03 Mar 2026
// PRESS RELEASE
24 Feb 2026
// PRESS RELEASE
30 Jan 2026
// PRESS RELEASE
Latest Content by PharmaCompass
 KEY PRODUCTS
KEY PRODUCTS KEY SERVICES
KEY SERVICES
Chunghwa provides cost-effective APIs & advanced intermediates with complete DMF or COS, ensuring quality & reliable production.
About
Industry Trade Show
Exhibiting
16-18 June, 2026
Industry Trade Show
Booth #2C114
06-08 October, 2026
PEGS Boston SummitPEGS Boston Summit
Industry Trade Show
Not Confirmed
11 May-15 November, 2026
CONTACT DETAILS






Events
Webinars & Exhibitions
Industry Trade Show
Exhibiting
16-18 June, 2026
Industry Trade Show
Booth #2C114
06-08 October, 2026
PEGS Boston SummitPEGS Boston Summit
Industry Trade Show
Not Confirmed
11 May-15 November, 2026
CORPORATE CONTENT #SupplierSpotlight
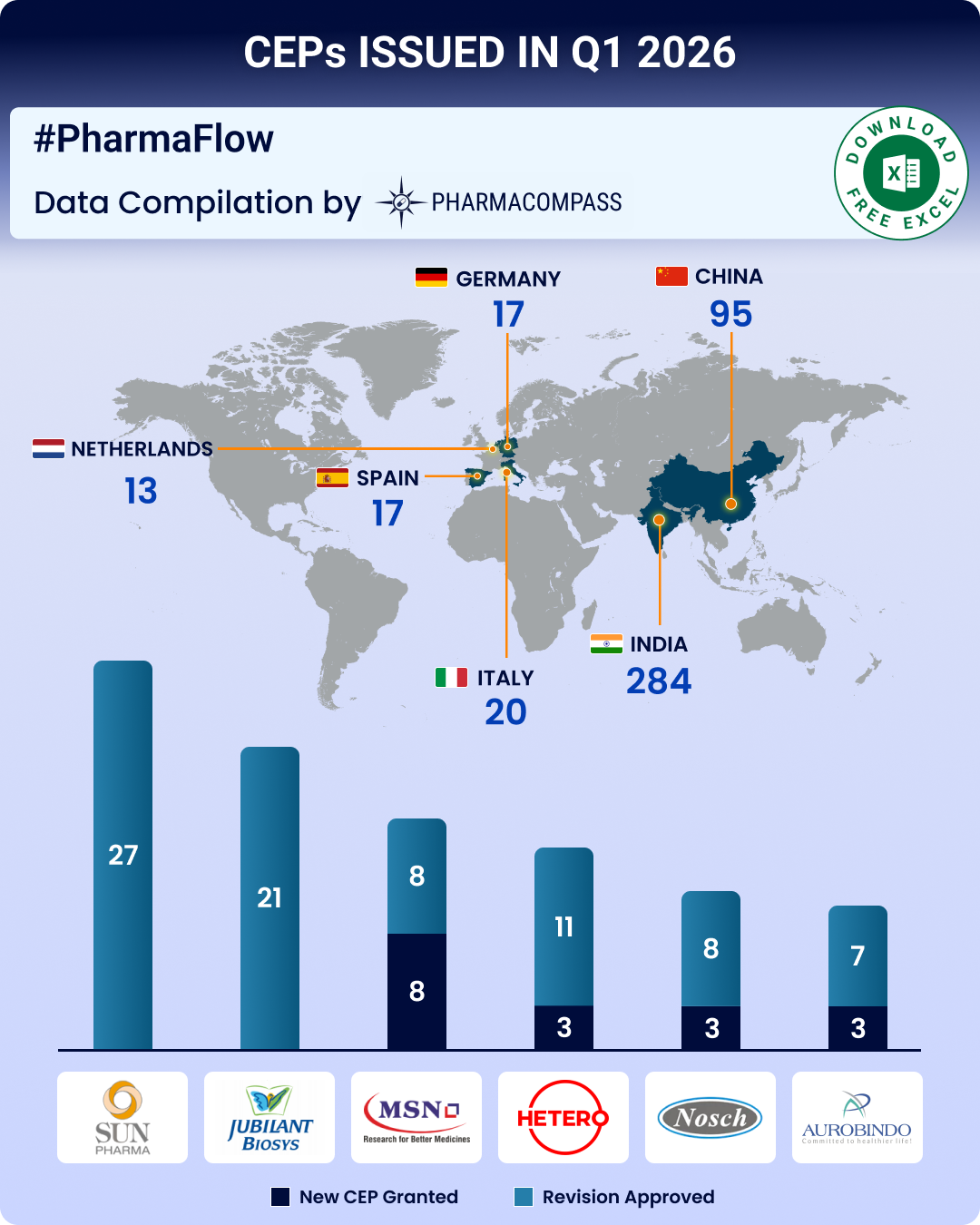
https://www.pharmacompass.com/radio-compass-blog/cep-q1-2026-update-cep-2-0-edqm-s-new-guidelines-strengthen-ecosystem-indian-firms-top-list-of-ceps-issued

03 Mar 2026
// PRESS RELEASE

24 Feb 2026
// PRESS RELEASE

30 Jan 2026
// PRESS RELEASE

29 Jan 2026
// PRESS RELEASE

12 Nov 2025
// PRESS RELEASE

09 Sep 2025
// PRESS RELEASE
Services
API Manufacturing
API & Drug Product Development
ABOUT THIS PAGE
Chunghwa Chemical Synthesis & Biotech is a supplier offers 51 products (APIs, Excipients or Intermediates).
Find Tacrolimus bulk with DMF, CEP, JDMF offered by Chunghwa Chemical Synthesis & Biotech
Find Everolimus bulk with DMF, CEP offered by Chunghwa Chemical Synthesis & Biotech
Find Mycophenolate Sodium bulk with DMF, CEP offered by Chunghwa Chemical Synthesis & Biotech
Find Sirolimus bulk with DMF, JDMF offered by Chunghwa Chemical Synthesis & Biotech
Find Pravastatin bulk with JDMF offered by Chunghwa Chemical Synthesis & Biotech
Find Baricitinib bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Brivaracetam bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Caspofungin bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Dexbrompheniramine Maleate bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Difelikefalin Acetate bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Eltrombopag bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Eslicarbazepine Acetate bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Everolimus bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Icosapent Ethyl bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Methocarbamol bulk with JDMF offered by Chunghwa Chemical Synthesis & Biotech
Find Midostaurin bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Mycophenolate Mofetil bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Mycophenolate Mofetil Hydrochloride bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Ozanimod bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Palbociclib bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Plecanatide bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Ribociclib bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Selumetinib bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Setmelanotide Acetate bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Tacrolimus bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Tofacitinib Citrate bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Trandolapril bulk with JDMF offered by Chunghwa Chemical Synthesis & Biotech
Find Trilaciclib Dihydrochloride bulk with DMF offered by Chunghwa Chemical Synthesis & Biotech
Find Abaloparatide bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Carfilzomib bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Dalbavancin bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Edoxaban Tosylate bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Etelcalcetide bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Everolimus bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Favipiravir bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Guaifenesin bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Ixazomib Citrate bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Lanreotide Acetate bulk offered by Chunghwa Chemical Synthesis & Biotech
Find LCZ 696 bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Metaxalone bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Methocarbamol bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Mycophenolic Acid bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Ozanimod bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Perindopril bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Ribociclib bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Ruxolitinib Phosphate bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Sacubitril Sodium bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Semaglutide bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Sugammadex Sodium bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Tenapanor Hydrochloride bulk offered by Chunghwa Chemical Synthesis & Biotech
Find Trilaciclib Dihydrochloride bulk offered by Chunghwa Chemical Synthesis & Biotech


 Chunghwa Chemical Synthesis & Biotech
Chunghwa Chemical Synthesis & Biotech