28 Jan 2026
// PRESS RELEASE
21 Jan 2026
// PRESS RELEASE
14 Jan 2026
// PRESS RELEASE
Latest Content by PharmaCompass
 KEY PRODUCTS
KEY PRODUCTS
Coral's API range includes Corticosteroids, Antihistamines. Hormones and Antineoplastics.
About
Industry Trade Show
Booth #E1D25
16-18 June, 2026
CPhI South East AsiaCPhI South East Asia
Industry Trade Show
Exhibiting
08-10 July, 2026
Industry Trade Show
Booth #4C87
06-08 October, 2026
CONTACT DETAILS






Events
Webinars & Exhibitions
Industry Trade Show
Booth #E1D25
16-18 June, 2026
CPhI South East AsiaCPhI South East Asia
Industry Trade Show
Exhibiting
08-10 July, 2026
Industry Trade Show
Booth #4C87
06-08 October, 2026
VLOG #PharmaReel
CORPORATE CONTENT #SupplierSpotlight
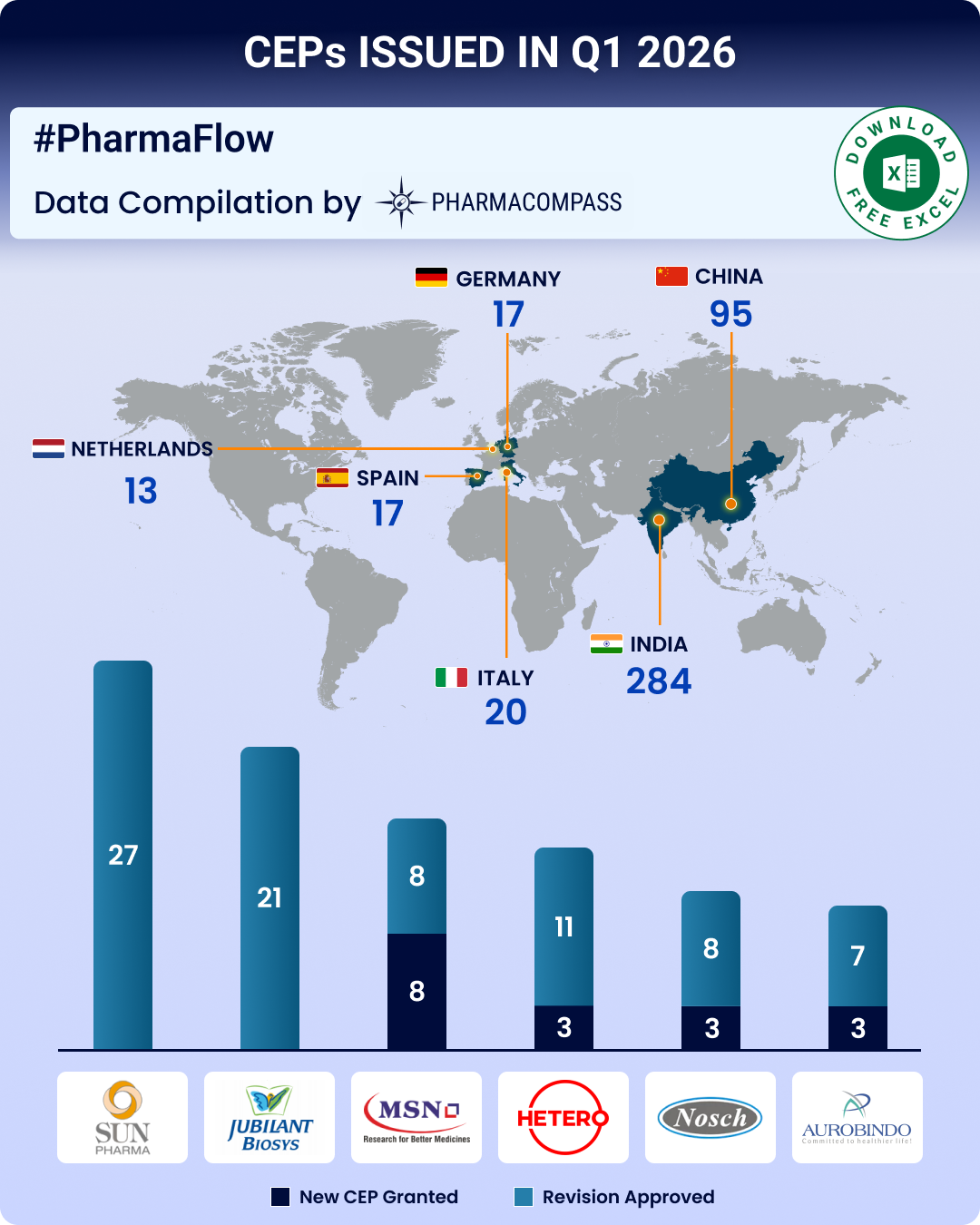
https://www.pharmacompass.com/radio-compass-blog/cep-q1-2026-update-cep-2-0-edqm-s-new-guidelines-strengthen-ecosystem-indian-firms-top-list-of-ceps-issued

28 Jan 2026
// PRESS RELEASE
https://www.coraldrugs.com/news/what-a-world-class-api-manufacturing-facility-looks-like

21 Jan 2026
// PRESS RELEASE
https://www.coraldrugs.com/news/precision-nano-sizing-shaping-the-future-of-advanced-apis

14 Jan 2026
// PRESS RELEASE
https://www.coraldrugs.com/news/precision-at-the-micron-level-for-reliable-apis

07 Jan 2026
// PRESS RELEASE
https://www.coraldrugs.com/news/indias-60-000-crore-api-push-and-its-impact-on-pharmaceutical-manufacturing

08 Oct 2025
// PRESS RELEASE
https://www.coraldrugs.com/news/coral-drugs-where-quality-meets-innovation-in-api-manufacturing

26 Aug 2025
// PRESS RELEASE
https://www.coraldrugs.com/news/us-fda-inspection-successfully-concluded-at-coral-drugs-facility
Services
API Manufacturing
API & Drug Product Development
ABOUT THIS PAGE
Coral Drugs Private Limited is a supplier offers 44 products (APIs, Excipients or Intermediates).
Find Azelastine Hydrochloride bulk with DMF, CEP, WC offered by Coral Drugs Private Limited
Find Budesonide bulk with DMF, CEP, WC offered by Coral Drugs Private Limited
Find Ciclesonide bulk with DMF, CEP, WC offered by Coral Drugs Private Limited
Find Exemestane bulk with DMF, CEP, WC offered by Coral Drugs Private Limited
Find Fluticasone Furoate bulk with DMF, CEP, WC offered by Coral Drugs Private Limited
Find Fluticasone Propionate bulk with DMF, CEP, WC offered by Coral Drugs Private Limited
Find Formoterol Fumarate bulk with DMF, CEP, WC offered by Coral Drugs Private Limited
Find Mometasone Furoate bulk with DMF, CEP, WC offered by Coral Drugs Private Limited
Find Triamcinolone Hexacetonide bulk with DMF, CEP, WC offered by Coral Drugs Private Limited
Find Fluticasone Furoate bulk with DMF, CEP offered by Coral Drugs Private Limited
Find Fluticasone Propionate bulk with DMF, CEP offered by Coral Drugs Private Limited
Find Triamcinolone Acetonide bulk with DMF, CEP offered by Coral Drugs Private Limited
Find Deflazacort bulk with DMF, WC offered by Coral Drugs Private Limited
Find Flutamide bulk with CEP, WC offered by Coral Drugs Private Limited
Find Triamcinolone bulk with CEP, WC offered by Coral Drugs Private Limited
Find Mometasone Furoate bulk with DMF offered by Coral Drugs Private Limited
Find Clobetasol Propionate bulk with WC offered by Coral Drugs Private Limited
Find Danazol bulk with WC offered by Coral Drugs Private Limited
Find Fluocinolone Acetonide bulk with WC offered by Coral Drugs Private Limited
Find Fluocinonide bulk with WC offered by Coral Drugs Private Limited
Find Isoflupredone Acetate bulk with WC offered by Coral Drugs Private Limited
Find Salmeterol Xinafoate bulk with WC offered by Coral Drugs Private Limited
Find Azelastine Hydrochloride bulk offered by Coral Drugs Private Limited
Find Budesonide bulk offered by Coral Drugs Private Limited
Find Ciclesonide bulk offered by Coral Drugs Private Limited
Find Clobetasol Propionate bulk offered by Coral Drugs Private Limited
Find Danazol bulk offered by Coral Drugs Private Limited
Find Exemestane bulk offered by Coral Drugs Private Limited
Find Flutamide bulk offered by Coral Drugs Private Limited
Find Formoterol Fumarate bulk offered by Coral Drugs Private Limited
Find Isoflupredone Acetate bulk offered by Coral Drugs Private Limited
Find Mometasone Furoate bulk offered by Coral Drugs Private Limited
Find Salmeterol Xinafoate bulk offered by Coral Drugs Private Limited
Find Triamcinolone bulk offered by Coral Drugs Private Limited
Find Beclomethasone Dipropionate bulk offered by Coral Drugs Private Limited
Find Diflucortolone Valerate bulk offered by Coral Drugs Private Limited
Find Glycopyrronium Bromide bulk offered by Coral Drugs Private Limited
Find Indacaterol Acetate bulk offered by Coral Drugs Private Limited
Find Indacaterol Maleate bulk offered by Coral Drugs Private Limited
Find Medroxyprogesterone Acetate bulk offered by Coral Drugs Private Limited
Find Megestrol Acetate bulk offered by Coral Drugs Private Limited
Find Norethisterone bulk offered by Coral Drugs Private Limited
Find Norethisterone Acetate bulk offered by Coral Drugs Private Limited
Find Norethisterone Enanthate bulk offered by Coral Drugs Private Limited


 Coral Drugs Private Limited
Coral Drugs Private Limited