America’s national health expenditure (NHE) grew by 4.6 percent to US$ 3.8
trillion, accounting for 17.7 percent of the gross domestic product (GDP).
During the year, prescription drug spend increased by 5.7
percent to US$ 369.7 billion. In comparison, Medicare spend grew 6.7
percent to US$ 799.4 billion.
President
Joe Biden recently stressed on the need to cap the prices of essential drugs,
and said that the average American pays the highest prices for prescription
drugs anywhere in the world. Americans pay 10 times as much as other countries for life-saving insulin — the top selling prescription drug covered by the Part D program.
Pharma
companies, on the other hand, have vehemently argued against any price cuts in
the US, saying price cuts would hinder drug research and development for all
diseases.
View US Medicare Part D 2019 Drug Spending (Free Excel Available)
Patented drugs account for 80.3 percent of total Part D spend
Medicare is the US federal government’s program that
provides health insurance to most people who are 65 years
or older. Medicare’s Part D plan provides outpatient drug coverage through private
insurance companies that have contracts with the federal government. Eligible
people have to choose and enroll in a private prescription drug plan for Part D
coverage. Medicare Part B, on the other hand, covers a wide variety of
medically necessary outpatient services and some preventative services.
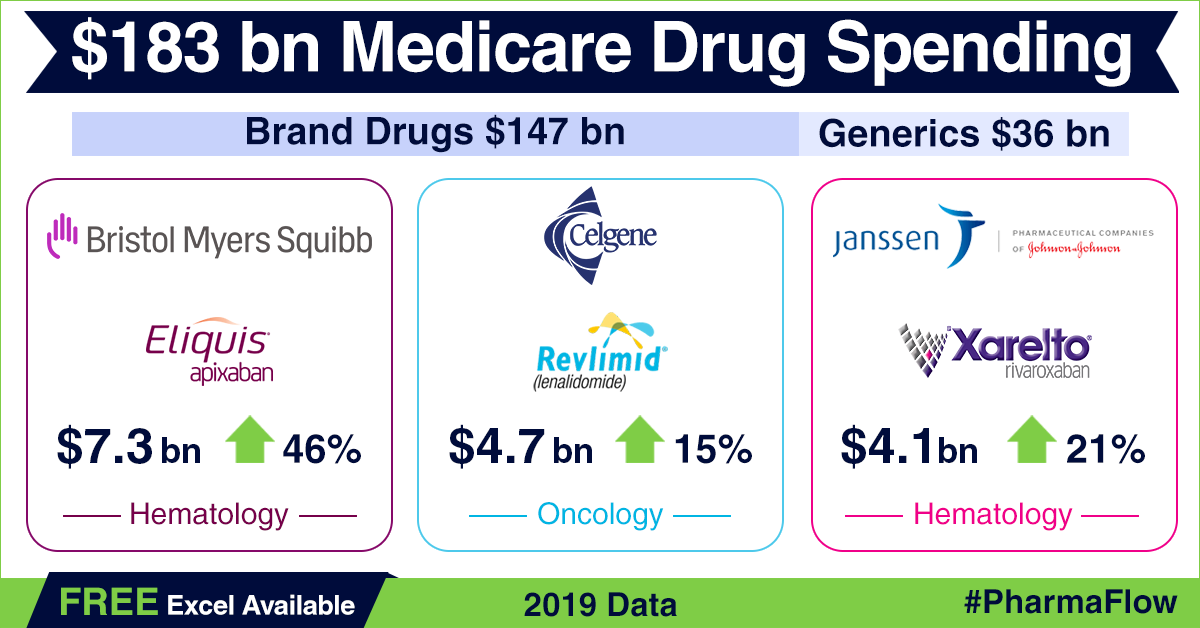
Prescription
drug coverage under Part D reached US$ 183 billion in 2019 — a growth of around 9 percent over 2018, when spending was US$ 168 billion. Spending
on patented drugs in 2019 accounted for around US$ 147 billion or 80.3 percent
of the total spend for the year. Generic drugs made up for the remaining 19.7
percent (approximately US$ 36 billion). In 2018, generic drugs worth US$ 35.8
billion were sold under Part D, accounting for 21 percent of the total spend
under the program.
View US Medicare Part D 2019 Drug Spending (Free Excel Available)
Eliquis ranks highest on Medicare’s brand drug spend
Under Part
D, endocrinology and oncology were the two therapeutic areas that generated
maximum revenue for pharma companies, driving home sales of over US$ 31.8
billion and US$ 23.5 billion, respectively. Neurology drugs generated sales of
around US$ 22.9 billion.
Among branded
drugs, Bristol Myers Squibb’s anticoagulant Eliquis (apixaban) was the most selling drug in 2019 under Part D, notching up about US$ 7.3 billion in sales — a rise of US$ 2.3 billion or 46 percent over 2018.
Celgene’s cancer drug Revlimid (lenalidomide) roped in US$ 4.7 billion (up
by 14.6 percent), while another anticoagulant drug Xarelto (rivaroxaban) by Janssen Pharma — a unit of Johnson & Johnson — fetched US$ 4.1 billion (up 20.6 percent) in sales through Part D. AbbVie’s anti-rheumatic drug Humira and Sanofi’s diabetes drug Lantus saw sales of around US$ 3.7 billion each
under the program.
Amongst
generics, the largest selling drug under Part D (by dosage units) was metformin (diabetes), followed by gabapentin (seizure), PEG3350 with
electrolyte (gastroenterology), metoprolol (hypertension) and atorvastatin (cholesterol). In 2019, the
overall dosage units sold also jumped higher by 2.25 billion units to 111.35
billion.
The sales
ranking of Part D does bare some similarities with the global ranking of
highest selling drugs. In 2020, Humira had retained its position as the highest
selling drug in the world, generating sales of US$ 20.4 billion. Both
Eliquis and Revlimid had retained their ranking as the third and fourth most
selling drugs, bringing home US$ 14.1 billion and US$ 12.1 billion in global
sales in 2020.
View US Medicare Part D 2019 Drug Spending (Free Excel Available)
Medicare’s inability to negotiate prices costs American taxpayers billions of dollars
Over the
years, drug companies have used Medicare’s
inability to negotiate prices under Part D to increase the prices of their
drugs significantly and rip off huge profits, a three-year-long US House
Oversight Committee investigation has revealed.
US taxpayers could have saved over US$ 25 billion in five years if the prices of just seven drugs — Humira, Imbruvica, Sensipar, Enbrel, Lantus, NovoLog and Lyrica — were negotiated by Medicare. Another US$ 16.7 billion could have been saved between
2011 and 2017 on insulin products manufactured by Eli Lilly, Novo Nordisk and Sanofi, which control 90 percent of the insulin market in the US, the committee’s report revealed.
Elsewhere in
the world, the same drugmakers are bending over backwards to get into medical
insurance programs. For instance, China reported that several international
pharma firms, many of them headquartered in the US, slashed the prices of their
drugs by up to 94 percent to get into the country’s national medical insurance coverage.
In the US — which accounted for around 46 percent of the global share of drugs in 2020 — senior citizens may have to pay more for medicines as the government announced a large hike in Medicare premiums for 2022
if an expensive Alzheimer’s drug, Aduhelm, is included in the list.
In order to
ensure inclusion in Medicare, Biogen slashed the price of Aduhelm by half — from US$ 56,000 to US$ 28,200 — just weeks before a crucial meeting called by the CMS. Clearly, this has set a precedent in an industry which is known for rampant price hikes and rarely for any price cuts. This could also be put forth as an example of what Medicare could achieve if it receives negotiation rights.
View US Medicare Part D 2019 Drug Spending (Free Excel Available)
Our view
President
Biden's Build Back Better legislation,
which the House passed last month, is up for vote in the Senate. The
legislation contains provisions that would allow Medicare to negotiate the
prices of some expensive drugs, penalize drugmakers who raise prices faster
than inflation and cap out-of-pocket costs for insulin at US$ 35 per month.
However, chances of the bill being passed in its present form are slim.
Even if the
Senate passes the bill, Medicare would be able to negotiate the prices of only 10 prescription drugs and insulin products in 2025.
The number would increase over the years, reaching 100 in six years, and hence
forth grow by 20 drugs a year.
It seems like 2022 won’t be the last year when January 1 will be braced with price hikes in the US by drugmakers. Looks like they will continue to make hay while the sun shines.
View US Medicare Part D 2019 Drug Spending (Free Excel Available)
Impressions: 2622
This week, PharmaCompass
reviews the recently released data on prescription drugs paid for under the
Medicare Part D Prescription Drug Program in the United States in calendar year
2016.
But first, let’s understand what is Medicare.
Medicare is the federal health insurance program in the US. In 2017, it covered 58.4 million people — 49.5 million aged 65 and older, and 8.9 million disabled.
Prescription drug coverage under this
program was started in 2006, and is known as Medicare Part D.
As part of this
coverage, the Centers for Medicare & Medicaid Services (CMS) contracts insurance
companies and other private companies, known as plan sponsors, that offer
prescription drug plans to their beneficiaries with varying drug coverage and
cost-sharing requirements.
In
2017, the Congressional Budget Office (CBO) had estimated that spending on
Medicare Part D would reach US$ 94 billion, or about 16 percent of all Medicare
expenditures for the year.
Click here to access the compilation of Medicare Part D
Prescriber Summary Report
According
to the CBO, Medicare Part D is the most significant expansion of the Medicare
program since it was created by Congress in 1965.
With
more than 1.48 billion claims from beneficiaries enrolled under the Part D
prescription drug benefit program under its umbrella, our analysis of Medicare
Part D provides valuable insights into how elderly Americans use prescription
drugs.
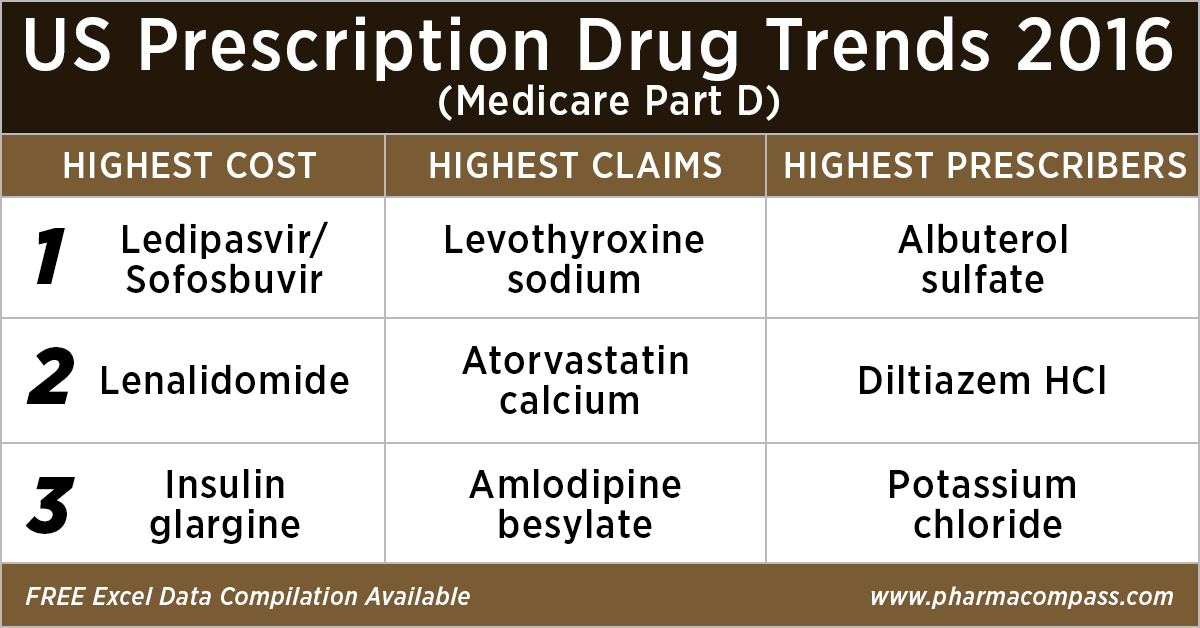
Top 10 drugs by
cost: The ones that bore the highest cost burden for Medicare
As in 2015, in 2016
too Gilead’s Hepatitis C treatment — Ledipasvir/Sofosbuvir (Harvoni) — remained the single drug highest payout under the Medicare Part D Prescription Drug Program with a total cost of US$ 4.4 billion.
As Gilead continued
to face competition from AbbVie and Merck in the Hepatitis C space, the spending on Harvoni was down
37 percent from US$ 7.03 billion in 2015.
Click here to access the compilation of Medicare Part D
Prescriber Summary Report
Celgene’s cancer treatment, Lenalidomide (Revlimid), Sanofi and Merck’s diabetes treatments and AstraZeneca’s Crestor (Rosuvastatin Calcium) for
cholesterol followed Harvoni. All together, they cost the Medicare program over US$ 10 billion.
Generic Name
Number of Medicare Part D Claims
Number of Medicare Beneficiaries
Number of Prescribers
Aggregate Cost Paid for Part D
Claims (In USD)
LEDIPASVIR/ SOFOSBUVIR (HARVONI)
141,665
52,782
12,097
4,398,534,465
LENALIDOMIDE
239,049
35,368
10,382
2,661,106,127
LANTUS SOLOSTAR (INSULIN
GLARGINE, HUM.REC.ANLOG )
5,028,485
1,075,248
245,447
2,526,048,766
SITAGLIPTIN PHOSPHATE
4,742,505
864,442
206,223
2,440,013,513
ROSUVASTATIN CALCIUM
6,012,444
1,560,050
249,981
2,322,724,007
FLUTICASONE/SALMETEROL
5,194,391
1,196,007
275,442
2,319,808,482
PREGABALIN
4,940,115
852,497
267,532
2,098,953,250
RIVAROXABAN
4,403,332
807,820
252,141
1,954,748,890
APIXABAN
4,455,782
826,969
231,631
1,926,107,484
TIOTROPIUM BROMIDE
4,153,162
903,494
235,564
1,818,857,361
Click here to access the compilation of Medicare Part D
Prescriber Summary Report
Top 10 drugs by claims: The most commonly
used drugs of 2016
With 46.6 million claims, the thyroid hormone deficiency treatment — Levothyroxine Sodium — retained its position of being the most claimed product under Medicare’s Part D Prescription Drug Program in 2016.
The number of
Medicare Part D claims includes original prescriptions and refills.
Following Levothyroxine Sodium was the lipid-lowering agent — Atorvastatin Calcium — which had 44.5 million Medicare Part D claims that
were filed by almost 9.4 million beneficiaries.
Generic
Name
Number
of Prescribers
Number
of Medicare Part D Claims
Number
of Medicare Beneficiaries
LEVOTHYROXINE SODIUM
669,999
46,617,109
8,091,785
ATORVASTATIN CALCIUM
494,973
44,595,686
9,435,633
AMLODIPINE BESYLATE
497,017
39,913,468
7,802,905
LISINOPRIL
490,452
39,469,840
8,009,954
OMEPRAZOLE
492,951
32,909,236
7,001,160
METFORMIN HCL
611,700
31,007,932
6,394,014
SIMVASTATIN
380,560
29,687,947
6,201,911
HYDROCODONE/ACETAMINOPHEN
660,617
28,595,150
7,265,882
FUROSEMIDE
488,352
27,878,243
5,421,598
GABAPENTIN
555,997
27,627,466
5,363,382
Click here
to access the compilation of Medicare Part D Prescriber Summary Report
Top 10 drugs by prescribers: Medicines that were most popular with
doctors
Among the prescribers, albuterol sulfate (salbutamol) and Diltiazem had
over 900,000 unique providers (or
doctors) prescribing the drug.
Albuterol (salbutamol) is
used to provide quick relief from wheezing and shortness
of breath while Diltiazem is used to prevent chest
pain (angina).
Also
on the list of popular drugs with prescribers is Hydrocodone-Acetaminophen.
With more doctors prescribing Hydrocodone-Acetaminophen (an
opioid) than commonly used antibiotics, such as Cephalexin, Ciprofloxacin and Amoxicillin, the
series of new FDA initiatives to combat the epidemic of opioid misuse and abuse
should change the position of opioids in the top 10 drugs by prescribers in the
coming years.
Click here to access the compilation of Medicare Part D
Prescriber Summary Report
Generic
Name
Number of
Prescribers
Number of
Medicare Part D Claims
Number of
Medicare Beneficiaries
ALBUTEROL SULFATE
985,427
13,100,354
5,417,718
DILTIAZEM HCL
931,159
8,142,004
1,982,550
POTASSIUM CHLORIDE
879,491
18,945,969
4,278,000
PEN NEEDLE, DIABETIC
677,210
5,281,778
1,795,046
LEVOTHYROXINE SODIUM
669,999
46,617,109
8,091,785
HYDROCODONE/ACETAMINOPHEN
660,617
28,595,150
7,265,882
METFORMIN HCL
611,700
31,007,932
6,394,014
CEPHALEXIN
597,647
5,603,879
3,933,373
CIPROFLOXACIN HCL
594,129
7,000,081
4,851,657
AZITHROMYCIN
591,028
7,958,625
5,734,122
What does the
future hold?
Although the Part D Prescriber PUF (public use file) has a wealth of information on payment and utilization for Medicare Part D prescriptions, the dataset has a number of limitations. Of particular importance is the fact that the data may not be representative of a physician’s entire practice or all of Medicare as it only includes information on beneficiaries enrolled in the Medicare Part D prescription drug program (i.e., approximately two-thirds of all Medicare beneficiaries).
Click here to access the compilation of Medicare Part D
Prescriber Summary Report
Last
month, the Office of the Inspector General (OIG)
reviewed
the Part D claims data for the years 2011 to 2015 for brand-name drugs.
The OIG’s report found that the total reimbursement for all brand-name drugs in Part D increased 77 percent from 2011 to 2015, despite a 17-percent decrease in the number of prescriptions for these drugs.
With soaring drug prices being an issue for
regular debate in the Unites States and President Trump announcing that his
team will use strategies to strengthen the negotiating powers under
Medicare Part D and Part B, it remains to be seen how the data on prescription drugs paid for under
the Medicare Part D Prescription Drug Program will change in the coming years.
Click here to access the compilation of Medicare Part D
Prescriber Summary Report
Impressions: 2500
Divi's Laboratories — an Indian active pharmaceutical ingredient (API) manufacturer and until recently the darling of the Indian bourses — received another setback this week.
The US Food and Drug Administration (FDA) placed its Unit
II, located in Visakhapatnam, on its import alert list for “refusing FDA foreign establishment inspection”
as also for “not meeting drug GMPs” (good manufacturing practices).
According to the FDA website “the refusal to permit inspection of a foreign facility or provide reasonable access to FDA’s inspectional personnel, combined with other evidence, provides an appearance that the firm’s products are manufactured, processed, or packed under insanitary conditions.” The firm is considered to have refused the FDA inspection.
This news comes as a surprise, as it adds an additional dimension to the problems that the FDA encountered at Divi’s facilities.
PharmaCompass
had
previously shared news that Health Canada had placed the company on its inspection tracker after data integrity and general GMP observations were reported by its “regulatory partner(s)”.
According to
media reports, the firm was inspected by the US FDA from November 29 to
December 6, 2016, and the regulators had issued an 18-page long Form 483 with five observations.
Second Divi’s group firm to refuse inspection
The news comes a few months after its affiliate – Divi’s Pharmaceutical Private Ltd was placed on FDA’s Import Alert List for refusing an inspection by the US
FDA.
The two
facilities of Divi’s Pharmaceutical that were placed on the FDA’s Red List were Divi Towers in Hyderabad, and a facility in Medak district, Telangana. Divi Towers is also the corporate headquarters of Divi’s Laboratories.
Divi’s Laboratories’ Unit-II is a major producer of APIs and a key supplier to leading companies around the world. The FDA has excluded some intermediates and the following APIs from the import alerts – Levetiracetam, Gabapentin, Lamotrigine, Capecitabine, Naproxen sodium , Raltegravir potassium and Atovaquone.
The exemptions did not help as far as the stock market was concerned. Divi’s stock price, which has already been under significant pressure after news of the inspection initially broke out in December, tanked another 20 percent on Tuesday.
Our view
While the details are yet to emerge on the exact nature of problems which the US FDA inspectors uncovered and encountered at Divi’s Laboratories Unit II, it is clear that Divi’s has a long road ahead insofar as regulatory compliance is concerned.
Impressions: 3525
This week, Phispers highlights more bad news for Israeli drugmaker Teva, along with news on the ‘overwhelming efficacy’ of blood thinner Rivaroxaban over Aspirin, Sanofi’s plans to resubmit its application for Sarilumab, Denmark’s entry into the tug of war for hosting the EMA headquarters and are routine round up of global non-compliance concerns.
Teva CEO steps down, as
another bribery probe emerges and discussions of a split start
The chief executive of Teva Pharmaceutical Industries, Erez Vigodman, stepped down after serving for
three years. He has been replaced by Chairman Yitzhak Peterburg for the interim period. Teva is the world's biggest maker of generic
drugs.
In the last five years, he is the third CEO to vacate the position. A sudden change in the company’s leadership came just two months after the resignation of Sigurdur Olafsson, the former head of Teva’s main business unit — generic medicines. Both the executives played an important role in Teva’s US $ 40.5 billion purchase of Actavis Generics last year, touting it as a move that would provide growth.
Instead, the acquisition led to more bad news.
In a short statement after leaving, Vigodman stated: “I believe that now is the right time for me to step down. It has been a privilege to lead Teva, and I am proud of all we have accomplished. I am confident that the company’s future is bright.”
A lot of bad news has already piled up for Teva’s investors since the New Year. This includes the following negative events:
The market is criticizing Teva’s acquisition of Actavis (Allergan’s generics division) for US $ 40 billion in cash and shares.
Teva’s acquisition of Mexican company Rimsa proved to be a catastrophe. Rimsa's plants are now shut down.
Apart from the bad decisions over acquisitions,
Teva is also involved in two legal wrangles. One was a case of bribery in
developing countries, in which Teva agreed to pay US $ 519 million to US
authorities after paying bribes to officials in Mexico, Ukraine and Russia to
boost sales. Another legal issue
involves the investigation of Teva over bribe allegations by Israeli
authorities which came up a day after Vigodman stepped down.
A US district court ruling invalidated four patents out of five on its top seller — the multiple sclerosis drug Copaxone. The ruling, issued in late January, may open the door to generic
competition (Novartis and Mylan) for thr drug that generates a fifth of Teva’s US $ 20 billion in annual sales.
In the company’s own words: “New products stemming from that asset (Copaxone) would be unexpectedly delayed, while prices of its copycat medicines are likely to remain under pressure in the US, prompting a cut to its 2017 profit forecast.”
Following the resignation, at the company's earnings call
earlier this week, analysts started asking if Teva would consider a split-up?
FDA issues Warning Letters to
Indian, Japanese & Chinese firms
Sato
Pharmaceutical, a company established in 1939 in Japan, received a warning letter from the FDA as it failed to establish an adequate system for monitoring the conditions of its cleanroom environments. Following the inspection, the firm revised its standard operating procedure related to the “Aseptic Production Area”, however,
the FDA found the response to be deficient.
FDA inspectors also uncovered that the company had not
performed the necessary smoke studies to evaluate air flow characteristics of
its open Restricted Access Barrier System (RABS). The company released sterile
products manufactured on the aseptic processing line, without studies to
demonstrate unidirectional airflow over the exposed sterile product during
processing.
Although, Sato renovated its RABS to use a closed design and
conducted validation studies, the response was found deficient as it does not
address the quality of the products which had already been released
to the U.S. market using the original open RABS design.
An active
pharmaceutical ingredient (API) manufacturer in India, Resonance Laboratories Private Limited also
received a warning letter from the FDA as the inspectors
raised concerns over the facilities water systems and cleaning validation
methods.
The FDA found that the firm’s response to the inspection observations had failed to perform a retrospective review of CGMP deficiencies on the quality of the products which had already been distributed within the United States.
PharmaCompass
had shared the news about the compliance troubles at
Resonance in November, 2016.
The FDA also issued warning letters this week to two Chinese firms who
had been placed on its Import Alert list last year. The warning letters sent to
Ausmetics Daily
Chemicals (Guangzhou) Co., Ltd. and Zhejiang Bangli Medical Products Co., Ltd.
showed that the companies failed to sufficiently test the batches of the final
product they produced and did not adequately confirm the quality of the
incoming active raw materials.
Bayer’s Rivaroxaban shows 'overwhelming efficacy' over aspirin
Back in 1897, a young scientist at a Bayer laboratory in Wuppertal, Germany — Dr. Felix Hoffmann — synthesized a chemically pure and stable form of acetylsalicylic acid (ASA), which
became the active ingredient in Aspirin™.
Since then, Aspirin has been an important medicine due to its
remarkable pain relief, as well as cardiovascular (CV) event prevention
properties. The medicine has truly stood the test of time.
Last week, Bayer AG and its development partner Janssen Research & Development announced the successful outcome of a
large-scale Phase 3 study -- COMPASS, involving 27,402 patients, that assessed the
effect of blood thinner Xarelto (rivaroxaban)
in preventing major adverse cardiac events (MACE).
The trial was scheduled to finish next year but
was stopped early on the advice of an independent Data Monitoring Committee, after the primary endpoint of prevention of MACE — which includes cardiovascular death, myocardial infarction and stroke —reached its pre-specified criteria for superiority over aspirin.
The drug could potentially be used on 30 million
patients with coronary artery disease (CAD) and peripheral artery disease (PAD), in addition to the roughly 25 million patients
it sees in the atrial fibrillation market, says Bayer.
Xarelto is currently the only non-vitamin K
antagonist oral anticoagulant (NOAC) currently under assessment in this
high-risk patient population. The drug is already on the market for reducing the risk of stroke and blood clots.
Sanofi fixes problems
in French plant, to resubmit application for Sarilumab
In 2014 and 2015, while reviewing new drug
applications, the US Food and Drug Administration (FDA) had raised manufacturing questions in only one Complete Response Letter (CRL) sent to the applicant. However, by mid-December, 2016 “an astonishing 40 percent were specifically tied to questions the agency raised about the manufacturing capabilities of a drugmaker or its contractor.”
Manufacturing issues derailed sales forecasts through new drug
approvals of Sanofi, AstraZeneca, Valeant, Bristol-Myers Squibb, Pfizer and many others.
In October 2016, Sanofi received the FDA’s Form 483 for it’s Le Trait facility in France since manufacturing deficiencies were
discovered during a routine good manufacturing practice (cGMP) inspection where
Sarilumab and Dupilumab are manufactured.
This plant is involved in one of the last steps in the manufacturing process of Sarilumab — an investigational interleukin-6 receptor (IL-6R) antibody for the treatment of adult patients with moderate to severely active rheumatoid arthritis (RA) which is a combined program of Sanofi and Regeneron. Due to the manufacturing issues, FDA issued a CRL regarding
the Biologics License Applications (BLA) for Sarilumab.
Sarilumab is said to become a blockbuster after beating the world’s best-selling drug AbbVie's Humira (adalimumab) in a head-to-head trial. Analysts have previously
predicted the drug could bring in more than US $ 1 billion by 2020.
In response to the letter received from the FDA, the French company has filed a comprehensive corrective action plan with the FDA and is “working towards a timely resolution that addresses these concerns.” Once the issues are addressed, both companies said they intend to seek a way to bring the drug to market.
In January 2017, Sanofi and its drug development partner Regeneron Pharmaceuticals said they have resolved manufacturing defects at Le Trait facility, which caused the delay for the approval of Sarilumab drug. Sanofi’s CEO Olivier Brandicourt said: “We worked closely with the US FDA to implement a corrective plan and got positive feedback".
Assuming the formal inspection will also play out
positively, the companies have decided to resubmit their application for
Sarilumab by the end of March.
Denmark
officially bids for relocation of EMA head office
The future location of the European Medicines Agency (EMA) — one of the regulatory jewels of the EU — has been a consistent topic of conversation since the outcome of the Brexit vote.
The intervention of the Japanese government in
early September 2016 brought the EMA issue further into the open when a 15-page letter came up where Japanese officials told their counterparts in the UK that if “the EMA were to transfer to other EU Member States, the appeal of London as an environment for the development of pharmaceuticals would be lost, which could possibly lead to a shift in the flow of R&D funds and personnel to Continental Europe.”
And now, Denmark is also in the list of countries that are bidding for EMA headquarters’ relocation. Copenhagen’s candidacy launch on February 8 comes in the wake of similar launches by
Amsterdam, Milan, Stockholm, Barcelona and Dublin. Only the Czech Republic and
Estonia have ruled themselves out, according to the Financial Times.
Therefore, we may see a 20-way tug of war amongst cities that want to host the EMA.
The Danish Medicines Agency is excited about the Danish government’s decision. Thomas Senderovitz, Director General of the Danish Medicines Agency, said: “The EMA is the most important European coordination forum in the pharmaceutical field, and Copenhagen offers a visionary and innovative life science cluster. Major international pharmaceutical companies have a presence in Copenhagen, and we offer a strong administration and unique culture for collaboration between the health sector and universities in Denmark and southern Sweden”. As the news came out, healthcare giant Novo Nordisk backed and supported the decision of the Danish Government.
India’s Strides plans to spin off API unit
Just two months after Perrigo agreed to sell its entire shareholding in Perrigo API India to Strides for INR 1000 million (US $14.8
million), Strides announced an organizational restructuring plan. As per the
plan, Strides has decided to move away from its business-to-business (B2B) model to a business-to-consumer (B2C) model, which
includes de-merging and listing its APIs business, exiting
probiotics and capping its investment in the biotech business which was also
approved by SeQuent Scientific, which bought into Shasun several years ago.
SeQuent also has a veterinary drug business. Strides Shasun plans to rename itself as Strides Pharma. Post restructuring, the new
Strides Pharma will comprise its retained formulations business having four US
FDA-approved plants in India, Europe and Singapore, and three research and
development (R&D) centers. This business will have a front-end presence in
the regulated markets of Australia, US and the UK and emerging markets of
Africa and India.
Last year, Strides Shasun had mentioned they plan to hive off its commodity focused API manufacturing unit as a separate business. Strides — with two API manufacturing facilities, one in India and one in the UK — is a global supplier of painkiller (Ibuprofen), anti-epileptic medication (Gabapentin) and anti-acidity medication (Ranitidine). Strides had said that it would retain API capacities required for
captive use while setting up a separate company for manufacturing low-margin APIs such as Ibuprofen, Gabapentin and Ranitidine.
Impressions: 5517
In less than three weeks, Donald Trump will assume office as the
President of the United States. He has mentioned that he wants Medicare (a
national social insurance program) to directly negotiate the price it pays for prescription drugs.
Medicare provides health insurance to Americans aged 65 or more, who
have worked and paid into the system through the payroll tax. It also provides
health insurance to younger people with some disabilities or end-stage renal
disease and amyotrophic lateral sclerosis.
In 2015, Medicare provided health insurance to over 55 million Americans — including 46 million people aged 65 or more, and nine million younger people.
As we flag off the New Year, PharmaCompass
provides insights into drug prices and prescription patterns in the US in order
to help professionals make informed decisions. We believe that the cost of
medicines in the US, which have been a subject of much public outcry and
discussions in the recent years, will continue to be scrutinized during 2017.
Medicare data for 2014
Medicare Part D, also known as the Medicare prescription drug benefit — the program which subsidizes the costs of prescription drugs and prescription drug insurance premiums for Medicare beneficiaries — published a data set (for calendar year 2014) which contains information from over one million healthcare providers
who collectively prescribed approximately US $121 billion worth of prescription
drugs paid for under this program.
For each prescriber and drug, the dataset
includes the total number of prescriptions that were dispensed (including
original prescriptions and any refills), and the total drug cost.
The total drug cost includes the ingredient cost of the medication, dispensing fees, sales tax, and any applicable administration fees. It’s based on the amounts paid by the Part D plan, the Medicare beneficiary, other government subsidies, and any other third-party payers (such as employers and liability insurers).
The total drug cost does not reflect any manufacturer rebates paid to Part D plan sponsors through direct and indirect remuneration or point-of sale rebates. In order to protect the beneficiary’s privacy, the Centers for Medicare & Medicaid Services (CMS) did not
include information in cases where 10 or fewer prescriptions were dispensed.
Top
Ten Drugs by Cost, 2014 [Most expensive for Medicare]
Drug Name
Total Claim Count
Beneficiary Count
Prescriber Count
Total Drug Cost
Sofosbuvir
109,543
33,028
7,323
$3,106,589,192
Esomeprazole Magnesium
7,537,736
1,405,570
286,927
$2,660,052,054
Rosuvastatin Calcium
9,072,799
1,752,423
266,499
$2,543,475,142
Aripiprazole
2,963,457
405,048
130,933
$2,526,731,476
Fluticasone/Salmeterol
6,093,354
1,420,515
281,775
$2,276,060,161
Tiotropium Bromide
5,852,258
1,211,919
253,277
$2,158,219,163
Lantus
Solostar
(Insulin Glargine)
4,441,782
972,882
224,710
$2,016,728,436
Sitagliptin Phosphate
4,495,964
789,828
190,741
$1,775,094,282
Lantus
(Insulin Glargine)
4,284,173
787,077
223,502
$1,725,391,907
Lenalidomide
178,373
27,142
9,337
$1,671,610,362
View the Medicare Part D National Prescriber Summary Report, Calendar Year 2014 (Excel version available) for FREE!
Top
Ten Drugs by Average Cost per Claim, 2014 [Most expensive drugs]
Drug Name
Total Claim Count
Beneficiary Count
Prescriber Count
Total Drug Cost
Average Cost Per Claim
Adagen
13
$1,224,835
$94,218
Elaprase
100
$6,560,225
$65,602
Cinryze
1,820
194
196
$96,155,785
$52,833
Carbaglu
60
$2,901,115
$48,352
Naglazyme
129
$6,189,045
$47,977
Berinert
538
73
68
$25,685,311
$47,742
Firazyr
1,568
269
232
$70,948,143
$45,248
H.P. Acthar
9,611
2,932
1,621
$391,189,653
$40,702
Procysbi
314
41
47
$12,542,911
$39,946
Folotyn
15
$598,210
$39,881
Top
Ten Drugs by Claims, 2014 [Most Commonly Used by Patients]
Generic Name
Total Claim Count
Beneficiary Count
Prescriber Count
Total Drug Cost
Lisinopril
38,278,860
7,454,940
464,747
$281,614,340
Levothyroxine Sodium
37,711,869
6,245,507
416,518
$631,855,415
Amlodipine Besylate
36,344,166
6,750,062
451,350
$303,779,661
Simvastatin
34,092,548
6,768,159
387,651
$346,677,118
Hydrocodone-Acetaminophen
33,446,696
8,005,790
677,865
$676,296,988
Omeprazole
33,032,770
6,707,964
475,122
$529,050,385
Atorvastatin Calcium
32,603,055
6,740,061
419,327
$747,635,818
Furosemide
27,133,430
5,176,582
456,047
$135,710,772
Metformin HCl
23,475,787
4,509,978
364,273
$203,948,989
Gabapentin
22,143,641
4,298,609
486,754
$492,557,255
View the Medicare Part D National Prescriber Summary Report, Calendar Year 2014 (Excel version available) for FREE!
Top
Ten Drugs by Prescribers, 2014 [Most Popular with Doctors]
Generic Name
Total Claim Count
Beneficiary Count
Prescriber Count
Total Drug Cost
Hydrocodone/Acetaminophen
33,446,696
8,005,790
677,865
$676,296,988
Ciprofloxacin HCl
7,253,018
4,926,835
568,201
$46,728,353
Amoxicillin
6,298,980
4,384,899
557,614
$31,193,739
Cephalexin
5,040,219
3,529,303
557,048
$36,987,401
Azithromycin
7,339,954
5,274,010
544,625
$70,699,119
Prednisone
11,032,986
4,505,821
536,108
$86,537,932
Tramadol HCl
14,250,227
4,272,724
515,816
$125,343,514
Sulfamethoxazole /Trimethoprim
4,833,758
3,090,944
500,790
$29,231,511
Gabapentin
22,143,641
4,298,609
486,754
$492,557,255
Amoxicillin/Potassium Clav
3,551,452
2,710,244
478,361
$61,713,432
The findings from CMS
data
The CY 2014 data represented a 17 percent
increase compared to the 2013 data set and a substantial part of the total estimated prescription drug spending (as estimated by the Department of Health and Human Services Office of the Assistant Secretary for Planning and Evaluation, or ASPE) in the United States — at about US $ 457 billion in 2015, which was 16.7 percent of the overall personal healthcare services.
Of that US $ 457 billion, US $ 328 billion (71.9 percent) was for retail
drugs and US $ 128 billion (28.1 percent) was for non-retail drugs.
The drug pricing process in the US is complex and
reflects the influence of numerous factors, including manufacturer list prices,
confidential negotiated discounts and rebates, insurance plan benefit designs,
and patient choices.
An IMS study found that across 12 therapy classes widely used in Medicare Part D,
medicine costs to plans and patients in Medicare Part D are 35 percent below
list prices.
View the Medicare Part D National Prescriber Summary Report, Calendar Year 2014 (Excel version available) for FREE!
While the CMS does not
currently have an established formulary, Part D drug coverage excludes drugs
not approved by the US Food and Drug Administration, those prescribed for off-label
use, drugs not available by prescription for
purchase in the US, and drugs for which payments would be available under Parts
A or B of Medicare.
Part D coverage
excludes drugs or classes of drugs excluded from Medicaid coverage,
such as:
Drugs used for anorexia, weight loss, or weight gain
Drugs used to promote fertility
Drugs used for erectile dysfunction
Drugs used for cosmetic purposes (hair growth, etc.)
Drugs used for the symptomatic relief of cough and colds
Prescription vitamins and mineral products, except prenatal vitamins and fluoride preparations
Drugs where the manufacturer requires (as a condition of sale) any associated tests or monitoring services to be purchased exclusively from that manufacturer or its designee
Our view
The Medicare program is designed such that the
federal government is not permitted to negotiate prices of drugs with the drug
companies, as federal agencies do under other programs.
For instance, the Department of Veterans Affairs — which is allowed to negotiate drug prices and establish a formulary — has been estimated to pay (on an average) between 40 to 58 percent less for drugs, as opposed to Medicare Part D.
If Trump administration kick starts direct
negotiations on Medicare drug prices with drug companies, 2017 will surely turn
out to be a year for the pharmaceutical industry to remember.
View the Medicare Part D National Prescriber Summary Report, Calendar Year 2014 (Excel version available) for FREE!
Impressions: 7923
This week, Phispers has lots on generics. While the global leader Teva has more troubles at hand, generic players in the US face fresh lawsuits, and Sanofi plans to sell its European generic unit. There is also talk of Novartis buying America’s generic-drugs maker Amneal. In other news, oncologists find problem with clinical trials, and China shuts plants to curb pollution
Teva braces declining sales, lawsuits
and closure of its Mexico plant
There is more bad news from Israel’s Teva Pharmaceutical Industries. First, its Rimsa plant in Mexico is said to be shut, and a lot of employees have been (reportedly) laid off. As per a news report, it’s difficult to make the Rimsa
plant operational anytime soon.
Teva had invested US $ 2.3 billion in the facility. There are reports that the company may
make a write-down on its investment in Rimsa. In September, the global leader
in generics had claimed that the Espinosa brothers, who had controlled Rimsa
until its sale to Teva, had deceived the regulatory authorities and patients
for years and sold defective and illegal drugs.
Teva’s troubles don’t end there. The company is also setting aside US $ 520 million in its bid to settle allegations of paying bribes in Russia, Mexico and Ukraine. In its latest earnings report released Tuesday, Teva noted that “advanced discussions” are under way with the federal courts in the US to resolve the incidents, which took place between 2007 and 2013.
Teva has
completed 12 acquisitions worth US $ 46 billion in the last four years. Teva’s blockbuster Copaxone,
which brings in 19 percent of its overall sales, has lost several patent
challenges in the US and is likely to face generic competition early next year,
putting more than US $ 4 billion in sales at risk. Even without a generic
competitor, sales declined 2.2 percent year-on-year in the third quarter this
year.
To control
pollution, north China industrial hub curbs drug production
If you live in Delhi, and are coping with the hazardous pollution levels, here’s something that will interest you. A wide-ranging ban has
been imposed in a northern Chinese industrial hub on production at drug plants,
steel mills and other businesses.
This is a
last-ditch attempt by the government of Shijiazhuang city to meet this year’s pollution control target — to reduce the levels of PM 2.5 (fine particles that pose a risk to human health) by 10 percent. Shijiazhuang is the capital of the northern Hebei province, which reported economic growth of 6.8 percent in the first three quarters of this year.
Last week,
the government of Shijiazhuang city said for the remaining 45 days of the year,
it will curb output at thermal power plants, halt all production at industries
such as steel and cement, and limit manufacturing of pharmaceuticals, chemicals
and even furniture.
In 2014,
President Xi Jinping had responded to public outrage over high smog levels. As
a result, local officials are trying hard to strike a balance between pollution
control and economic growth.
Shijiazhuang
is home to major active pharmaceutical ingredient (API) producers such as North China Pharmaceutical, CSPC Zhongnuo Pharmaceutical, CSPC Ouyi Pharmaceutical and many others. These companies are critical to the
global antibiotic supply chain as they provide the building blocks for
antibiotic manufacturing, such as 6-APA and 7-ACA, along with commonly used
antibiotics such as Penicillin, Amoxicillin, Amipicillin and Azithromycin.
PharmaCompass has been routinely
covering the Chinese bulk drug industry and its impact on the environment. In April this year, PharmaCompass
had reported how school children in China were wearing gas masks due to pollution concerns. And prior to that, we had
carried an article on how dependent the world has become on bulk drugs from China.
More trouble
for generic drug-makers in the US as unions file lawsuits
In a fresh
salvo at the generic drug industry, a union representing sergeants of the New
York Police Department is attempting to hit some companies with civil penalties. The generic industry is already facing charges from a
two-year US Justice Department antitrust probe.
The union
has filed two lawsuits against two groups of drug-makers, which includes Novartis AG’s generic drug unit, Ireland-based Perrigo Co., India’s Wockhardt and Taro Pharmaceutical Industries (Israeli subsidiary of India’s Sun Pharma). The union has alleged that the companies colluded to
raise prices of two dermatological creams by as much as 1,000 percent since
2013.
Besides
this, at least four other unions
have filed lawsuits of their own, with two of them adding Actavis Inc., acquired in August by Teva, to the list of
defendants. All the unions manage health benefits for their members. The
unions say they overpaid for the drugs due to the price collusion. They point
to data that the drug-makers took price hikes on certain medicines by nearly
the same amounts within months of each other.
A lawyer for
the New York sergeants’ union said he expects a judge will call a conference in December
to decide if the cases can be combined.
Novartis may
buy generic drug-maker Amneal for US $ 8 billion
Swiss
healthcare major Novartis AG is in talks to acquire American generic-drugs maker Amneal Pharmaceuticals. Through this acquisition, Novartis plans to strengthen
its Sandoz
business. According to Bloomberg, Novartis and Amneal may reach an agreement soon. Amneal
makes the antiviral acyclovir (to treat herpes) and gabapentin (for epilepsy and pain). The
acquisition could cost Novartis around US $ 8 billion. Amneal is a family-owned
business led by co-founders Chintu and Chirag Patel and has operations in North
America, Australia, Europe and Asia. Its portfolio of generic treatments
includes around 115 approved molecules in the US.
Sanofi to
sell off European generic drug unit
French drug
maker Sanofi
confirmed it has decided to sell off its generic drug unit in Europe. The decision will affect two
manufacturing plants in the Czech Republic and Romania.
Sanofi CEO Olivier Brandicourt recently informed investors that the company has “made a definitive decision to initiate a carve-out process and divest the generics portfolio in Europe.” The move is part of the company’s 2020 strategic roadmap. He, however, did not provide details.
Sanofi had
acquired Zentiva, a Czech generic business, in 2008 for US $ 2.6 billion. And Sanofi’s generic business is centered around this acquisition. The business is particularly strong in the Czech Republic, Romania and Turkey.
On Monday, Zentiva Romania informed
the Bucharest Stock Exchange that its majority shareholder Sanofi has decided
to sell its Romanian generic drug plant as part of a major divestment plan of
its EU generic drugs business.
A company spokesperson said the planned scope of the divestment is the generics business “related to Europe,” so it excludes Russia, the Commonwealth of Independent States (CIS) and Turkey. And it includes the two “dedicated manufacturing sites producing and distributing generics for the European market,” one in Prague (Czech Republic), and the other in Bucharest (Romania).
Former
Valeant executives arrested for fraud
Last week,
two former executives of Valeant Pharmaceuticals — Gary Tanner and Andrew Davenport, who had been the CEO of Philidor — were arrested on charges of running a fraud scheme that swindled millions of dollars out of Valeant. The fraud was allegedly conducted with the help of a mail-order pharmacy, that is now defunct.
According to
Preet Bharara, US Attorney for the Southern District of New York, the arrests
were part of an ongoing probe of the scheme.
The criminal
complaint alleges that Tanner and Davenport conspired to enrich themselves with
Valeant funds. The two helped Valeant set up Philidor in early 2013, which was primarily a vehicle to market and distribute
Valeant drugs.
According to
the complaint, Tanner focused on building Philidor’s business, resisted his superiors’ directives to line up other distributors for Valeant’s products and ultimately received a US $10 million kickback from Davenport.
The complaint alleges that in 2014, the two orchestrated Valeant’s agreement to buy an option to purchase Philidor, which cost Valeant at least US $ 133 million. More than US $ 40 million of that went to shell companies controlled by Davenport. One such shell company — called ‘End Game LP — gave a kickback of US $10 million to Tanner.
Homeopathy
products in the US may carry caveats soon
In a report on homeopathic advertising, the Federal Trade Commission (FTC) in the US said that homeopathic drugs should “be held to the same truth-in-advertising standards as other products claiming health benefits.”
Only the US
Food and Drug Administration (FDA) can prevent homeopathic marketers from
selling their products. The FTC has no teeth in the matter.
But very soon, homeopathic products could include statements such as ‘there is no scientific evidence backing homeopathic health claims’ and ‘homeopathic claims are based only on theories from the 1700s that are not accepted by modern medical experts.’
However, this may not affect sales of homeopathic products. There are claims that such statements could backfire because homeopaths and those who believe in homoeopathy don’t trust modern medicine. They could also believe that if
homeopathy has been around for that long, it must work.
This is not the first-time homeopathic medicines would carry caveats. In 1988, the FDA had struck a deal where it agreed that homeopaths could be self-regulating, if they include a disclaimer that their claims haven’t been evaluated by the FDA.
In February
this year, PharmaCompass had carried a news nugget on Professor
Paul Glasziou, a leading academic in evidence-based medicine at Bond
University, who had declared homeopathy as a “therapeutic dead-end”
after a systematic review concluded the controversial treatment was no more
effective than placebo drugs.
Cancer
clinical trials exaggerate benefits of new drugs, say oncologists
Two cancer physicians argue that large clinical trials — required for approval of new cancer drugs in the US — often overstate the effectiveness of the treatment in the real world.
During
cancer clinical trials, some volunteers take the experimental drug, while
others receive standard care with existing drugs. The groups are then compared
to see if their tumors have shrunk, how long it takes for the tumors to return,
and how long do the patients survive. This way, the trial sees whether the
experimental drug is safe and effective and can be sold to patients in the US.
The process
is based on the premise that trials give an accurate indication of safety
and efficacy among cancer patients in general, and not only those who are
eligible for and selected for the trial.
The trouble
is, participants in clinical trials are unlike the overall cancer population,
point out oncologists Dr. Sham Mailankody of Memorial Sloan Kettering Cancer
Center and Dr. Vinay Prasad of the Oregon Health and Science University in JAMA
Oncology. They’re younger, healthier, wealthier, better plugged in to the healthcare system, and better educated.
According to
these oncologists, if cancer patients are similar in age, socio-economic
status, have presence of other (similar) illnesses, and other
characteristics as those in a clinical trial, they might do as well. But for
everyone else, the trial results probably promise more than the drug can
deliver.
Impressions: 4478