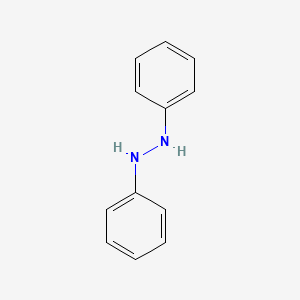

1. Hydrazobenzene
1. Hydrazobenzene
2. 122-66-7
3. 1,2-diphenyldiazane
4. N,n'-bianiline
5. N,n'-diphenylhydrazine
6. Hydrazobenzen
7. Benzene, Hydrazodi-
8. Hydrazine, 1,2-diphenyl-
9. (sym)-diphenylhydrazine
10. Rcra Waste Number U109
11. Nci-c01854
12. Benzene, 1,1'-hydrazobis-
13. Diphenylhydrazine
14. Nsc 3510
15. 1g3cs09tuk
16. 1,2-diphenylhydrazine (9ci)
17. Nsc-3510
18. 38622-18-3
19. Dsstox_cid_710
20. Dsstox_rid_75751
21. Dsstox_gsid_20710
22. Hydrazobenzen [czech]
23. 1,1'-hydrazodibenzene
24. Diphenylhydrazine, 1,2-
25. Cas-122-66-7
26. Symmetrical Diphenyl Hydrazine
27. Ccris 337
28. Hydrazine, Diphenyl-
29. 1,2-diphenyldiazane (1,2-diphenylhydrazine)
30. Hsdb 2882
31. Einecs 204-563-5
32. Unii-1g3cs09tuk
33. Brn 0639793
34. Hydrazobenzol
35. Ai3-15365
36. Hydrazine,2-diphenyl-
37. N,n'-biphenylhydrazine
38. 1,2-diphenyl-hydrazine
39. Benzene,1'-hydrazobis-
40. Wln: Rmmr
41. Mr-ii-10
42. Schembl51179
43. Hydrazobenzene [hsdb]
44. 4-15-00-00056 (beilstein Handbook Reference)
45. Chembl558459
46. Dtxsid7020710
47. Ybqzxxmejhzymb-uhfffaoysa-
48. Nsc3510
49. Bdbm233141
50. Zinc1666823
51. Tox21_202323
52. Tox21_300492
53. Mfcd00003012
54. Stl283944
55. Akos015889796
56. Ncgc00091240-01
57. Ncgc00091240-02
58. Ncgc00091240-03
59. Ncgc00091240-04
60. Ncgc00254433-01
61. Ncgc00259872-01
62. Db-005623
63. Ft-0600571
64. Ft-0629583
65. H0180
66. F15405
67. Phenylbutazone Impurity C [ep Impurity]
68. A804943
69. Q424993
70. W-108426
71. 1,2-diphenylhydrazine, Analytical Standard, Ampule Of 100 Mg
| Molecular Weight | 184.24 g/mol |
|---|---|
| Molecular Formula | C12H12N2 |
| XLogP3 | 2.9 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 3 |
| Exact Mass | 184.100048391 g/mol |
| Monoisotopic Mass | 184.100048391 g/mol |
| Topological Polar Surface Area | 24.1 Ų |
| Heavy Atom Count | 14 |
| Formal Charge | 0 |
| Complexity | 128 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Thin layer chromatographic analysis of the urine of rats given an oral dose of 200 or 400 mg/kg revealed the presence of hydrazobenzene, benzidine, and two other unidentified metabolites. At an ip dose of 100 or 200 mg/kg, hydrazobenzene, aniline, benzidine, p-aminophenol, and o-aminophenol were identified in the urine. After iv or intratracheal administration of hydrazobenzene, only one urinary metabolite, which appeared to be phenolic, was found.
USEPA; Chemical Hazard Information Profile: Hydrazobenzene (Draft) p.6 (1981)
In the stomach, 1,2-diphenylhydrazine can be converted into benzidine, a known human carcinogen.
USEPA; Ambient Water Quality Criteria Doc: Diphenylhydrazine p.A-1 (1980) EPA 440/5-80-062
The enzyme systems in rat liver and lung responsible for the oxidative metabolism of hydrazine derivatives were studied to determine whether these enzymes, cytochrome P450 and monoamine oxidase, were responsible for metabolically activating hydrazines to carcinogenic/toxic metabolites. Cytochrome P450 preferentially oxidized the nitrogen to nitrogen bond of 1,2-disubstituted hydrazines and hydrazides, while monoamine oxidase oxidized the nitrogen to nitrogen bond of all the classes of hydrazine derivatives that were tested. Oxidation of the nitrogen to nitrogen bond led to the formation of stable azo intemediates in the case of 1,2-disubstituted hydrazines and to unstable monoazo (diazene) metabolites in the case of monosubstituted hydrazines and hydrazides. /Substituted hydrazines/
PMID:3271870 Erikson JM, Prough RA; J Biochem Toxicol 1 (1): 41-52 (1986)
BUILDING BLOCK


