
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
VMF
0
Australia
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Annual Reports
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Azol
2. Cyclomen
3. Danatrol
4. Danazant
5. Danazol Ratiopharm
6. Danazol-ratiopharm
7. Danocrine
8. Danol
9. Danoval
10. Ladogal
11. Norciden
12. Panacrine
1. Danocrine
2. 17230-88-5
3. Chronogyn
4. Winobanin
5. Danol
6. Cyclomen
7. Danazolum
8. Ladogal
9. Danzol
10. Danatrol
11. Danazol, Usp
12. Win-17757
13. Win 17,757
14. Danazole
15. N29qww3buo
16. (1r,3as,3br,10ar,10bs,12as)-1-ethynyl-10a,12a-dimethyl-2,3,3a,3b,4,5,10,10a,10b,11,12,12a-dodecahydro-1h-cyclopenta[7,8]phenanthro[3,2-d]isoxazol-1-ol
17. Win 17757
18. Danovaol
19. Chebi:4315
20. Danokrin
21. Nsc-270916
22. Ncgc00164400-01
23. Danogen
24. Pregna-2,4-dien-20-yno(2,3-d)isoxazol-17-ol, (17alpha)-
25. Dsstox_cid_2880
26. Danazolum [inn-latin]
27. Nsc 270916
28. Dsstox_rid_76772
29. Dsstox_gsid_22880
30. Bonzol
31. Danocrine (tn)
32. Smr000058321
33. Ccris 6747
34. Einecs 241-270-1
35. Unii-n29qww3buo
36. Vasaloc
37. 17-alpha-pregn-4-en-20-yno(2,3-d)isoxazol-17-ol
38. Optina
39. Pregna-2,4-dien-20-yno[2,3-d]isoxazol-17-ol, (17alpha)-
40. 17-alpha-2,4-pregnadien-20-yno(2,3-d)isoxazol-17-ol
41. 17alpha-pregna-2,4-dien-20-yno(2,3-d)isoxazol-17-ol
42. Nsc270916
43. Win 17, 757
44. Prestwick_150
45. Cas-17230-88-5
46. Danazol [usan:usp:inn:ban:jan]
47. Danazol [vandf]
48. Ethynyl(dimethyl)[?]ol
49. 2,3-isoxazolethisterone
50. Danazol [usan]
51. Danazol [inn]
52. Danazol [jan]
53. Danazol [mi]
54. Danazol [mart.]
55. Prestwick0_000105
56. Prestwick1_000105
57. Prestwick2_000105
58. Prestwick3_000105
59. Danazol [usp-rs]
60. Danazol [who-dd]
61. Chembl1479
62. Schembl21107
63. Bspbio_000090
64. (1s,2r,13r,14s,17r,18s)-17-ethynyl-2,18-dimethyl-7-oxa-6-azapentacyclo[11.7.0.0^{2,10}.0^{4,8}.0^{14,18}]icosa-4(8),5,9-trien-17-ol
65. Mls001066617
66. Mls001306473
67. Danazol (jp17/usp/inn)
68. Danazol [orange Book]
69. Spbio_002029
70. Bpbio1_000100
71. Gtpl6942
72. Danazol [usp Monograph]
73. Dtxsid2022880
74. 2,4,17alpha-pregnadien-20-yno[2,3-d]-isoxa-zol-17-ol
75. Win-17,757
76. Hms1568e12
77. Hms2090a22
78. Hms2095e12
79. Hms2231m08
80. Hms3259m10
81. Hms3712e12
82. Pregna-2,4-dien-20-yno(2,3-d)isoxazol-17-ol,(17alpha)-
83. [1,2]oxazolo[4',5':2,3]-17alpha-pregn-4-en-20-yn-17-ol
84. 17.alpha.-pregna-2, {4-dien-20-yno[2,3-d]isoxazol-17-ol}
85. Bcp11914
86. Hy-b1029
87. Zinc3881958
88. Tox21_112114
89. Tox21_301940
90. Bdbm50423541
91. Mfcd00056838
92. S9506
93. Akos015961192
94. Tox21_112114_1
95. Ac-6836
96. Ccg-220105
97. Cs-4547
98. Danazol 100 Microg/ml In Acetonitrile
99. Db01406
100. Nc00557
101. Ncgc00179665-01
102. Ncgc00179665-02
103. Ncgc00179665-04
104. Ncgc00255335-01
105. (1r,3as,3br,10ar,10bs,12as)-1-ethynyl-10a,12a-dimethyl-2,3,3a,3b,4,5,10,10a,10b,11,12,12a-dodecahydro-1h-cyclopenta[7,8]phenanthro[3,2-d][1,2]oxazol-1-ol
106. 1-ethynyl-10a,12a-dimethyl-2,3,3a,3b,4,5,10,10a,10b,11,12,12a-dodecahydro-1h-cyclopenta[7,8]phenanthro[3,2-d]isoxazol-1-ol
107. As-13035
108. C06938
109. D00289
110. 230d885
111. Q419652
112. Sr-01000760722
113. Sr-05000000445
114. Sr-01000760722-2
115. Sr-05000000445-2
116. W-107864
117. 17a-pregna-2,4-dien-20-yno[2,3-d]isoxazol-17-ol
118. Brd-k48970916-001-03-0
119. (17a)-pregna-2,4-dien-20-yno[2,3-d]isoxazol-17-ol
120. 17a-pregna-2,4-dien-20-yne-[2,3-d]isoxazole-17b-ol
121. 1-(p-tosyl)-3,4,4-trimethyl-2-imidazoliniumiodide
122. 17 Alpha-pregna-2,4-dien-20-yno[2,3-d] Isoxazol-17 Beta-ol
123. 17.alpha.-pregna-2,4-dien-20-yno(2,3)-disoxazol-17-ol
124. Pregna-2, {4-dien-20-yno[2,3-d]isoxazol-17-ol,} (17.alpha.)-
125. Pregna-2,4-dien-20-yno(2,3-d)isoxazol-17-ol,(17.alpha.)-
126. (1s,2r,13r,14s,17r,18s)-17-ethynyl-2,18-dimethyl-7-oxa-6-azapentacyclo[11.7.0.02,10.04,8.014,18]icosa-4(8),5,9-trien-17-ol
127. 1-ethynyl-2,3,3a,3b,4,5,10,10a,10b,11,12,12a-dodecahydro-10a,12a-dimethyl-1h-cyclopenta[7,8]phenanthro[3,2-d]isoxazol-1-ol
128. 1h-cyclopenta[7,8]phenanthro[3,2-d]isoxazole- Pregna-2,4-dien-20-yno[2,3-d]isoxazol-17-ol Deriv.
| Molecular Weight | 337.5 g/mol |
|---|---|
| Molecular Formula | C22H27NO2 |
| XLogP3 | 3.8 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 1 |
| Exact Mass | 337.204179104 g/mol |
| Monoisotopic Mass | 337.204179104 g/mol |
| Topological Polar Surface Area | 46.3 Ų |
| Heavy Atom Count | 25 |
| Formal Charge | 0 |
| Complexity | 677 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 6 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Danazol |
| PubMed Health | Danazol (By mouth) |
| Drug Classes | Endocrine-Metabolic Agent |
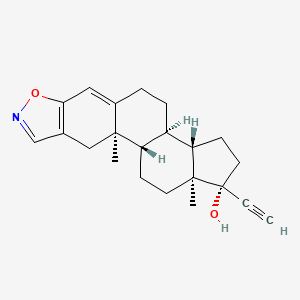
| Drug Label | Danazol is a synthetic steroid derived from ethisterone. It is a white to pale yellow crystalline powder, practically insoluble or insoluble in water, and sparingly soluble in alcohol. Chemically, danazol is 17-Pregna-2, 4-dien-20-yno [2, 3- ]-isox... |
| Active Ingredient | Danazol |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 200mg; 100mg; 50mg |
| Market Status | Prescription |
| Company | Lannett; Barr |
| 2 of 2 | |
|---|---|
| Drug Name | Danazol |
| PubMed Health | Danazol (By mouth) |
| Drug Classes | Endocrine-Metabolic Agent |
| Drug Label | Danazol is a synthetic steroid derived from ethisterone. It is a white to pale yellow crystalline powder, practically insoluble or insoluble in water, and sparingly soluble in alcohol. Chemically, danazol is 17-Pregna-2, 4-dien-20-yno [2, 3- ]-isox... |
| Active Ingredient | Danazol |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 200mg; 100mg; 50mg |
| Market Status | Prescription |
| Company | Lannett; Barr |
For the treatment of endometriosis and fibrocystic breast disease (in patients unresponsive to simple measures). Also used for the prophylactic treatment of all types of hereditary angioedema in males and females.
Danazol is a derivative of the synthetic steroid ethisterone, a modified testosterone. It was approved by the U.S. Food and Drug Administration (FDA) as the first drug to specifically treat endometriosis, but its role as a treatment for endometriosis has been largely replaced by the gonadotropin-releasing hormone (GnRH) agonists. Danazol has antigonadotropic and anti-estrogenic activities. Danazol acts as an anterior pituitary suppressant by inhibiting the pituitary output of gonadotropins. It possesses some androgenic properties.
Estrogen Antagonists
Compounds which inhibit or antagonize the action or biosynthesis of estrogenic compounds. (See all compounds classified as Estrogen Antagonists.)
G - Genito urinary system and sex hormones
G03 - Sex hormones and modulators of the genital system
G03X - Other sex hormones and modulators of the genital system
G03XA - Antigonadotropins and similar agents
G03XA01 - Danazol
Hepatic, to principal metabolites, ethisterone and 17-hydroxymethylethisterone.
Approximately 24 hours.
As a gonadotropin inhibitor, danazol suppresses the pituitary-ovarian axis possibly by inhibiting the output of pituitary gonadotropins. Danazol also depresses the preovulatory surge in output of follicle-stimulating hormone (FSH) and luteinizing hormone (LH), thereby reducing ovarian estrogen production. Danazol may also directly inhibits ovarian steroidogenesis; bind to androgen, progesterone, and glucocorticoid receptors; bind to sex-hormone-binding globulin and corticosteroid-binding globulin; and increases the metabolic clearance rate of progesterone. Another mechanism of action by which danazol may use to facilitate regression of endometriosis is by decreasing IgG, IgM, and IgA concentrations, as well as phospholipid and IgG isotope autoantibodies. In the treatment of endometriosis, as a consequence of suppression of ovarian function, danazol causes both normal and ectopic endometrial tissues to become inactive and atrophic. This leads to anovulation and associated amenorrhea. In fibrocystic breast disease, the exact mechanism of action of danazol is unknown, but may be related to suppressed estrogenic stimulation as a result of decreased ovarian production of estrogen. A direct effect on steroid receptor sites in breast tissue is also possible. This leads to a disappearance of nodularity, relief of pain and tenderness, and possibly changes in the menstrual pattern. In terms of hereditary angioedema, danazol corrects the underlying biochemical deficiency by increasing serum concentrations of the deficient C1 esterase inhibitor, resulting in increased serum concentrations of the C4 component of the complement system. (Source: PharmGKB)
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
52
PharmaCompass offers a list of Danazol API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Danazol manufacturer or Danazol supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Danazol manufacturer or Danazol supplier.
A Danazol manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Danazol, including repackagers and relabelers. The FDA regulates Danazol manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Danazol API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Danazol manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Danazol supplier is an individual or a company that provides Danazol active pharmaceutical ingredient (API) or Danazol finished formulations upon request. The Danazol suppliers may include Danazol API manufacturers, exporters, distributors and traders.
click here to find a list of Danazol suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Danazol DMF (Drug Master File) is a document detailing the whole manufacturing process of Danazol active pharmaceutical ingredient (API) in detail. Different forms of Danazol DMFs exist exist since differing nations have different regulations, such as Danazol USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Danazol DMF submitted to regulatory agencies in the US is known as a USDMF. Danazol USDMF includes data on Danazol's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Danazol USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Danazol suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Danazol Drug Master File in Korea (Danazol KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Danazol. The MFDS reviews the Danazol KDMF as part of the drug registration process and uses the information provided in the Danazol KDMF to evaluate the safety and efficacy of the drug.
After submitting a Danazol KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Danazol API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Danazol suppliers with KDMF on PharmaCompass.
A Danazol written confirmation (Danazol WC) is an official document issued by a regulatory agency to a Danazol manufacturer, verifying that the manufacturing facility of a Danazol active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Danazol APIs or Danazol finished pharmaceutical products to another nation, regulatory agencies frequently require a Danazol WC (written confirmation) as part of the regulatory process.
click here to find a list of Danazol suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Danazol as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Danazol API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Danazol as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Danazol and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Danazol NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Danazol suppliers with NDC on PharmaCompass.
Danazol Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Danazol GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Danazol GMP manufacturer or Danazol GMP API supplier for your needs.
A Danazol CoA (Certificate of Analysis) is a formal document that attests to Danazol's compliance with Danazol specifications and serves as a tool for batch-level quality control.
Danazol CoA mostly includes findings from lab analyses of a specific batch. For each Danazol CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Danazol may be tested according to a variety of international standards, such as European Pharmacopoeia (Danazol EP), Danazol JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Danazol USP).

