
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
DRUG PRODUCT COMPOSITIONS
Regulatory FDF Prices
NA
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
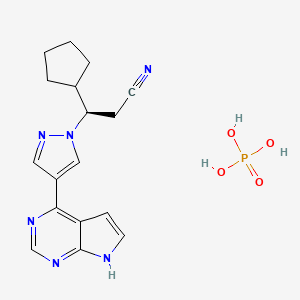

1. 3r)-3-cyclopentyl-3-(4-(7h-pyrrolo(2,3-d)pyrimidin-4-yl)pyrazol-1-yl)propanenitrile
2. Inc-424
3. Inc424
4. Inca24
5. Incb-018424
6. Incb-018424 Phosphate
7. Incb-018424 Salt
8. Incb-18424
9. Incb-18424 Phosphate
10. Incb018424
11. Incb018424 Phosphate
12. Jakafi
13. Jakavi
14. Opzelura
15. Ruxolitinib
16. Ruxolitinib (as Phosphate)
17. Ruxolitinib Monophosphate
1. 1092939-17-7
2. Jakafi
3. (r)-3-(4-(7h-pyrrolo[2,3-d]pyrimidin-4-yl)-1h-pyrazol-1-yl)-3-cyclopentylpropanenitrile Phosphate
4. Jakavi
5. Ruxolitinib (phosphate)
6. Ruxolitinib Monophosphate
7. Incb018424 Phosphate
8. Ruxolitinib (as Phosphate)
9. Incb-018424 Phosphate
10. Incb-018424 Salt
11. Ruxolitinib Phosphate [usan]
12. Incb-18424 Phosphate
13. Incb018424 Salt
14. Chebi:66917
15. 436lru32h5
16. 1092939-17-7 (phosphate)
17. (betar)-beta-cyclopentyl-4-(7h-pyrrolo[2,3-d]pyrimidin-4-yl)-1h-pyrazole-1-propanenitrile Phosphate
18. Incb-18424
19. Ruxolitinib Phosphate Salt
20. (3r)-3-cyclopentyl-3-[4-(7h-pyrrolo[2,3-d]pyrimidin-4-yl)-1h-pyrazol-1-yl]propanenitrile Phosphate
21. (3r)-3-cyclopentyl-3-(4-(7h-pyrrolo(2,3-d)pyrimidin-4-yl)-1h-pyrazol-1-yl)propanenitrile Phosphate (1:1)
22. Phosphenoperoxoic Acid Compound With (r)-3-(4-(7h-pyrrolo[2,3-d]pyrimidin-4-yl)- 1h-pyrazol-1-yl)-3-cyclopentylpropanenitrile And Dihydrogen (1:1:1)
23. Opzelura
24. Unii-436lru32h5
25. (3r)-3-cyclopentyl-3-[4-(7h-pyrrolo[2,3-d]pyrimidin-4-yl)-1h-pyrazol-1-yl]propanenitrilephosphoricacid
26. Jakafi (tn)
27. Jakavi (tn)
28. Ruxolitinib Phosphate(incb018424)
29. Inc 424 Phosphate
30. Incb 018424 Phosphate
31. Incb-424
32. Schembl1369365
33. Chembl1795071
34. Amy5620
35. Dtxsid00911086
36. Ruxolitinib Phosphate (jan/usan)
37. Ruxolitinib Phosphate [mi]
38. Ruxolitinib Phosphate [jan]
39. Ex-a2660
40. Cs1956
41. Mfcd18452860
42. S5243
43. Akos024464417
44. Ruxolitinib (incb-18424) Phosphate
45. Ruxolitinib Phosphate [who-dd]
46. Bcp9000783
47. Ccg-268687
48. Cs-0326
49. 1h-pyrazole-1-propanenitrile, Beta-cyclopentyl-4-(7h-pyrrolo(2,3-d)pyrimidin-4-yl)-,(betar)-, Phosphate (1:1)
50. Ac-30901
51. As-74723
52. Hy-50858
53. Ruxolitinib Phosphate [orange Book]
54. Ruxolitinib (as Phosphate) [ema Epar]
55. D09960
56. J-501793
57. Q27135517
58. (3r)-3-cyclopentyl-3-(4-{7h-pyrrolo[2,3-d]pyrimidin-4-yl}-1h-pyrazol-1-yl)propanenitrile; Phosphoric Acid
59. (3r)-3-cyclopentyl-3-[4-(7h-pyrrolo[2,3-d]pyrimidin-4-yl)pyrazol-1-yl]propanenitrile;phosphoric Acid
60. (r)-3-(4-(7h-pyrrolo[2,3-d]pyrimidin-4-yl)-1h-pyrazol-1-yl)-3-cyclopentylpropanenitrilephosphate
61. 1h-pyrazole-1-propanenitrile,.beta.-cyclopentyl-4-(7h-pyrrolo(2,3-d)pyrimidin-4-yl)-,(.beta.r)-,phosphate (1:1)
62. Phosphoric Acid--3-cyclopentyl-3-[4-(1h-pyrrolo[2,3-d]pyrimidin-4-yl)-1h-pyrazol-1-yl]propanenitrile (1/1)
| Molecular Weight | 404.4 g/mol |
|---|---|
| Molecular Formula | C17H21N6O4P |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 4 |
| Exact Mass | 404.13619017 g/mol |
| Monoisotopic Mass | 404.13619017 g/mol |
| Topological Polar Surface Area | 161 Ų |
| Heavy Atom Count | 28 |
| Formal Charge | 0 |
| Complexity | 503 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 2 | |
|---|---|
| Drug Name | Jakafi |
| PubMed Health | Ruxolitinib (By mouth) |
| Drug Classes | Antineoplastic Agent |
| Active Ingredient | Ruxolitinib phosphate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 5mg base; eq 20mg base; eq 15mg base; eq 10mg base; eq 25mg base |
| Market Status | Prescription |
| Company | Incyte |
| 2 of 2 | |
|---|---|
| Drug Name | Jakafi |
| PubMed Health | Ruxolitinib (By mouth) |
| Drug Classes | Antineoplastic Agent |
| Active Ingredient | Ruxolitinib phosphate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 5mg base; eq 20mg base; eq 15mg base; eq 10mg base; eq 25mg base |
| Market Status | Prescription |
| Company | Incyte |
* Myelofibrosis (MF):
Jakavi is indicated for the treatment of disease related splenomegaly or symptoms in adult patients with primary myelofibrosis (also known as chronic idiopathic myelofibrosis), post polycythaemia vera myelofibrosis or post essential thrombocythaemia myelofibrosis.
* Polycythaemia vera (PV):
Jakavi is indicated for the treatment of adult patients with polycythaemia vera who are resistant to or intolerant of hydroxyurea.
* Graft versus host disease (GvHD):
Jakavi is indicated for the treatment of patients aged 12 years and older with acute graft versus host disease or chronic graft versus host disease who have inadequate response to corticosteroids or other systemic therapies (see section 5. 1).
Treatment of chronic Graft versus Host Disease (cGvHD)
Treatment of acute graft-versus-host disease (aGvHD)
Treatment of vitiligo
L01EJ01
 Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 41012
Submission : 2024-12-31
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39763
Submission : 2024-03-27
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2015-11-16
Pay. Date : 2015-07-31
DMF Number : 29480
Submission : 2015-05-29
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39494
Submission : 2024-03-29
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 38506
Submission : 2023-07-13
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2021-12-16
Pay. Date : 2021-09-29
DMF Number : 32221
Submission : 2017-11-30
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39500
Submission : 2024-03-28
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 38709
Submission : 2023-09-30
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
About the Company : Biophore, founded in 2007, develops and manufactures niche and complex pharmaceutical products. With USFDA- and EU-approved API facilities, a dedicated intermediates site and an R&...
About the Company : Transo-Pharm is a licensed distributor of pharmaceutical components for human and veterinary markets. The company supports the full API life cycle, from development to sales, and a...
 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
About the Company : LGM Pharma is a global leader in sourcing APIs, including hard-to-find drug substances, for pharmaceutical and biotech industries. LGM also operates as a full-service drug product ...
 Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
About the Company : Established in 2004, Metrochem API is one of the fastest-growing APIs, pellets & intermediates manufacturers. It has 6 dedicated manufacturing facilities for its 3 core product gro...
 IKF/Pharmasynthese have been with fine chemicals market and APIs performance for more than 40 years.
IKF/Pharmasynthese have been with fine chemicals market and APIs performance for more than 40 years.
About the Company : Inabata France, established in 1990 and part of the Inabata Group, acquired Pharmasynthèse in 2006. Today, Inabata-Pharmasynthèse offers sourcing, manufacturing, and development ...
 China’s top API & intermediate manufacturer, delivering advanced CMO/CDMO solutions to 60+ countries, empowering global health progress
China’s top API & intermediate manufacturer, delivering advanced CMO/CDMO solutions to 60+ countries, empowering global health progress
About the Company : Shandong Chenghui Shuangda Pharmaceutical Co., Ltd. specializes in API and advanced intermediate R&D and manufacturing, delivering comprehensive CDMO/CRO/OEM services and Finished ...
 Aarti Pharmalabs is a partner of choice for pharmaceutical companies for APIs & Intermediates. Largest Indian producer of Caffeine.
Aarti Pharmalabs is a partner of choice for pharmaceutical companies for APIs & Intermediates. Largest Indian producer of Caffeine.
About the Company : Aarti Pharmalabs is generic APIs & Intermediates manufacturing company & small molecule drug substance CDMO and the largest Indian manufacturer of Xanthine Derivatives (Caffeine, e...
 Tagoor's product development expertise, backed by our comprehensive understanding of the processes, helps us offer high-quality APIs.
Tagoor's product development expertise, backed by our comprehensive understanding of the processes, helps us offer high-quality APIs.
About the Company : Tagoor Laboratories, established in 2018 and part of the Tagoor Group, provides APIs, advanced intermediates, and key starting materials for critical and high-growth therapeutic ar...
 Chunghwa provides cost-effective APIs & advanced intermediates with complete DMF or COS, ensuring quality & reliable production.
Chunghwa provides cost-effective APIs & advanced intermediates with complete DMF or COS, ensuring quality & reliable production.
About the Company : Chunghwa Chemical Synthesis & Biotech Co. Ltd. (CCSB) offers cost-effective APIs with speed, supported by a skilled team. As the first Southeast Asian company to receive FDA approv...
About the Company : Sichuan Qingmu Pharmaceutical is an innovation-driven company specializing in generic APIs, advanced intermediates, and CDMO/CMO services for small-molecule drugs. Approved by US F...
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Results are from an exploratory analysis of ABT-263 (navitoclax) plus ruxolitinib from Cohort 3 of the Phase 2 REFINE study in JAK inhibitor naïve myelofibrosis patients.
Lead Product(s): Navitoclax,Ruxolitinib Phosphate
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: Phase IIIProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable June 10, 2022
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Navitoclax,Ruxolitinib Phosphate
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
AbbVie Presents Investigational Navitoclax Preliminary Data in JAK Inhibitor Naïve Myelofibrosis ...
Details : Results are from an exploratory analysis of ABT-263 (navitoclax) plus ruxolitinib from Cohort 3 of the Phase 2 REFINE study in JAK inhibitor naïve myelofibrosis patients.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
June 10, 2022
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Navitoclax is an investigational, oral BCL-XL/BCL-2 inhibitor. The BCL-2 family of proteins are known regulators of the apoptosis pathway. Its safety and efficacy are under evaluation as part of ongoing Phase 2 and registrational Phase 3 studies.
Lead Product(s): Navitoclax,Ruxolitinib Phosphate
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: Phase IIIProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable April 12, 2022
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Navitoclax,Ruxolitinib Phosphate
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Navitoclax is an investigational, oral BCL-XL/BCL-2 inhibitor. The BCL-2 family of proteins are known regulators of the apoptosis pathway. Its safety and efficacy are under evaluation as part of ongoing Phase 2 and registrational Phase 3 studies.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
April 12, 2022
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Navitoclax is a Other Small Molecule drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Primary Myelofibrosis.
Lead Product(s): Navitoclax,Ruxolitinib Phosphate
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: Phase IIIProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable July 15, 2020
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Navitoclax,Ruxolitinib Phosphate
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Navitoclax is a Other Small Molecule drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Primary Myelofibrosis.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
July 15, 2020
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Navitoclax is a Other Small Molecule drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Primary Myelofibrosis.
Lead Product(s): Navitoclax,Ruxolitinib Phosphate
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: Phase IIIProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable July 13, 2020
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Navitoclax,Ruxolitinib Phosphate
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Navitoclax is a Other Small Molecule drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Primary Myelofibrosis.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
July 13, 2020
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Ruxolitinib is a Other Small Molecule drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of Leukemia, Myeloid, Acute.
Lead Product(s): Ruxolitinib Phosphate,Venetoclax
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: Phase IProduct Type: Miscellaneous
Sponsor: Brian Druker
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable March 14, 2019
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Ruxolitinib Phosphate,Venetoclax
Therapeutic Area : Oncology
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Brian Druker
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Ruxolitinib is a Other Small Molecule drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of Leukemia, Myeloid, Acute.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
March 14, 2019
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Ruxolitinib is a Other Small Molecule drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Primary Myelofibrosis.
Lead Product(s): Ruxolitinib Phosphate,Navitoclax
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: Phase IIProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable July 19, 2017
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Ruxolitinib Phosphate,Navitoclax
Therapeutic Area : Oncology
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Ruxolitinib is a Other Small Molecule drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Primary Myelofibrosis.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
July 19, 2017
Details:
Madecassoside is a plant extract/herbal drug, which is currently being evaluated in Phase II clinical studies for the treatment of graft vs host disease.
Lead Product(s): Madecassoside,Ruxolitinib Phosphate,Belumosudil
Therapeutic Area: Immunology Brand Name: Undisclosed
Study Phase: Phase IIProduct Type: Plant Extract/Herbal
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable May 26, 2026

Lead Product(s) : Madecassoside,Ruxolitinib Phosphate,Belumosudil
Therapeutic Area : Immunology
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Madecassoside is a plant extract/herbal drug, which is currently being evaluated in Phase II clinical studies for the treatment of graft vs host disease.
Product Name : Undisclosed
Product Type : Plant Extract/Herbal
Upfront Cash : Inapplicable
May 26, 2026

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Ruxolitinib is a small molecule drug, which is currently being evaluated in Phase I clinical studies for the treatment of undisclosed medical condition.
Lead Product(s): Ruxolitinib Phosphate,Povorcitinib
Therapeutic Area: Undisclosed Brand Name: Undisclosed
Study Phase: Phase IProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable May 14, 2026

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Ruxolitinib Phosphate,Povorcitinib
Therapeutic Area : Undisclosed
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Ruxolitinib is a small molecule drug, which is currently being evaluated in Phase I clinical studies for the treatment of undisclosed medical condition.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
May 14, 2026

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
L19IL2 (Bifikafusp Alfa) is a antibody-protein conjugate drug, which is currently being evaluated in Phase I clinical studies for the treatment of carcinoma, renal cell.
Lead Product(s): Bifikafusp Alfa,Ruxolitinib Phosphate
Therapeutic Area: Oncology Brand Name: L19IL2
Study Phase: Phase IProduct Type: Antibody-protein Conjugate
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable May 05, 2026

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Bifikafusp Alfa,Ruxolitinib Phosphate
Therapeutic Area : Oncology
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : L19IL2 (Bifikafusp Alfa) is a antibody-protein conjugate drug, which is currently being evaluated in Phase I clinical studies for the treatment of carcinoma, renal cell.
Product Name : L19IL2
Product Type : Antibody-protein Conjugate
Upfront Cash : Inapplicable
May 05, 2026

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Ruxolitinib, a miscellaneous product targeting Tyrosine-protein kinase JAK1 and JAK2, shows promise in treating Myelofibrosis, Polycythemia Vera, and Graft-Versus-Host Disease.
Lead Product(s): Ruxolitinib Phosphate,Inapplicable
Therapeutic Area: Oncology Brand Name: Jakafi
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable May 01, 2026

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Ruxolitinib Phosphate,Inapplicable
Therapeutic Area : Oncology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Incyte Reveals FDA Approval of Jakafi XRTM for Myelofibrosis, Polycythemia Vera, and GVHD
Details : Ruxolitinib, a miscellaneous product targeting Tyrosine-protein kinase JAK1 and JAK2, shows promise in treating Myelofibrosis, Polycythemia Vera, and Graft-Versus-Host Disease.
Product Name : Jakafi
Product Type : Miscellaneous
Upfront Cash : Inapplicable
May 01, 2026

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Regulatory Info :
Registration Country : Iran
Brand Name : Relata
Dosage Form : Tablet
Dosage Strength : 5MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Iran
Packaging :
Regulatory Info :
Dosage : Tablet
Dosage Strength : 5MG
Brand Name : Relata
Approval Date :
Application Number :
Registration Country : Iran
Regulatory Info :
Registration Country : Iran
Brand Name : Relata
Dosage Form : Tablet
Dosage Strength : 10MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Iran
Packaging :
Regulatory Info :
Dosage : Tablet
Dosage Strength : 10MG
Brand Name : Relata
Approval Date :
Application Number :
Registration Country : Iran
Regulatory Info :
Registration Country : Iran
Brand Name : Relata
Dosage Form : Tablet
Dosage Strength : 15MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Iran
Packaging :
Regulatory Info :
Dosage : Tablet
Dosage Strength : 15MG
Brand Name : Relata
Approval Date :
Application Number :
Registration Country : Iran
Regulatory Info :
Registration Country : Iran
Brand Name : Relata
Dosage Form : Tablet
Dosage Strength : 20MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Iran
Packaging :
Regulatory Info :
Dosage : Tablet
Dosage Strength : 20MG
Brand Name : Relata
Approval Date :
Application Number :
Registration Country : Iran
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Related Excipient Companies

Dosage Form : Tablet
Grade : Oral
Dosage Form : Tablet
Grade : Oral
Application : Fillers, Diluents & Binders
Excipient Details : Microlose (Lactose Monohydrate & Microcrystalline Cellulose) is used as a diluent in oral dosage forms such as tablets.
Pharmacopoeia Ref : USP-NF,BP,EP,IP
Technical Specs : Lactose Monohydrate – 40%, Microcrystalline cellulose – 60%
Ingredient(s) : Lactose Monohydrate
Dosage Form : Capsule
Grade : Oral
Application : Fillers, Diluents & Binders
Excipient Details : ProBlend (SMCC) is a co-processed excipient consists of microcrystalline cellulose & colloidal silicon dioxide, used as a diluent & binder in OSDs.
Pharmacopoeia Ref : USP-NF,BP,EP,IP
Technical Specs : NA
Ingredient(s) : Microcrystalline Cellulose Excipients
Dosage Form : Tablet
Grade : Oral
Application : Fillers, Diluents & Binders
Excipient Details : Starlose P40 (Starch & Lactose Monohydrate) is used as a diluent in oral dosage forms such as tablets.
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
98
PharmaCompass offers a list of Ruxolitinib Phosphate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ruxolitinib Phosphate manufacturer or Ruxolitinib Phosphate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Ruxolitinib Phosphate manufacturer or Ruxolitinib Phosphate supplier.
A Ruxolitinib Phosphate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Ruxolitinib Phosphate, including repackagers and relabelers. The FDA regulates Ruxolitinib Phosphate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Ruxolitinib Phosphate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Ruxolitinib Phosphate manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Ruxolitinib Phosphate supplier is an individual or a company that provides Ruxolitinib Phosphate active pharmaceutical ingredient (API) or Ruxolitinib Phosphate finished formulations upon request. The Ruxolitinib Phosphate suppliers may include Ruxolitinib Phosphate API manufacturers, exporters, distributors and traders.
click here to find a list of Ruxolitinib Phosphate suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Ruxolitinib Phosphate DMF (Drug Master File) is a document detailing the whole manufacturing process of Ruxolitinib Phosphate active pharmaceutical ingredient (API) in detail. Different forms of Ruxolitinib Phosphate DMFs exist exist since differing nations have different regulations, such as Ruxolitinib Phosphate USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Ruxolitinib Phosphate DMF submitted to regulatory agencies in the US is known as a USDMF. Ruxolitinib Phosphate USDMF includes data on Ruxolitinib Phosphate's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Ruxolitinib Phosphate USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Ruxolitinib Phosphate suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Ruxolitinib Phosphate Drug Master File in Korea (Ruxolitinib Phosphate KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Ruxolitinib Phosphate. The MFDS reviews the Ruxolitinib Phosphate KDMF as part of the drug registration process and uses the information provided in the Ruxolitinib Phosphate KDMF to evaluate the safety and efficacy of the drug.
After submitting a Ruxolitinib Phosphate KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Ruxolitinib Phosphate API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Ruxolitinib Phosphate suppliers with KDMF on PharmaCompass.
A Ruxolitinib Phosphate written confirmation (Ruxolitinib Phosphate WC) is an official document issued by a regulatory agency to a Ruxolitinib Phosphate manufacturer, verifying that the manufacturing facility of a Ruxolitinib Phosphate active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Ruxolitinib Phosphate APIs or Ruxolitinib Phosphate finished pharmaceutical products to another nation, regulatory agencies frequently require a Ruxolitinib Phosphate WC (written confirmation) as part of the regulatory process.
click here to find a list of Ruxolitinib Phosphate suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Ruxolitinib Phosphate as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Ruxolitinib Phosphate API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Ruxolitinib Phosphate as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Ruxolitinib Phosphate and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Ruxolitinib Phosphate NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Ruxolitinib Phosphate suppliers with NDC on PharmaCompass.
Ruxolitinib Phosphate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Ruxolitinib Phosphate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ruxolitinib Phosphate GMP manufacturer or Ruxolitinib Phosphate GMP API supplier for your needs.
A Ruxolitinib Phosphate CoA (Certificate of Analysis) is a formal document that attests to Ruxolitinib Phosphate's compliance with Ruxolitinib Phosphate specifications and serves as a tool for batch-level quality control.
Ruxolitinib Phosphate CoA mostly includes findings from lab analyses of a specific batch. For each Ruxolitinib Phosphate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Ruxolitinib Phosphate may be tested according to a variety of international standards, such as European Pharmacopoeia (Ruxolitinib Phosphate EP), Ruxolitinib Phosphate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Ruxolitinib Phosphate USP).

