
Synopsis
0
CEP/COS
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Europe
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
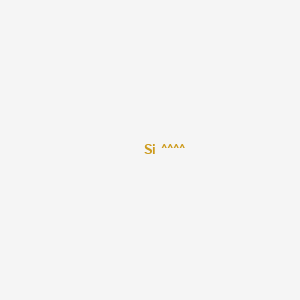
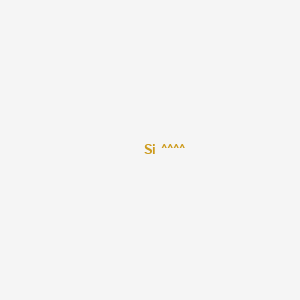
1. Silicon 28
2. Silicon-28
3. Silicone
4. Silicones
1. 7440-21-3
2. Si
3. Silicone
4. Silicon Powder
5. Z4152n8iui
6. Mfcd00085311
7. 90337-93-2
8. Silicon Metal
9. Defoamer S-10
10. Silicon, Isotope Ofmass 32
11. Flots 100sco
12. Silicio
13. Silicon Dust
14. Silicon, Elemental
15. Silicon Powder, Crystalline, -325 Mesh
16. Polyeristalline Silicon Powder
17. Ccris 6599
18. Ccris 6831
19. Silicon Powder, Crystalline, -100+200 Mesh
20. Hsdb 5033
21. Hsdb 6351
22. Einecs 231-130-8
23. Einecs 232-263-4
24. Un1346
25. Un2203
26. Unii-z4152n8iui
27. Silicium
28. Silizium
29. Silicon Atom
30. Silicon Chips
31. Silicon, Disks, 15.9mm, Thickness 0.38mm, Single Crystal, N-type, 100%
32. Silicon Nanopowder
33. Silane, Compressed
34. Silicon, Cp
35. Unii-5j076063r1
36. Silicon Random Pieces
37. Silicon [inci]
38. Silicon, 99.0%
39. Silane [hsdb]
40. Silicon [ii]
41. Silicon [mi]
42. Silicon [mart.]
43. Silicon Powder, Amorphous
44. Silane [mi]
45. Silicon [who-dd]
46. 14si
47. Silicon Powder (amorphous)
48. Ec 231-130-8
49. Ec 232-263-4
50. Silicon (si), >98%
51. Dtxsid0051441
52. Dtxsid6052534
53. Silicon Powder (nanocrystalline)
54. Chebi:27573
55. Silicon, Elemental [hsdb]
56. Amy37004
57. Silicon Granular, Virgin Poly Fines
58. Silicon, Containing More Than 99.99 Per Cent By Weight Of Silicon
59. Akos004909675
60. Akos024438062
61. Silicon Rod, 2.5cm (0.98in) Dia
62. Silicon Rod, 5.08cm (2.0in) Dia
63. Db12982
64. Silicon Lump, 10cm (3.9in) & Down
65. Silicon Lump, 15cm (5.9in) & Down
66. Silicon Metal, Nist(r) Srm(r) 57b
67. Silicon Plate, 12.5mm (0.5in) Thick
68. Silicon Plate, 9.5mm (0.37in) Thick
69. Silicon Powder, Crystalline, -100 Mesh
70. Q670
71. Silicon Plate, 6.35mm (0.25in) Thick
72. Silicon Powder, Crystalline, Aps <=50nm
73. Silicon Powder, Nodular, Aps 1-5 Micron
74. Silicon Rod, 0.828cm (0.326in) Dia
75. Silicon Powder, Crystalline, Aps C=100nm
76. Silicon Powder, Crystalline, -100+325 Mesh
77. Silicon, Pieces, 99.95% Trace Metals Basis
78. Silicon Lump, 0.1-2.5cm (0.04-1.0in)
79. Silane, Compressed [un2203] [flammable Gas]
80. 803s625
81. Silicon Metal Powder Si Grade Ax 05 (h?gan?s)
82. Silicon Metal Powder Si Grade Ax 10 (h?gan?s)
83. Silicon Metal Powder Si Grade Ax 20 (h?gan?s)
84. Polysilicon, Monocrystalline Silicon, Silicon Ingots
85. Silicon Standard For Aas, Ready-to-use, In Hno3: Hf
86. Silicon, Powder, -325 Mesh, 99% Trace Metals Basis
87. Silicon Powder, Amorphous [un1346] [flammable Solid]
88. Polydispersed Silicon Nanowires, D X L 40 Nm X 1-20 Mum
89. Silicon, Aas Standard Solution, Specpure?, Si 1000?g/ml
90. Silicon, Powder, -60 Mesh, 99.999% Trace Metals Basis
91. Silicon, Rod, 100mm, Diameter 2.0mm, Crystalline, 100%
92. Silicon, Rod, 100mm, Diameter 25mm, Crystalline, 100%
93. Silicon, Rod, 100mm, Diameter 5.0mm, Crystalline, 100%
94. Silicon, Rod, 10mm, Diameter 2.0mm, Crystalline, 100%
95. Silicon, Rod, 25mm, Diameter 2.0mm, Crystalline, 100%
96. Silicon, Rod, 50mm, Diameter 2.0mm, Crystalline, 100%
97. Silicon, Rod, 50mm, Diameter 5.0mm, Crystalline, 100%
98. Silicon, Single Crystal, -111, Size, Thickness 0.4 Mm
99. Silicon Rod, Polycrystalline, Unpolished, 1.6cm (0.6in) Dia
100. Silicon, Nanopowder, <100 Nm (bet), <3% Oxygen Passivation
101. Silicon, Oil Based Standard Solution, Specpure, Si 5000g/g
102. Silicon, Oil Based Standard Solution, Specpure?, Si 1000?g/g
103. Silicon, Plasma Standard Solution, Specpure?, Si 10,000?g/ml
104. Silicon, Plasma Standard Solution, Specpure?, Si 1000?g/ml
105. Silicon, Rod, 20 Mm Diameter, Length 50 Mm, Purity 99.999%
106. Silicon, Rod, 20 Mm Diameter, Length 80 Mm, Purity 99.999%
107. Silicon Optical Window, 25.4mm (1.0in) Dia X 1mm (0.04in) Thick
108. Silicon Slugs, 9.5mm (0.375in) Dia X 9.5mm (0.375in) Length
109. Silicon, Disks, 13mm, Thickness 0.38mm, Single Crystal, 100%
110. Silicon, Lump, 25 Mm Max. Lump Size, Weight 50 G, Purity 99.95%
111. Silicon, Lump, 25 Mm Max. Lump Size, Weight 50 G, Purity 99.99%
112. Silicon, Lump, 5 Mm Max. Lump Size, Weight 100 G, Purity 99.99%
113. Silicon, Lump, 5 Mm Max. Lump Size, Weight 200 G, Purity 99.99%
114. Silicon, Lump, 5 Mm Max. Lump Size, Weight 50 G, Purity 99.99%
115. Silicon, Lump, 5 Mm Max. Lump Size, Weight 500 G, Purity 99.99%
116. Silicon, Rod, 25 Mm Diameter, Length 150 Mm, Purity 99.999%
117. Silicon, Rod, Length 100 Mm, 20 Mm Diameter, Purity 99.999%
118. Silicon, Sphere, 0.48 Mm Diameter, 10_pcs, Tolerance +/-2.5um
119. Silicon, Sphere, 0.48 Mm Diameter, 20 Pcs, Tolerance +/-2.5um
120. Silicon, Sphere, 0.48 Mm Diameter, 50 Pcs, Tolerance +/-2.5um
121. Silicon, Sphere, 10pcs, Diameter 2.0mm, Precision Sphere Grade 25
122. Silicon, Sphere, 20pcs, Diameter 2.0mm, Precision Sphere Grade 25
123. Silicon, Sphere, 50pcs, Diameter 2.0mm, Precision Sphere Grade 25
124. Silicon 99.999% Polycrystalinedisc And Sputtering Targets Available Also
125. Silicon Sputtering Target, 50.8mm (2.0in) Dia X 3.18mm (0.125in) Thick
126. Silicon Sputtering Target, 50.8mm (2.0in) Dia X 6.35mm (0.250in) Thick
127. Silicon Sputtering Target, 76.2mm (3.0in) Dia X 6.35mm (0.250in) Thick
128. Silicon, Lump, 25 Mm Max. Lump Size, Weight 100 G, Purity 99.95%
129. Silicon, Lump, 25 Mm Max. Lump Size, Weight 100 G, Purity 99.99%
130. Silicon, Lump, 25 Mm Max. Lump Size, Weight 100 G, Purity 99.999%
131. Silicon, Lump, 25 Mm Max. Lump Size, Weight 200 G, Purity 99.95%
132. Silicon, Lump, 25 Mm Max. Lump Size, Weight 200 G, Purity 99.99%
133. Silicon, Lump, 25 Mm Max. Lump Size, Weight 200 G, Purity 99.999%
134. Silicon, Lump, 25 Mm Max. Lump Size, Weight 50 G, Purity 99.999%
135. Silicon, Lump, 25 Mm Max. Lump Size, Weight 500 G, Purity 99.95%
136. Silicon, Lump, 25 Mm Max. Lump Size, Weight 500 G, Purity 99.99%
137. Silicon, Powder, 45 Max. Part. Size (micron), Weight 50 G, Purity 99.5%
138. Silicon, Rod, 40mm, Diameter 20mm, Single Crystal, -100, 99.999%
139. Silicon, Rod, 40mm, Diameter 20mm, Single Crystal, -111, 99.999%
140. Silicon, Rod, 50mm, Diameter 5mm, Single Crystal, -100, 99.999%
141. Silicon, Rod, 50mm, Diameter 5mm, Single Crystal, -111, 99.999%
142. Silicon, Rod, 50mm, Diameter 6mm, Single Crystal, -111, 99.999%
143. Silicon, Rod, 80mm, Diameter 20mm, Single Crystal, -100, 99.999%
144. Silicon, Sheet, 10x10mm, Thickness 0.5mm, Single Crystal, -100, 100%
145. Silicon, Sheet, 10x10mm, Thickness 0.6mm, Single Crystal, -100, 100%
146. Silicon, Sheet, 14x14mm, Thickness 1.0mm, Single Crystal, -100, 100%
147. Silicon, Sheet, 14x14mm, Thickness 1.0mm, Single Crystal, -111, 100%
148. Silicon, Sheet, 25x25mm, Thickness 1.0mm, Polycrystalline, 99.999%
149. Silicon, Sheet, 25x25mm, Thickness 1.0mm, Single Crystal, -100, 100%
150. Silicon, Sheet, 40x40mm, Thickness 3.0mm, Single Crystal, P-type, 100%
151. Silicon, Sheet, 52x52mm, Thickness 1.0mm, Polycrystalline, 99.999%
152. Silicon, Nanopowder, <100 Nm Particle Size (tem), >=98% Trace Metals Basis
153. Silicon, Powder, 150 Max. Part. Size (micron), Weight 1000 G, Purity 97.5%
154. Silicon, Powder, 150 Max. Part. Size (micron), Weight 200 G, Purity 97.5%
155. Silicon, Powder, 150 Max. Part. Size (micron), Weight 2000 G, Purity 97.5%
156. Silicon, Powder, 45 Max. Part. Size (micron), Purity 99.95%, Weight 200 G
157. Silicon, Powder, 45 Max. Part. Size (micron), Weight 10 G, High Purity 99.999%
158. Silicon, Powder, 45 Max. Part. Size (micron), Weight 100 G, High Purity 99.998%
159. Silicon, Powder, 45 Max. Part. Size (micron), Weight 100 G, Purity 99.95%
160. Silicon, Powder, 45 Max. Part. Size (micron), Weight 20 G, High Purity 99.998%
161. Silicon, Powder, 45 Max. Part. Size (micron), Weight 50 G, Purity 99.95%
162. Silicon, Powder, Max. Particle Size 150 Micron, Weight 100 G, Purity 97.5%
163. Silicon, Powder, Max. Particle Size 150 Micron, Weight 500 G, Purity 97.5%
164. Silicon, Powder, Max. Particle Size 45 Micron, Weight 100 G, Purity 99.5%
165. Silicon, Powder, Max. Particle Size 45 Micron, Weight 200 G, Purity 99.5%
166. Silicon, Powder, Max. Particle Size 45 Micron, Weight 50 G, High Purity 99.998%
167. Silicon, Rod, 100mm, Diameter 10.0mm, Single Crystal - Random Orientation, 100%
168. Silicon, Rod, 100mm, Diameter 12.7mm, Single Crystal - Random Orientation, 100%
169. Silicon, Rod, 25mm, Diameter 3.15mm, Single Crystal - Random Orientation, 100%
170. Silicon, Rod, 50mm, Diameter 3.15mm, Single Crystal - Random Orientation, 100%
171. Silicon, Single Crystal, -111, P-type, Boron Doped, Diameter 57mm, Length 1.0mm, 100%
172. Silicon, Nist(r) Srm(r) 640e, Powder, Line Position And Line Shape Standard For Powder Diffraction
173. Silicon, Single Crystal, -100, N-type, Phosphorus Doped, Diameter 150mm, Length 0.65mm, 100%
174. Silicon, Single Crystal, -100, N-type, Phosphorus Doped, Diameter 76.2mm, Length 0.38mm, 100%
175. Silicon, Single Crystal, -100, N-type, Phosphorus Doped, Diameter 76.2mm, Length 2.0mm, 100%
176. Silicon, Single Crystal, -100, P-type, Boron Doped, Diameter 76.2mm, Length 0.38mm, 100%
177. Silicon, Single Crystal, -111, N-type, Phosphorus Doped, Diameter 76.2mm, Length 0.38mm, 100%
178. Silicon, Single Crystal, -111, N-type, Phosphorus Doped, Diameter 76.2mm, Length 0.50mm, 100%
179. Silicon, Single Crystal, -111, N-type, Phosphorus Doped, Diameter 76.2mm, Length 0.75mm, 100%
180. Silicon, Single Crystal, -111, P-type, Boron Doped, Diameter 76.2mm, Length 0.38mm, 100%
181. Silicon, Sputtering Target, Diam. X Thickness 2.00 In. X 0.25 In., 99.999% Trace Metals Basis
182. Silicon, Wafer (single Side Polished), <100>, N-type, Contains No Dopant, Diam. X Thickness 2 In. X 0.5 Mm
183. Silicon, Wafer (single Side Polished), <100>, N-type, Contains No Dopant, Diam. X Thickness 3 In. X 0.5 Mm
184. Silicon, Wafer (single Side Polished), <100>, N-type, Contains Phosphorus As Dopant, Diam. X Thickness 2 In. X 0.5 Mm
185. Silicon, Wafer (single Side Polished), <100>, N-type, Contains Phosphorus As Dopant, Diam. X Thickness 3 In. X 0.5 Mm
186. Silicon, Wafer (single Side Polished), <100>, P-type, Contains Boron As Dopant, Diam. X Thickness 2 In. X 0.5 Mm
187. Silicon, Wafer (single Side Polished), <100>, P-type, Contains Boron As Dopant, Diam. X Thickness 3 In. X 0.5 Mm
188. Silicon, Wafer (single Side Polished), <111>, N-type, Contains No Dopant, Diam. X Thickness 2 In. X 0.5 Mm
189. Silicon, Wafer (single Side Polished), <111>, N-type, Contains No Dopant, Diam. X Thickness 3 In. X 0.5 Mm
190. Silicon, Wafer (single Side Polished), <111>, P-type, Contains Boron As Dopant, Diam. X Thickness 3 In. X 0.5 Mm
191. Silicon, Wafer (single Side Polished), Contains Phosphorus As Dopant, <111>, N-type, Diam. X Thickness 2 In. X 0.5 Mm
192. Silicon, Wafer, <111>, P-type, Contains Boron As Dopant, Diam. X Thickness 2 In. X 0.3 Mm
| Molecular Weight | 28.085 g/mol |
|---|---|
| Molecular Formula | Si |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 0 |
| Rotatable Bond Count | 0 |
| Exact Mass | 27.976926534 g/mol |
| Monoisotopic Mass | 27.976926534 g/mol |
| Topological Polar Surface Area | 0 Ų |
| Heavy Atom Count | 1 |
| Formal Charge | 0 |
| Complexity | 0 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Trace Elements
A group of chemical elements that are needed in minute quantities for the proper growth, development, and physiology of an organism. (From McGraw-Hill Dictionary of Scientific and Technical Terms, 4th ed) (See all compounds classified as Trace Elements.)
In 36 patients suffering from chronic renal failure (mean creatinine clearance 26 mL/min), serum silicon levels were significantly increased (mean 0.52 microgram/mL compared with 0.265 microgram/mL in normals; p less than 0.005). Urinary silicon excretion per 24 hr was significantly decreased (15.71 mg/24 hr compared with 21.4 mg/24 hr in normals; p less than 0.001). Fractional excretion of silicon (FESi) was significantly increased in chronic renal failure (p less than 0.001), with overall tubular secretion of silicon in 33% of patients. Urinary excretion of silicon was significantly related to urinary calcium excretion (p less than 0.0001) urinary magnesium excretion (p less than 0.0001) creatinine clearance (p less than 0.05) and sodium excretion (p less than 0.05). It is suggested that urinary silicon is in the form of orthosilicate, principally bound to calcium and magnesium; and that in chronic renal failure the increase in FESi, and the decrease in absorbed Si from the gastrointestinal tract, moderate the increase in plasma silicon levels and prevent excessive entry of silicon into the tissues.
PMID:3748249 Adler AJ, Berlyne GM; Nephton 44 (1): 36-9 (1986)
The substance can be absorbed into the body by inhalation.
International Program on Chemical Safety/Commission of the European Communities; International Chemical Safety Card on Silicon (November 2003). Available from, as of May 20, 2010: https://www.inchem.org/pages/icsc.html
 Click Us!
Click Us! 
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 20262
Submission : 2007-02-07
Status : Active
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 23097
Submission : 2009-09-03
Status : Active
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 23967
Submission : 2010-07-12
Status : Active
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 25985
Submission : 2012-04-20
Status : Active
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 18419
Submission : 2005-06-07
Status : Active
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 18429
Submission : 2005-06-13
Status : Active
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 18431
Submission : 2005-06-15
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 20283
Submission : 2007-02-07
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 20838
Submission : 2007-09-03
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 21465
Submission : 2008-03-18
Status : Inactive
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Related Excipient Companies

Excipients by Applications
ABOUT THIS PAGE
69
PharmaCompass offers a list of Silicon API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Silicon manufacturer or Silicon supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Silicon manufacturer or Silicon supplier.
A Silicon manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Silicon, including repackagers and relabelers. The FDA regulates Silicon manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Silicon API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Silicon supplier is an individual or a company that provides Silicon active pharmaceutical ingredient (API) or Silicon finished formulations upon request. The Silicon suppliers may include Silicon API manufacturers, exporters, distributors and traders.
click here to find a list of Silicon suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Silicon DMF (Drug Master File) is a document detailing the whole manufacturing process of Silicon active pharmaceutical ingredient (API) in detail. Different forms of Silicon DMFs exist exist since differing nations have different regulations, such as Silicon USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Silicon DMF submitted to regulatory agencies in the US is known as a USDMF. Silicon USDMF includes data on Silicon's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Silicon USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Silicon suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Silicon Drug Master File in Japan (Silicon JDMF) empowers Silicon API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Silicon JDMF during the approval evaluation for pharmaceutical products. At the time of Silicon JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Silicon suppliers with JDMF on PharmaCompass.
Silicon Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Silicon GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Silicon GMP manufacturer or Silicon GMP API supplier for your needs.
A Silicon CoA (Certificate of Analysis) is a formal document that attests to Silicon's compliance with Silicon specifications and serves as a tool for batch-level quality control.
Silicon CoA mostly includes findings from lab analyses of a specific batch. For each Silicon CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Silicon may be tested according to a variety of international standards, such as European Pharmacopoeia (Silicon EP), Silicon JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Silicon USP).

