
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
DRUG PRODUCT COMPOSITIONS
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
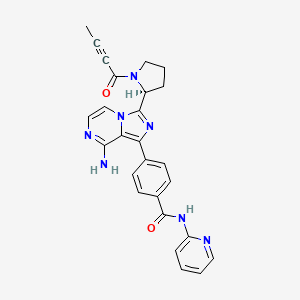

1. Acp-196
2. Calquence
1. 1420477-60-6
2. Acp-196
3. Calquence
4. Acalabrutinib (acp-196)
5. Acalabrutinib [inn]
6. I42748elqw
7. 4-[8-amino-3-[(2s)-1-but-2-ynoylpyrrolidin-2-yl]imidazo[1,5-a]pyrazin-1-yl]-n-pyridin-2-ylbenzamide
8. Benzamide, 4-(8-amino-3-((2s)-1-(1-oxo-2-butyn-1-yl)-2-pyrrolidinyl)imidazo(1,5-a)pyrazin-1-yl)-n-2-pyridinyl-
9. (s)-4-(8-amino-3-(1-(but-2-ynoyl)pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-yl)-n-(pyridin-2-yl)benzamide
10. Acalabrutinib(acp196)
11. Acalabrutinib [usan:inn]
12. Acalabrutinibum
13. Unii-i42748elqw
14. Calquence (tn)
15. Benzamide, 4-[8-amino-3-[(2s)-1-(1-oxo-2-butyn-1-yl)-2-pyrrolidinyl]imidazo[1,5-a]pyrazin-1-yl]-n-2-pyridinyl-
16. Acp-196;acalabrutinib
17. Acalabrutinib [mi]
18. Acalabrutinib [jan]
19. Acalabrutinib [usan]
20. Acalabrutinib [who-dd]
21. Gtpl8912
22. Acalabrutinib (jan/usan/inn)
23. Chembl3707348
24. Schembl14637368
25. Acp 196
26. Amy5290
27. Ex-a881
28. Chebi:167707
29. Dtxsid401026209
30. Acalabrutinib [orange Book]
31. Bdbm50175583
32. Mfcd29472294
33. Nsc791164
34. Nsc800976
35. S8116
36. Zinc208774715
37. Ccg-269407
38. Cs-5356
39. Db11703
40. Ds-3326
41. Nsc-791164
42. Nsc-800976
43. Ncgc00479074-01
44. Hy-17600
45. Example 6 [us20140155385 A1]
46. D10893
47. A857446
48. J-690166
49. Q23668732
50. (s)-4-(8-amino-3-(1-(but-2-ynoyl)pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-yl)-n-(pyridin-2-yl)benzamide;acalabrutinib
51. (s)-4-(8-amino-3-(1-but-2-ynoylpyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-yl)-n-(pyridin-2-yl)benzamide
52. 4-[8-amino-3-[(2s)-1-(1-oxo-2-butyn-1-yl)-2-pyrrolidinyl]imidazo[1,5-a]pyrazin-1-yl]-n-2-pyridinyl-benzamide
53. 4-[8-amino-3-[(2s)-1-but-2-ynoylpyrrolidin-2-yl]imidazo[1,5-a]pyrazin-1-yl]-n-(2-pyridyl)benzamide
54. 4-{8-amino-3-[(2s)-1-(but-2-ynoyl)pyrrolidin-2-yl]imidazo[1,5-a]pyrazin-1-yl}-n-(pyridin-2-yl)benzamide
| Molecular Weight | 465.5 g/mol |
|---|---|
| Molecular Formula | C26H23N7O2 |
| XLogP3 | 3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 4 |
| Exact Mass | 465.19132300 g/mol |
| Monoisotopic Mass | 465.19132300 g/mol |
| Topological Polar Surface Area | 119 Ų |
| Heavy Atom Count | 35 |
| Formal Charge | 0 |
| Complexity | 845 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 1 | |
|---|---|
| Drug Name | CALQUENCE |
| Active Ingredient | ACALABRUTINIB |
| Company | ASTRAZENECA (Application Number: N210259. Patents: 9290504, 9758524, 9796721) |
Acalabrutinib is currently indicated for the treatment of adult patients with Mantle Cell Lymphoma (MCL) who have received at least one prior therapy. It has also been recently approved for chronic lymphocytic leukemia and small lymphocytic lymphoma.
FDA Label
Calquence as monotherapy or in combination with obinutuzumab is indicated for the treatment of adult patients with previously untreated chronic lymphocytic leukaemia (CLL).
Calquence as monotherapy is indicated for the treatment of adult patients with chronic lymphocytic leukaemia (CLL) who have received at least one prior therapy.
Acalabrutinib is a Bruton Tyrosine Kinase inhibitor that prevents the proliferation, trafficking, chemotaxis, and adhesion of B cells. It is taken every 12 hours and can cause other effects such as atrial fibrillation, other malignancies, cytopenia, hemorrhage, and infection.
Antineoplastic Agents
Substances that inhibit or prevent the proliferation of NEOPLASMS. (See all compounds classified as Antineoplastic Agents.)
L01XE51
L - Antineoplastic and immunomodulating agents
L01 - Antineoplastic agents
L01E - Protein kinase inhibitors
L01EL - Bruton's tyrosine kinase (btk) inhibitors
L01EL02 - Acalabrutinib
Absorption
The geometric mean absolute bioavailability of acalabrutinib is 25% with a median time to peak plasma concentrations (Tmax) of 0.75 hours.
Route of Elimination
After administration of a single 100 mg radiolabelled acalabrutinib dose in healthy subjects, 84% of the dose was recovered in the feces and 12% of the dose was recovered in the urine. An irradiated dose of acalabrutinib was 34.7% recovered as the metabolite ACP-5862; 8.6% was recovered as unchanged acalabrutinub; 10.8 was recovered as a mixture of the M7, M8, M9, M10, and M11 metabolites; 5.9% was the M25 metabolite; 2.5% was recovered as the M3 metabolite.
Volume of Distribution
The mean steady-state volume of distribution is approximately 34 L.
Clearance
Acalabrutinib's mean apparent oral clearance (CL/F) is observed to be 159 L/hr with similar PK between patients and healthy subjects, based on population PK analysis.
Acalabrutinib is mainly metabolized by CYP3A enzymes. ACP-5862 is identified to be the major active metabolite in plasma with a geometric mean exposure (AUC) that is about 2-3 times greater than the exposure of acalabrutinib. ACP-5862 is about 50% less potent than acalabrutinib in regards to the inhibition of BTK.
After administering a single oral dose of 100 mg acalabrutinib, the median terminal elimination half-life of the drug was found to be 0.9 (with a range of 0.6 to 2.8) hours. The half-life of the active metabolite, ACP-5862, is about 6.9 hours.
Mantle Cell Lymphoma (MCL) is a rare yet aggressive type of B-cell non-Hodgkin lymphoma (NHL) with poor prognosis. Subsequently, relapse is common in MCL patients and ultimately represents disease progression. Lymphoma occurs when immune system lymphocytes grow and multiply uncontrollably. Such cancerous lymphocytes may travel to many parts of the body, including the lymph nodes, spleen, bone marrow, blood, and other organs where they can multiply and form a mass(es) called a tumor. One of the main kinds of lymphocytes that can develop into cancerous lymphomas are the body's own B-lymphocytes (B-cells). Bruton Tyrosine Kinase (BTK) is a signalling molecule of the B-cell antigen receptor and cytokine receptor pathways. Such BTK signaling causes the activation of pathways necessary for B-cell proliferation, trafficking, chemotaxis, and adhesion. Acalabrutinib is a small molecule inhibitor of BTK. Both acalabrutinib and its active metabolite, ACP-5862, act to form a covalent bond with a cysteine residue (Cys481) in the BTK active site, leading to inhibition of BTK enzymatic activity. As a result, acalabrutinib inhibits BTK-mediated activation of downstream signaling proteins CD86 and CD69, which ultimately inhibits malignant B-cell proliferation and survival Whereas ibrutinib is typically recognized as the first-in-class BTK inhibitor, acalabrutinib is considered a second generation BTK inhibitor primarily because it demonstrates highter selectivity and inhibition of the targeted activity of BTK while having a much greater IC50 or otherwise virtually no inhibition on the kinase activities of ITK, EGFR, ERBB2, ERBB4, JAK3, BLK, FGR, FYN, HCK, LCK, LYN, SRC, and YES1. In effect, acalabrutinib was rationally designed to be more potent and selective than ibrutinib, all the while demonstrating fewer adverse effects - in theory - because of the drug's minimized off target effects.

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
16
PharmaCompass offers a list of Acalabrutinib API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Acalabrutinib manufacturer or Acalabrutinib supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Acalabrutinib manufacturer or Acalabrutinib supplier.
PharmaCompass also assists you with knowing the Acalabrutinib API Price utilized in the formulation of products. Acalabrutinib API Price is not always fixed or binding as the Acalabrutinib Price is obtained through a variety of data sources. The Acalabrutinib Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Acalabrutinib manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Acalabrutinib, including repackagers and relabelers. The FDA regulates Acalabrutinib manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Acalabrutinib API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Acalabrutinib manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Acalabrutinib supplier is an individual or a company that provides Acalabrutinib active pharmaceutical ingredient (API) or Acalabrutinib finished formulations upon request. The Acalabrutinib suppliers may include Acalabrutinib API manufacturers, exporters, distributors and traders.
click here to find a list of Acalabrutinib suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Acalabrutinib DMF (Drug Master File) is a document detailing the whole manufacturing process of Acalabrutinib active pharmaceutical ingredient (API) in detail. Different forms of Acalabrutinib DMFs exist exist since differing nations have different regulations, such as Acalabrutinib USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Acalabrutinib DMF submitted to regulatory agencies in the US is known as a USDMF. Acalabrutinib USDMF includes data on Acalabrutinib's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Acalabrutinib USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Acalabrutinib suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Acalabrutinib Drug Master File in Korea (Acalabrutinib KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Acalabrutinib. The MFDS reviews the Acalabrutinib KDMF as part of the drug registration process and uses the information provided in the Acalabrutinib KDMF to evaluate the safety and efficacy of the drug.
After submitting a Acalabrutinib KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Acalabrutinib API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Acalabrutinib suppliers with KDMF on PharmaCompass.
A Acalabrutinib written confirmation (Acalabrutinib WC) is an official document issued by a regulatory agency to a Acalabrutinib manufacturer, verifying that the manufacturing facility of a Acalabrutinib active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Acalabrutinib APIs or Acalabrutinib finished pharmaceutical products to another nation, regulatory agencies frequently require a Acalabrutinib WC (written confirmation) as part of the regulatory process.
click here to find a list of Acalabrutinib suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Acalabrutinib as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Acalabrutinib API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Acalabrutinib as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Acalabrutinib and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Acalabrutinib NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Acalabrutinib suppliers with NDC on PharmaCompass.
Acalabrutinib Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Acalabrutinib GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Acalabrutinib GMP manufacturer or Acalabrutinib GMP API supplier for your needs.
A Acalabrutinib CoA (Certificate of Analysis) is a formal document that attests to Acalabrutinib's compliance with Acalabrutinib specifications and serves as a tool for batch-level quality control.
Acalabrutinib CoA mostly includes findings from lab analyses of a specific batch. For each Acalabrutinib CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Acalabrutinib may be tested according to a variety of international standards, such as European Pharmacopoeia (Acalabrutinib EP), Acalabrutinib JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Acalabrutinib USP).
