
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
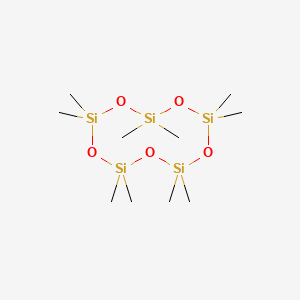

1. D5-sil
1. 541-02-6
2. Cyclopentasiloxane, Decamethyl-
3. Cyclomethicone 5
4. 2,2,4,4,6,6,8,8,10,10-decamethyl-1,3,5,7,9,2,4,6,8,10-pentaoxapentasilecane
5. Dimethylsiloxane Pentamer
6. Dekamethylcyklopentasiloxan
7. Cyclomethicone
8. Ciclopentasiloxane
9. Cyclomethicone D5
10. 0tht5pci0r
11. Ddecamethylcyclopentasiloxane
12. Dow Corning 345 Fluid
13. 2,2,4,4,6,6,8,8,10,10-decamethyl-1,3,5,7,9,2,4,6,8,10-pentoxapentasilecane
14. Dow Corning 345
15. Nuc Silicone Vs 7158
16. Silicon Sf 1202
17. Cyclopentasiloxane, 2,2,4,4,6,6,8,8,10,10-decamethyl-
18. Cyclic Dimethylsiloxane Pentamer
19. Mfcd00046966
20. Union Carbide 7158 Silicone Fluid
21. Kf 995
22. Vs 7158
23. D5-sil
24. Ccris 1328
25. Hsdb 5683
26. Dekamethylcyklopentasiloxan [czech]
27. Einecs 208-764-9
28. Unii-0tht5pci0r
29. Decamethyl Cyclopentasiloxane
30. Sf 1202
31. Brn 1800166
32. D5 Cyclomethicone
33. D5
34. Dimethylcyclopentasiloxane
35. Decamethylcylopentasiloxane
36. Dsstox_cid_7184
37. Jeesilc Cps-211
38. Ec 208-764-9
39. Dsstox_rid_78338
40. Dsstox_gsid_27184
41. Schembl28497
42. N-propylheptamethyltrisiloxane
43. Xiameter Pmx-0245
44. 4-04-00-04128 (beilstein Handbook Reference)
45. Cyclopentasiloxane (d5)
46. Chembl1885178
47. Dtxsid1027184
48. Cyclopentasiloxane [inci]
49. Chebi:191092
50. Decamethylcyclopentasiloxane, 97%
51. Cyclomethicone 5 [usp-rs]
52. Cyclomethicone 5 [who-dd]
53. Bcp15826
54. Tox21_303170
55. Cd3770
56. Kf-995
57. Akos008901199
58. Zinc169743678
59. Cs-w009767
60. Db11244
61. Dow Corning St Cyclomethicone 5
62. Decamethylcyclopentasiloxane [mi]
63. Ncgc00163981-01
64. Ncgc00257224-01
65. Octamethylcyclotetrasiloxane (d5)
66. As-59731
67. Cas-541-02-6
68. Decamethylcyclopentasiloxane [hsdb]
69. Kp-545 Component Cyclomethicone 5
70. D1890
71. D3770
72. Decamethylcyclopentasiloxane (cyclic Monomer)
73. Ft-0665531
74. Dow Corning Up-1002 Ultra Pure Fluid
75. D78203
76. S05475
77. Decamethylcyclopentasiloxane, Analytical Standard
78. Q414350
79. Decamethyl-1,3,5,7,9,2,4,6,8,10-pentaoxapentasilecane
80. Cyclomethicone 5, United States Pharmacopeia (usp) Reference Standard
81. 2,2,4,4,6,6,8,8,10,10-decamethyl-1,3,5,7,9,2,4,6,8,10-pentaoxapentasilecane #
82. D5 Cyclomethicone, Pharmaceutical Secondary Standard; Certified Reference Material
| Molecular Weight | 370.77 g/mol |
|---|---|
| Molecular Formula | C10H30O5Si5 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 0 |
| Exact Mass | 370.09395673 g/mol |
| Monoisotopic Mass | 370.09395673 g/mol |
| Topological Polar Surface Area | 46.2 Ų |
| Heavy Atom Count | 20 |
| Formal Charge | 0 |
| Complexity | 258 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
To assess the fate of D5 absorbed from the skin in intact animals, (14)C-D5 was applied to the dorsal surface of male and female rats. Hair at the application site was clipped prior to application and the application site was covered with a non-occlusive elastic wrap. The study was designed to permit differentiation between D5 exhaled after absorption and D5 evaporating from the application site. After application of D5, the animals were transferred to metabolic cages for the collection of urine and feces. The majority (about 85%) of the applied (14)C-D5 volatilized from the skin. After 96 hr, 0.35% of the administered D5 remained at the application site and less than 1% of the applied (14)C-activity was recovered in urine and carcass with trace levels of (14)C-activity recovered in feces, CO2 traps, and tissues. The total amount of D5 absorbed was <1%.
Dekant W, Klaunig JE; Toxicology of decamethylcyclopentasiloxane (D5), Regulatory Toxicology and Pharmacology. p.2 (2015)
Octamethylcyclotetrasiloxane (D4) and decamethylcyclopentasiloxane (D5) are cyclic siloxanes used as chemical intermediates with some applications in consumer products. The in vitro percutaneous absorption of (14)C-D4 and (14)C-D5 was studied in flow-through diffusion cells. Single doses were applied neat and in antiperspirant formulations to dermatomed human skin for 24 hr. The majority of applied D4 and D5 (approximately 90%) volatilized before being absorbed. Only 0.5% of applied D4 was absorbed while the absorption of D5 (0.04%) was one order of magnitude lower. The largest percentage (>90%) of the absorbed D4 and D5 was found in the skin. The fate of D4 and D5 absorbed in the skin was studied in rat in vivo. A single dose of (14)C-D4 (10, 4.8 and 2 mg/sq cm) and (14)C-D5 (10 mg/sq cm) was topically applied inside a dosing chamber attached to the dorsal area. Rats were housed in metabolism cages up to 24 hr to enable collection of urine, feces, expired/escaped volatiles. The majority of applied D4 or D5 had volatilized from the skin surface. Less than 1.0% of the applied D4 and only 0.2% of applied D5 was absorbed with approximately 60% of absorbed D4 and 30% of absorbed D5 reaching systemic compartments. The amount absorbed into the skin decreased with time showing that residual D4 and D5 diffused back to the skin surface and continued to evaporate. Overall, a low tendency to pass through the skin into systemic compartments was demonstrated for both D4 (< or = 0.5% of applied dose) and D5 (<0.1% of applied dose).
PMID:18178298 Jovanovic ML et al; Regul Toxicol Pharmacol 50 (2): 239-48 (2008)
Toxicokinetic studies were performed in rats after dosing by gavage with (14)C-D5 (single dose of 1000 mg/kg bw) dissolved in different vehicles (corn oil and simethicone fluid) and as a neat material. The carrier had a significant influence on the extent of absorption of (14)C-D5. After administration of neat D5, approximately 10% of the dose was absorbed from the gastrointestinal tract. Based on blood area under the curve (AUC), absorption increased after administration of D5 in corn oil and decreased after administration in simethicone fluid. Elimination half-lives for D5-associated radioactivity in blood ranged from 45 (simethicone) to 240 hr (corn oil), and were between 117 (neat) and 242 hr (simethicone) for parent D5. The radioactivity eliminated in the urine consisted entirely of polar metabolites of D5. Mass balance analysis indicated that approximately 60-80 % of the administered D5 was excreted unchanged in the feces, and up to 20% of administered D5 as water soluble metabolites in urine. Half of the systemically available D5 was eliminated as unchanged D5 in exhaled air. However, the kinetics and tissue distribution observed after oral dosing were qualitatively different from the distributions after inhalation or dermal exposures. Higher relative concentrations of D5 were noted in liver and spleen as compared to exposure to D5 by inhalation and dermal application.
Dekant W, Klaunig JE; Toxicology of decamethylcyclopentasiloxane (D5), Regulatory Toxicology and Pharmacology. p.2 (2015)
Separate 77-d fish feeding studies were conducted on the cyclic volatile methylsiloxane (cVMS) chemicals octamethylcyclotetrasiloxane and decamethylcyclopentasiloxane with the rainbow trout, Oncorhynchus mykiss, with the determination of biomagnification factor (BMF) and lipid-adjusted BMF (BMF(L)) values as the final experimental metrics. The studies used fish food concentrations of about 500 ug/g for exposure periods of 35 d, followed by a depuration period of 42 d with clean food. The fish tissue concentrations of D4 and D5 achieved empirical steady-state by day 21 in each study. By day 7 of exposure, total (14)C activity of both compounds had moved from the fish gastrointestinal (GI) tract into surrounding tissue. An absence of significant fish growth during the initial depuration phase allowed for measurement of empirical depuration rate constants (k2) independent of growth dilution for D4 and D5 of 0.035 and 0.040/d, respectively, corresponding to elimination half-lives of approximately 20d. These rate constants indicated that about 70-75% of steady-state was achieved during exposure in both studies, resulting in empirical steady-state BMF and BMF(L) values of 0.28 and 0.66 for D4, respectively, and 0.32 and 0.85 for D5, respectively. Kinetic modeling using simple first-order uptake and depuration dynamics produced good agreement with experimental data, with D4 and D5 assimilation efficiencies of 40% and 44%, respectively. Growth-corrected depuration rate constants modeled over the entire study data set indicated slower elimination kinetics for D4 (k2 of 0.007/d or half-life of 100 d) compared to D5 (k2 of 0.010/d or elimination half-life of 69 d). Kinetic BMFk values (i.e., k1/k2) for D4 and D5 were 1.7 and 1.3, respectively, with lipid-adjusted BMFk(L) values of 4.0 and 3.4, respectively.
PMID:23200569 Woodburn K et al; Chemosphere 93 (5): 779-88 (2013)
For more Absorption, Distribution and Excretion (Complete) data for DECAMETHYLCYCLOPENTASILOXANE (13 total), please visit the HSDB record page.
Major metabolites of D5 were identified in urine collected from Fischer (F-344) rats administered ... (14)C-D5 orally and via intravenous injection ... The metabolites of D5 are as follows: Me(2)Si(OH)(2), MeSi(OH)(3), MeSi(OH)(2)OSi(OH)(3), MeSi(OH)(2)OSi(OH)(2)Me, MeSi(OH)(2)OSi(OH)Me(2), Me(2)Si(OH)OSi(OH)Me(2), Me(2)Si(OH)OSiMe(2)OSi(OH)Me(2), nonamethylcyclopentasiloxanol, and hydroxymethylnonamethylcyclopentasiloxane. No parent ... D5 was present in urine. The presence of certain metabolites such as ... Me(2)Si(OH)(2) ... clearly established the occurrence of demethylation at the silicon-methyl bonds. Metabolites of the linear siloxane are structurally different from that obtained for cyclic siloxane except for the commonly present Me(2)Si(OH)(2). Mechanistic pathways for the formation of the metabolites were proposed.
PMID:12527702 Varaprath S et al; Drug Metab Dispos 31 (2): 206-14 (2003)
Toxicokinetic studies were performed in rats after dosing by gavage with (14)C-D5 (single dose of 1000 mg/kg bw) dissolved in different vehicles (corn oil and simethicone fluid) and as a neat material. The carrier had a significant influence on the extent of absorption of (14)C-D5. ... Half of the systemically available D5 was eliminated as unchanged D5 in exhaled air. ...
Dekant W, Klaunig JE; Toxicology of decamethylcyclopentasiloxane (D5), Regulatory Toxicology and Pharmacology. p.2 (2015)
Separate 77-d fish feeding studies were conducted on the cyclic volatile methylsiloxane (cVMS) chemicals octamethylcyclotetrasiloxane and decamethylcyclopentasiloxane with the rainbow trout, Oncorhynchus mykiss, with the determination of biomagnification factor (BMF) and lipid-adjusted BMF (BMF(L)) values as the final experimental metrics. The studies used fish food concentrations of about 500 ug/g for exposure periods of 35 d, followed by a depuration period of 42 d with clean food. ... An absence of significant fish growth during the initial depuration phase allowed for measurement of empirical depuration rate constants (k2) independent of growth dilution for D4 and D5 of 0.035 and 0.040/d, respectively, corresponding to elimination half-lives of approximately 20d.
PMID:23200569 Woodburn K et al; Chemosphere 93 (5): 779-88 (2013)
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
68
PharmaCompass offers a list of Decamethylcyclopentasiloxane API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Decamethylcyclopentasiloxane manufacturer or Decamethylcyclopentasiloxane supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Decamethylcyclopentasiloxane manufacturer or Decamethylcyclopentasiloxane supplier.
A Decamethylcyclopentasiloxane manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Decamethylcyclopentasiloxane, including repackagers and relabelers. The FDA regulates Decamethylcyclopentasiloxane manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Decamethylcyclopentasiloxane API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Decamethylcyclopentasiloxane supplier is an individual or a company that provides Decamethylcyclopentasiloxane active pharmaceutical ingredient (API) or Decamethylcyclopentasiloxane finished formulations upon request. The Decamethylcyclopentasiloxane suppliers may include Decamethylcyclopentasiloxane API manufacturers, exporters, distributors and traders.
click here to find a list of Decamethylcyclopentasiloxane suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Decamethylcyclopentasiloxane DMF (Drug Master File) is a document detailing the whole manufacturing process of Decamethylcyclopentasiloxane active pharmaceutical ingredient (API) in detail. Different forms of Decamethylcyclopentasiloxane DMFs exist exist since differing nations have different regulations, such as Decamethylcyclopentasiloxane USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Decamethylcyclopentasiloxane DMF submitted to regulatory agencies in the US is known as a USDMF. Decamethylcyclopentasiloxane USDMF includes data on Decamethylcyclopentasiloxane's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Decamethylcyclopentasiloxane USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Decamethylcyclopentasiloxane suppliers with USDMF on PharmaCompass.
Decamethylcyclopentasiloxane Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Decamethylcyclopentasiloxane GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Decamethylcyclopentasiloxane GMP manufacturer or Decamethylcyclopentasiloxane GMP API supplier for your needs.
A Decamethylcyclopentasiloxane CoA (Certificate of Analysis) is a formal document that attests to Decamethylcyclopentasiloxane's compliance with Decamethylcyclopentasiloxane specifications and serves as a tool for batch-level quality control.
Decamethylcyclopentasiloxane CoA mostly includes findings from lab analyses of a specific batch. For each Decamethylcyclopentasiloxane CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Decamethylcyclopentasiloxane may be tested according to a variety of international standards, such as European Pharmacopoeia (Decamethylcyclopentasiloxane EP), Decamethylcyclopentasiloxane JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Decamethylcyclopentasiloxane USP).

