
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Canada
0
Australia
0
South Africa
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
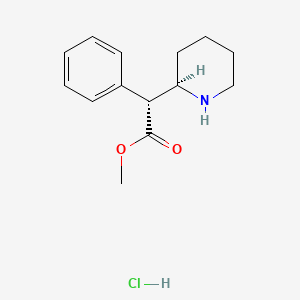

1. Dexmethylphenidate
2. Focalin
3. Focalin Xr
4. Hydrochloride, Dexmethylphenidate
5. Xr, Focalin
1. Dexmethylphenidate Hcl
2. Focalin
3. D-threo-methylphenidate Hydrochloride
4. 19262-68-1
5. Focalin Xr
6. 23655-65-4
7. Metadate Cd
8. Metadate Er
9. Equasym Xl
10. Dexmethylphenidate Hydrochloride [usan]
11. Ritalin
12. 4b3sc438hi
13. Dex-methylphenidate Hydrochloride
14. Methyl (2r)-2-phenyl-2-[(2r)-piperidin-2-yl]acetate;hydrochloride
15. 1678ok0e08
16. Methylphenidate D-threo-form Hydrochloride
17. Methyl (2r)-phenyl((2r)-piperidin-2-yl)acetate Hydrochloride
18. 19262-68-1 (hcl)
19. Methyl (2r)-2-phenyl-2-[(2r)-piperidin-1-ium-2-yl]acetate;chloride
20. Quillichew
21. Quillivant
22. Aptensio
23. Foquest
24. Oros Mph
25. Aptensio Xr
26. Methylin Er
27. Ritalin La
28. Ritalin Sr
29. Chembl904
30. (r)-methyl 2-phenyl-2-((r)-piperidin-2-yl)acetate Hydrochloride
31. Daytrana
32. Dexmethylphenidate Hydrochloride (usan)
33. Spd544
34. Spd-544
35. Dl-threo-methylphenidate Hcl
36. Biphentin
37. Equasym
38. Methypatch
39. Metadate Mr
40. Unii-1678ok0e08
41. Equasym Ir
42. Ritalin Qd
43. Focalin (tn)
44. Dl-threo-methylphenidate Hydrochloride
45. Adhansia Xr
46. Jornay Pm
47. Methyl .alpha.-phenyl-2-piperidineacetate Hydrochloride
48. Unii-4b3sc438hi
49. Nwp-06
50. Dtxsid50940927
51. D-threo Methylphenidate Hydrochloride
52. Akos027321667
53. Rac-threo-methylphenidate Hydrochloride
54. 2-piperidineacetic Acid, Alpha-phenyl-, Methyl Ester, Hydrochloride, (alphar,2r)-
55. 2-piperidineacetic Acid, Alpha-phenyl-, Methyl Ester, Hydrochloride, (r*,r*)- (+-)-
56. 2-piperidineacetic Acid, Alpha-phenyl-, Methyl Ester, Hydrochloride, (r-(r*,r*))-
57. Methylphenidate Hydrochloride [mi]
58. (+/-)-threo-methylphenidate Hydrochloride
59. Methylphenidate Hydrochloride [jan]
60. Methylphenidate Hydrochloride [mart.]
61. Methylphenidate Hydrochloride [vandf]
62. Methylphenidate Hydrochloride [who-dd]
63. D03721
64. Dexmethylphenidate Hydrochloride [mart.]
65. Dexmethylphenidate Hydrochloride [vandf]
66. Dexmethylphenidate Hydrochloride [who-dd]
67. Methylphenidate Hydrochloride [orange Book]
68. Methylphenidate Hydrochloride Cii [usp-rs]
69. Methylphenidate Hydrochloride [ep Monograph]
70. Methylphenidate Hydrochloride [usp Monograph]
71. Dexmethylphenidate Hydrochloride [orange Book]
72. Methylphenidate D-threo-form Hydrochloride [mi]
73. Q47487562
74. Azstarys Component Dexmethylphenidate Hydrochloride
75. Dexmethylphenidate Hydrochloride Component Of Azstarys
76. Methyl Phenyl(piperidin-2-yl)acetate--hydrogen Chloride (1/1)
77. Methylphenidate Hydrochloride 1.0 Mg/ml In Methanol (as Free Base)
78. 2-piperidineacetic Acid, .alpha.-phenyl-, Methyl Ester, Hydrochloride, (r*,r*)-(+/-)-
79. 2-piperidineacetic Acid, .alpha.-phenyl-, Methyl Ester, Hydrochloride,(.alpha.r,2r)-
| Molecular Weight | 269.77 g/mol |
|---|---|
| Molecular Formula | C14H20ClNO2 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 4 |
| Exact Mass | 269.1182566 g/mol |
| Monoisotopic Mass | 269.1182566 g/mol |
| Topological Polar Surface Area | 38.3 Ų |
| Heavy Atom Count | 18 |
| Formal Charge | 0 |
| Complexity | 249 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 6 | |
|---|---|
| Drug Name | Dexmethylphenidate hydrochloride |
| Drug Label | Focalin (dexmethylphenidate hydrochloride) is the d-threo-enantiomer of racemic methylphenidate hydrochloride, which is a 50/50 mixture of the d-threo and l-threo-enantiomers. Focalin is a central nervous system (CNS) stimulant, available in 3 tabl... |
| Active Ingredient | Dexmethylphenidate hydrochloride |
| Dosage Form | Tablet; Capsule, extended release |
| Route | oral; Oral |
| Strength | 2.5mg; 5mg; 25mg; 30mg; 10mg; 15mg; 40mg; 20mg |
| Market Status | Tentative Approval; Prescription |
| Company | Watson Labs; Mylan Pharms; Teva Pharms; Intellipharmaceutics; Teva Pharms Usa |
| 2 of 6 | |
|---|---|
| Drug Name | Focalin |
| PubMed Health | Dexmethylphenidate (By mouth) |
| Drug Classes | CNS Stimulant |
| Drug Label | Focalin (dexmethylphenidate hydrochloride) is the d-threo-enantiomer of racemic methylphenidate hydrochloride, which is a 50/50 mixture of the d-threo and l-threo-enantiomers. Focalin is a central nervous system (CNS) stimulant, available in 3 tabl... |
| Active Ingredient | Dexmethylphenidate hydrochloride |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 2.5mg; 5mg; 10mg |
| Market Status | Prescription |
| Company | Novartis |
| 3 of 6 | |
|---|---|
| Drug Name | Focalin xr |
| PubMed Health | Dexmethylphenidate (By mouth) |
| Drug Classes | CNS Stimulant |
| Drug Label | Focalin XR is an extended-release formulation of dexmethylphenidate with a bi-modal release profile. Focalin XR uses the proprietary SODAS (Spheroidal Oral Drug Absorption System) technology. Each bead-filled Focalin XR capsule contains half the dose... |
| Active Ingredient | Dexmethylphenidate hydrochloride |
| Dosage Form | Capsule, extended release |
| Route | Oral |
| Strength | 25mg; 30mg; 15mg; 5mg; 10mg; 40mg; 20mg; 35mg |
| Market Status | Prescription |
| Company | Novartis |
| 4 of 6 | |
|---|---|
| Drug Name | Dexmethylphenidate hydrochloride |
| Drug Label | Focalin (dexmethylphenidate hydrochloride) is the d-threo-enantiomer of racemic methylphenidate hydrochloride, which is a 50/50 mixture of the d-threo and l-threo-enantiomers. Focalin is a central nervous system (CNS) stimulant, available in 3 tabl... |
| Active Ingredient | Dexmethylphenidate hydrochloride |
| Dosage Form | Tablet; Capsule, extended release |
| Route | oral; Oral |
| Strength | 2.5mg; 5mg; 25mg; 30mg; 10mg; 15mg; 40mg; 20mg |
| Market Status | Tentative Approval; Prescription |
| Company | Watson Labs; Mylan Pharms; Teva Pharms; Intellipharmaceutics; Teva Pharms Usa |
| 5 of 6 | |
|---|---|
| Drug Name | Focalin |
| PubMed Health | Dexmethylphenidate (By mouth) |
| Drug Classes | CNS Stimulant |
| Drug Label | Focalin (dexmethylphenidate hydrochloride) is the d-threo-enantiomer of racemic methylphenidate hydrochloride, which is a 50/50 mixture of the d-threo and l-threo-enantiomers. Focalin is a central nervous system (CNS) stimulant, available in 3 tabl... |
| Active Ingredient | Dexmethylphenidate hydrochloride |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 2.5mg; 5mg; 10mg |
| Market Status | Prescription |
| Company | Novartis |
| 6 of 6 | |
|---|---|
| Drug Name | Focalin xr |
| PubMed Health | Dexmethylphenidate (By mouth) |
| Drug Classes | CNS Stimulant |
| Drug Label | Focalin XR is an extended-release formulation of dexmethylphenidate with a bi-modal release profile. Focalin XR uses the proprietary SODAS (Spheroidal Oral Drug Absorption System) technology. Each bead-filled Focalin XR capsule contains half the dose... |
| Active Ingredient | Dexmethylphenidate hydrochloride |
| Dosage Form | Capsule, extended release |
| Route | Oral |
| Strength | 25mg; 30mg; 15mg; 5mg; 10mg; 40mg; 20mg; 35mg |
| Market Status | Prescription |
| Company | Novartis |
Dopamine Uptake Inhibitors
Drugs that block the transport of DOPAMINE into axon terminals or into storage vesicles within terminals. Most of the ADRENERGIC UPTAKE INHIBITORS also inhibit dopamine uptake. (See all compounds classified as Dopamine Uptake Inhibitors.)
Central Nervous System Stimulants
A loosely defined group of drugs that tend to increase behavioral alertness, agitation, or excitation. They work by a variety of mechanisms, but usually not by direct excitation of neurons. The many drugs that have such actions as side effects to their main therapeutic use are not included here. (See all compounds classified as Central Nervous System Stimulants.)
 Siegfried – A global CDMO delivering integrated pharmaceutical development and manufacturing solutions.
Siegfried – A global CDMO delivering integrated pharmaceutical development and manufacturing solutions.
DEXMETHYLPHENIDATE HYDROCHLORIDE
NDC Package Code : 0792-0619
Start Marketing Date : 1998-09-21
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
DEXMETHYLPHENIDATE HYDROCHLORIDE
NDC Package Code : 49812-0076
Start Marketing Date : 2019-09-12
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (100g/100g)
Marketing Category : BULK INGREDIENT
DEXMETHYLPHENIDATE HYDROCHLORIDE
NDC Package Code : 73548-2193
Start Marketing Date : 2025-05-01
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
 Curia - Accelerating therapies from discovery to commercialization with flexible, scalable CDMO solutions.
Curia - Accelerating therapies from discovery to commercialization with flexible, scalable CDMO solutions.
DEXMETHYLPHENIDATE HYDROCHLORIDE
NDC Package Code : 17180-1725
Start Marketing Date : 2012-06-26
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
DEXMETHYLPHENIDATE HYDROCHLORIDE
NDC Package Code : 50683-0428
Start Marketing Date : 2017-09-04
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

DEXMETHYLPHENIDATE HYDROCHLORIDE
NDC Package Code : 0406-1039
Start Marketing Date : 2016-08-12
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

DEXMETHYLPHENIDATE HYDROCHLORIDE
NDC Package Code : 53834-201
Start Marketing Date : 2019-07-19
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1g/g)
Marketing Category : BULK INGREDIENT

DEXMETHYLPHENIDATE HYDROCHLORIDE
NDC Package Code : 65392-3418
Start Marketing Date : 2012-06-26
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
DEXMETHYLPHENIDATE HYDROCHLORIDE
NDC Package Code : 61960-6900
Start Marketing Date : 2009-07-01
End Marketing Date : 2027-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
NDC Package Code : 61960-1724
Start Marketing Date : 2014-06-01
End Marketing Date : 2027-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Siegfried – A global CDMO delivering integrated pharmaceutical development and manufacturing solutions.
Siegfried – A global CDMO delivering integrated pharmaceutical development and manufacturing solutions.
About the Company : Siegfried is a global Contract Development and Manufacturing Organization providing integrated services for pharmaceutical ingredients & finished dosage forms. With 13 production s...
About the Company : Veranova is a global leader in developing and manufacturing specialist and complex APIs for pharma and biotech customers, with over 50 years of experience supporting the healthcare...
 Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Dexmethylphenidate Hydrochloride
About the Company : Cohance Lifesciences is a leading CDMO and API platform delivering products and services across the full molecule lifecycle, from development to commercialization. With strong expe...
 Noramco is a partner to the pharmaceutical industry for controlled substance development & manufacturing.
Noramco is a partner to the pharmaceutical industry for controlled substance development & manufacturing.
Dexmethylphenidate Hydrochloride
About the Company : Noramco, founded in 1979, specializes in developing and manufacturing APIs for opioid and non-opioid products. It excels in controlled substance development, offering supply-chain ...
 Curia - Accelerating therapies from discovery to commercialization with flexible, scalable CDMO solutions.
Curia - Accelerating therapies from discovery to commercialization with flexible, scalable CDMO solutions.
Dexmethylphenidate Hydrochloride
About the Company : Curia is a leading global CDMO with over 30 years of experience supporting clients across drug discovery, development, and commercial manufacturing. Leveraging deep scientific expe...
About the Company : Back in the year 1973, a team of individuals came up with such an idea – The idea called Alkem. Looking back at our 5 decades, illustrious journey, that one small idea has Not on...

About the Company : Centaur accepts change as a constant, and continuously innovates to remain significant. Centaur has built knowledge sharing relationships with the pharmaceutical majors in areas of...

Dexmethylphenidate Hydrochloride
About the Company : We have been supporting the pharmaceutical and biopharmaceutical industries with drug development for over 30 years, assisting our clients in developing and bringing new drugs to m...

Dexmethylphenidate Hydrochloride
About the Company : Harman Finochem Limited is a leading India-based Pharmaceutical Company which specializes in the manufacture and export of more than 45 Active Pharmaceutical Ingredients (APls) of ...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
55
PharmaCompass offers a list of Dexmethylphenidate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Dexmethylphenidate manufacturer or Dexmethylphenidate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Dexmethylphenidate manufacturer or Dexmethylphenidate supplier.
A Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride, including repackagers and relabelers. The FDA regulates Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride supplier is an individual or a company that provides Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride active pharmaceutical ingredient (API) or Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride finished formulations upon request. The Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride suppliers may include Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride API manufacturers, exporters, distributors and traders.
click here to find a list of Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride DMF (Drug Master File) is a document detailing the whole manufacturing process of Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride active pharmaceutical ingredient (API) in detail. Different forms of Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride DMFs exist exist since differing nations have different regulations, such as Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride DMF submitted to regulatory agencies in the US is known as a USDMF. Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride USDMF includes data on Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride suppliers with USDMF on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride suppliers with NDC on PharmaCompass.
Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride GMP manufacturer or Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride GMP API supplier for your needs.
A Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride CoA (Certificate of Analysis) is a formal document that attests to Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride's compliance with Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride specifications and serves as a tool for batch-level quality control.
Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride CoA mostly includes findings from lab analyses of a specific batch. For each Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride may be tested according to a variety of international standards, such as European Pharmacopoeia (Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride EP), Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Novartis Pharmaceutical Brand of Dexmethylphenidate Hydrochloride USP).

