
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Australia

0
South Africa

DRUG PRODUCT COMPOSITIONS
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
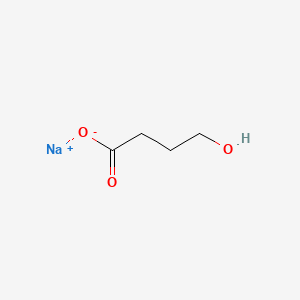

1. 4 Hydroxybutyrate Sodium
2. 4-hydroxybutyrate Sodium
3. Gamma Hydroxybutyrate
4. Gamma-hydroxybutyrate
5. Oxybate Sodium
6. Oxybate, Sodium
7. Oxybutyrate, Sodium
8. Sodium Gamma Hydroxybutyrate
9. Sodium Gamma-hydroxybutyrate
10. Sodium Oxybutyrate
11. Somsanit
12. Xyrem
1. 502-85-2
2. Sodium 4-hydroxybutyrate
3. Anetamin
4. 4-hydroxybutyric Acid Sodium Salt
5. Sodium 4-hydroxybutanoate
6. Somsanit
7. Oxybate Sodium
8. Gamma Oh
9. Sodium Oxybate [usan]
10. Sodium Oxybutyrate
11. Catabate
12. Sodium Oxybat
13. Oxybate (sodium)
14. 4-hydroxybutyrate Sodium
15. Wy-3478
16. Butanoic Acid, 4-hydroxy-, Monosodium Salt
17. Gamma-hydroxybutyrate Sodium
18. Sodium Gamma-hydroxybutyrate
19. Sodium;4-hydroxybutanoate
20. Gamma-hydroxy Sodium Butyrate
21. Butanoic Acid, 4-hydroxy-, Sodium Salt
22. Eb 27
23. Hydroxybutyric Acid Monosodium Salt
24. 4-hydroxybutyric Acid Monosodium Salt
25. Wy 3478
26. .gamma.-hydroxybutyrate Sodium Salt
27. Nsc-84223
28. Sodium Oxybate (usan)
29. 7g33012534
30. Butyric Acid, 4-hydroxy-, Monosodium Salt
31. Butyric Acid, 4-hydroxy-, Sodium Salt
32. Natrium 4-hydroxybutyrat
33. Sodium-4-hydroxybutyrate
34. Sodium Oxybate (sodium 4-hydroxybutanoate)
35. Ghb Sodium Salt (sodium Gammahydroxybutyrate)
36. 4-hydroxybuttersaeure Natriumsalz
37. Gamma-hydroxybutyrate Sodium Salt
38. Ncgc00247714-01
39. Einecs 207-953-3
40. Nsc 84223
41. Gamma-hydroxybutyric Acid, Sodium Salt
42. Jzp-6
43. Sodium4-hydroxybutyrate
44. Acetamide,2,2-dichloro-n-[(1r,2r)-2-hydroxy-1-(hydroxymethyl)-2-(4-nitrophenyl)ethyl]-,rel-
45. Unii-7g33012534
46. Xyrem (tn)
47. Sodium Gammahydroxybutyrate
48. Dsstox_cid_28866
49. Dsstox_rid_83135
50. Dsstox_gsid_48940
51. Schembl61823
52. Sodium Oxybate [hsdb]
53. Sodium Oxybate [vandf]
54. Dea No. 2012
55. Sodium Oxybate [mart.]
56. Chembl1200682
57. Dtxsid3048940
58. Oxybate Sodium [who-dd]
59. Sodium Oxybate [ema Epar]
60. Hms2091e15
61. Hy-b1187
62. Tox21_112871
63. Sodium Oxybate [orange Book]
64. Gamma Hydroxybutyric Acid Preparations
65. Xywav Component Sodium Oxybate
66. Akos006221428
67. Ccg-212465
68. Cs-4796
69. Db09072
70. Sodium Oxybate Component Of Xywav
71. Cas-502-85-2
72. Db-071156
73. Ft-0626615
74. D05866
75. H-4040
76. Sodium Salt Of Gamma-hydroxybutyric Acid
77. .gamma.-hydroxybutyrate Sodium Salt [mi]
78. Butanoic Acid, 4-hydroxy-, Sodium Salt (1:1)
79. Q7553347
80. 90318-49-3
| Molecular Weight | 126.09 g/mol |
|---|---|
| Molecular Formula | C4H7NaO3 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 3 |
| Exact Mass | 126.02928836 g/mol |
| Monoisotopic Mass | 126.02928836 g/mol |
| Topological Polar Surface Area | 60.4 Ų |
| Heavy Atom Count | 8 |
| Formal Charge | 0 |
| Complexity | 64.3 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 2 | |
|---|---|
| Drug Name | Xyrem |
| PubMed Health | Sodium Oxybate (By mouth) |
| Drug Classes | Central Nervous System Agent |
| Drug Label | Sodium oxybate, a CNS depressant, is the active ingredient in Xyrem. The chemical name for sodium oxybate is sodium 4-hydroxybutyrate. The molecular formula is C4H7NaO3, and the molecular weight is 126.09 g/mole. The chemical structure is:Sodium oxyb... |
| Active Ingredient | Sodium oxybate |
| Dosage Form | Solution |
| Route | Oral |
| Strength | 500mg/ml |
| Market Status | Prescription |
| Company | Jazz Pharms |
| 2 of 2 | |
|---|---|
| Drug Name | Xyrem |
| PubMed Health | Sodium Oxybate (By mouth) |
| Drug Classes | Central Nervous System Agent |
| Drug Label | Sodium oxybate, a CNS depressant, is the active ingredient in Xyrem. The chemical name for sodium oxybate is sodium 4-hydroxybutyrate. The molecular formula is C4H7NaO3, and the molecular weight is 126.09 g/mole. The chemical structure is:Sodium oxyb... |
| Active Ingredient | Sodium oxybate |
| Dosage Form | Solution |
| Route | Oral |
| Strength | 500mg/ml |
| Market Status | Prescription |
| Company | Jazz Pharms |
For the treatment of cataplexy and excessive daytime sleepiness (EDS) associated with narcolepsy.
FDA Label
Treatment of narcolepsy with cataplexy in adult patients.
Substitution treatment for alcohol dependence within a framework of careful medical supervision along with continuous psychosocial support and social rehabilitation. Treatment should be initiated only in patients resistant to existing interventions or in patients for whom existing therapies are contra-indicated or not recommended. ,
Sodium oxybate works through an unknown mechanism to treat narcolepsy by inducing sleep within about 5-15 minutes of administration.
Adjuvants, Anesthesia
Agents that are administered in association with anesthetics to increase effectiveness, improve delivery, or decrease required dosage. (See all compounds classified as Adjuvants, Anesthesia.)
Anesthetics, Intravenous
Ultrashort-acting anesthetics that are used for induction. Loss of consciousness is rapid and induction is pleasant, but there is no muscle relaxation and reflexes frequently are not reduced adequately. Repeated administration results in accumulation and prolongs the recovery time. Since these agents have little if any analgesic activity, they are seldom used alone except in brief minor procedures. (From AMA Drug Evaluations Annual, 1994, p174) (See all compounds classified as Anesthetics, Intravenous.)
N07XX04
N07BB
B05XA02
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
N - Nervous system
N01 - Anesthetics
N01A - Anesthetics, general
N01AX - Other general anesthetics
N01AX11 - Sodium oxybate
N - Nervous system
N07 - Other nervous system drugs
N07X - Other nervous system drugs
N07XX - Other nervous system drugs
N07XX04 - Sodium oxybate
Absorption
Absolute bioavailability is approximately 88%. Tmax of 30.7-51.9min.
Route of Elimination
The major metabolite is carbon dioxide which is cleared by expiration, less then 5% appears as the unchanged drug in the urine within 6-8 hours after dosing.
Volume of Distribution
Vd of 37.7-67.7
Clearance
Total clearance of 895-1361mL/min.
Animal studies indicate that the major elimination pathway is metabolism by the creation of carbon dioxide and water through the Krebs cycle and secondarily by beta-oxidation. In the primary pathway hydroxyacid-oxoacid transhydrogenase catalyzes the conversion of sodium oxybate to succinic semialdehyde which is then transformed to succinic acid by succinic semmialdehyde dehydrogenase. Succinic acid is then turned into carbon dioxide and water in the Krebs cycle. Succinic semialdehyde is also metabolised into carbon dioxide and water by a transhydrofenase in the presence of alpha ketoglutarate.
0.5 to 1 hour.
The exact mechanism of action is unknown. It is the sodium salt of the endogenous compound gamma hydroxybutyrate which is a metabolite of the GABA neurotransmitter and it's thought that it's therapeutic effects are mediated via GABA B actions at noradrenergic, dopaminergic and thalamocortical neurons.

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
65
PharmaCompass offers a list of Gamma-Hydroxybutyric Acid Sodium API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Gamma-Hydroxybutyric Acid Sodium manufacturer or Gamma-Hydroxybutyric Acid Sodium supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Gamma-Hydroxybutyric Acid Sodium manufacturer or Gamma-Hydroxybutyric Acid Sodium supplier.
PharmaCompass also assists you with knowing the Gamma-Hydroxybutyric Acid Sodium API Price utilized in the formulation of products. Gamma-Hydroxybutyric Acid Sodium API Price is not always fixed or binding as the Gamma-Hydroxybutyric Acid Sodium Price is obtained through a variety of data sources. The Gamma-Hydroxybutyric Acid Sodium Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Sodium Oxybate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Sodium Oxybate, including repackagers and relabelers. The FDA regulates Sodium Oxybate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Sodium Oxybate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Sodium Oxybate manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Sodium Oxybate supplier is an individual or a company that provides Sodium Oxybate active pharmaceutical ingredient (API) or Sodium Oxybate finished formulations upon request. The Sodium Oxybate suppliers may include Sodium Oxybate API manufacturers, exporters, distributors and traders.
click here to find a list of Sodium Oxybate suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Sodium Oxybate DMF (Drug Master File) is a document detailing the whole manufacturing process of Sodium Oxybate active pharmaceutical ingredient (API) in detail. Different forms of Sodium Oxybate DMFs exist exist since differing nations have different regulations, such as Sodium Oxybate USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Sodium Oxybate DMF submitted to regulatory agencies in the US is known as a USDMF. Sodium Oxybate USDMF includes data on Sodium Oxybate's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Sodium Oxybate USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Sodium Oxybate suppliers with USDMF on PharmaCompass.
A Sodium Oxybate written confirmation (Sodium Oxybate WC) is an official document issued by a regulatory agency to a Sodium Oxybate manufacturer, verifying that the manufacturing facility of a Sodium Oxybate active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Sodium Oxybate APIs or Sodium Oxybate finished pharmaceutical products to another nation, regulatory agencies frequently require a Sodium Oxybate WC (written confirmation) as part of the regulatory process.
click here to find a list of Sodium Oxybate suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Sodium Oxybate as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Sodium Oxybate API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Sodium Oxybate as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Sodium Oxybate and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Sodium Oxybate NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Sodium Oxybate suppliers with NDC on PharmaCompass.
Sodium Oxybate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Sodium Oxybate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Sodium Oxybate GMP manufacturer or Sodium Oxybate GMP API supplier for your needs.
A Sodium Oxybate CoA (Certificate of Analysis) is a formal document that attests to Sodium Oxybate's compliance with Sodium Oxybate specifications and serves as a tool for batch-level quality control.
Sodium Oxybate CoA mostly includes findings from lab analyses of a specific batch. For each Sodium Oxybate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Sodium Oxybate may be tested according to a variety of international standards, such as European Pharmacopoeia (Sodium Oxybate EP), Sodium Oxybate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Sodium Oxybate USP).
