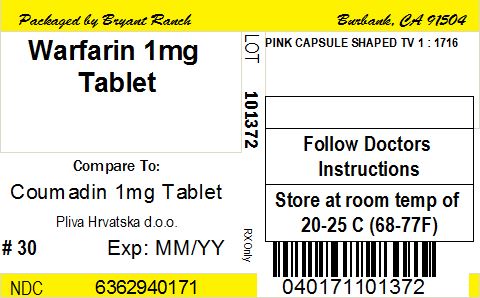
NDC Code(s) : 63629-4017-1, 63629-4017-2, 63629-4017-3, 63629-4017-4, 63629-4017-5, 63629-4017-6, 63629-4017-7, 63629-4017-8
Packager : Bryant Ranch Prepack
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| Warfarin SodiumWarfarin Sodium TABLET | ||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
PRINCIPAL DISPLAY PANEL