
Synopsis
Synopsis
0
EU WC
0
KDMF
0
VMF
0
FDA Orange Book

0
Europe

0
Australia

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Ardeydorm
2. Ardeytropin
3. L Tryptophan
4. L Tryptophan Ratiopharm
5. L-tryptophan
6. L-tryptophan-ratiopharm
7. Levotryptophan
8. Lyphan
9. Naturruhe
10. Optimax
11. Pms Tryptophan
12. Pms-tryptophan
13. Ratio Tryptophan
14. Ratio-tryptophan
15. Trofan
16. Tryptacin
17. Tryptan
18. Tryptophan Metabolism Alterations
1. L-tryptophan
2. 73-22-3
3. L-tryptophane
4. H-trp-oh
5. (s)-tryptophan
6. Tryptophane
7. Trofan
8. Tryptacin
9. Optimax
10. Ardeytropin
11. (2s)-2-amino-3-(1h-indol-3-yl)propanoic Acid
12. Pacitron
13. Indole-3-alanine
14. Kalma
15. L-beta-3-indolylalanine
16. L-tryptofan
17. L-trp
18. L-(-)-tryptophan
19. 3-indol-3-ylalanine
20. Tryptan
21. Lyphan
22. Tryptophan (van)
23. 1-beta-3-indolylalanine
24. Tryptophan (h-3)
25. Triptofano [spanish]
26. Tryptophanum [latin]
27. 1h-indole-3-alanine
28. Tryptophan, L-
29. 1beta-3-indolylalanine
30. (-)-tryptophan
31. 2-amino-3-indolylpropanoic Acid
32. L(-)-tryptophan
33. Triptofano
34. Tryptophanum
35. (s)-alpha-amino-1h-indole-3-propanoic Acid
36. Tryptophane [french]
37. (l)-tryptophan
38. Alpha'-amino-3-indolepropionic Acid
39. Tryptophan [usan:inn]
40. L-alpha-amino-3-indolepropionic Acid
41. L-alpha-aminoindole-3-propionic Acid
42. Sedanoct
43. (s)-alpha-aminoindole-3-propionic Acid
44. 1h-indole-3-alanine (van)
45. Eh 121
46. Trp
47. Alanine, 3-indol-3-yl-
48. Ccris 617
49. L-alanine, 3-(1h-indol-3-yl)-
50. 1h-indole-3-alanine, (s)-
51. Alpha-amino-3-indolepropionic Acid, L-
52. Hsdb 4142
53. Trytophan-
54. (s)-alpha-amino-beta-(3-indolyl)-propionic Acid
55. Nci-c01729
56. Ai3-18478
57. (s)-2-amino-3-(3-indolyl)propionic Acid
58. Indole-3-propionic Acid, Alpha-amino-
59. 1h-indole-3-propanoic Acid, Alpha-amino-, (s)-
60. Propionic Acid, 2-amino-3-indol-3-yl-
61. Chebi:16828
62. Lopac-t-0254
63. Tryptophan ((-),l,s)
64. 8duh1n11bx
65. (s)-alpha-amino-beta-indolepropionic Acid
66. Chembl54976
67. (s)-2-amino-3-(1h-indol-3-yl)propanoic Acid
68. Nsc-13119
69. Mfcd00064340
70. Dsstox_cid_1419
71. Dsstox_rid_76152
72. Dsstox_gsid_21419
73. 80206-30-0
74. L-b-3-indolylalanine
75. L-tryptophan-13c11,15n2
76. D-trp-oh
77. Cas-73-22-3
78. Propionic Acid, 2-amino-3-indol-3-yl
79. L-tryptophan (9ci)
80. Tryptophan (usp/inn)
81. (s)-a-amino-b-indolepropionic Acid
82. (s)-a-aminoindole-3-propionic Acid
83. Alanine, 3-indol-3-yl
84. Einecs 200-795-6
85. Nsc 13119
86. Unii-8duh1n11bx
87. (2s)-2-amino-3-(1h-indol-3-yl)propanoate
88. Trytophan
89. (s)-a-amino-1h-indole-3-propanoic Acid
90. Trp-01
91. L-trytophan
92. 1qaw
93. L-tryptophan,(s)
94. L-trp-oh
95. 2a4m
96. H-l-trp-oh
97. Tryptophan [ii]
98. Tryptophan [mi]
99. L-tryptophan (jp17)
100. Tryptophan [inn]
101. S(-)-1-alpha-aminoindole-3-propionic Acid
102. Tryptophan [hsdb]
103. Tryptophan [inci]
104. Tryptophan [usan]
105. Tryptophan (l-tryptophan)
106. Tryptophan [vandf]
107. Tryptophan, L- (8ci)
108. Bmse000050
109. Bmse000868
110. Bmse001017
111. Epitope Id:136043
112. Ec 200-795-6
113. T 0254
114. L-tryptophan [fcc]
115. L-tryptophan [jan]
116. Schembl7328
117. Tryptophan [mart.]
118. 2-amino-3-indolylpropanoate
119. (s)-(-)-2-amino-3-(3-indolyl)propionic Acid
120. (s)-1h-indole-3-alanine
121. Lopac0_001183
122. Gtpl717
123. L-tryptophan [vandf]
124. Mls001056750
125. Divk1c_000457
126. L-tryptophan [usp-rs]
127. (s)-a-amino-b-indolepropionate
128. 151a3008-4cfe-40c9-ac0b-467ef0cb50ea
129. Dtxsid5021419
130. (s)-a-aminoindole-3-propionate
131. Bdbm21974
132. Hms501g19
133. Kbio1_000457
134. Zinc83315
135. Tryptophan [ep Monograph]
136. 3-(1h-indol-3-yl)-l-alanine
137. L-a-amino-3-indolepropionic Acid
138. Ninds_000457
139. Alpha-aminoindole-3-propionic Acid
140. Hms3263n07
141. Pharmakon1600-01500600
142. Tryptophan [usp Monograph]
143. Tryptophan, L- [who-dd]
144. Act08662
145. Hy-n0623
146. Str02722
147. (s)-alpha-aminoindole-3-propionate
148. Tox21_201246
149. Tox21_300359
150. Tox21_501183
151. Nsc757373
152. S3987
153. (s)-alpha-amino-beta-indolepropionate
154. L-tryptophan, Vetec(tm), 98.5%
155. (s)-a-amino-1h-indole-3-propanoate
156. Akos015854052
157. Indoe-3-propionic Acid, Alpha-amino-
158. Am82273
159. Ccg-205257
160. Cs-w020011
161. Db00150
162. Lp01183
163. Nsc-757373
164. Sdccgsbi-0051150.p002
165. Idi1_000457
166. Ncgc00015994-01
167. Ncgc00094437-01
168. Ncgc00094437-02
169. Ncgc00094437-03
170. Ncgc00094437-04
171. Ncgc00094437-08
172. Ncgc00254424-01
173. Ncgc00258798-01
174. Ncgc00261868-01
175. (s)-alpha-amino-1h-indole-3-propanoate
176. Ac-17050
177. Bp-13286
178. Smr000326686
179. Ts-04426
180. Db-029986
181. L-tryptophan, Bioultra, >=99.5% (nt)
182. A7403
183. Eu-0101183
184. T0541
185. (s)-tryptophan 1h-indole-3-alanine, (s)-
186. 73t223
187. C00078
188. D00020
189. L-.alpha.-amino-3-indolepropionic Acid
190. L-tryptophan, Reagent Grade, >=98% (hplc)
191. M02943
192. P16427
193. Ab00373874_05
194. L-tryptophan, Vetec(tm) Reagent Grade, >=98%
195. (s)-2-amino-3-(1h-indol-3-yl)-propionic Acid
196. A837752
197. L-tryptophan, Cell Culture Reagent (h-l-trp-oh)
198. Q181003
199. Sr-01000075590
200. 4-(3-methoxyanilino)-4-oxobut-2-enoicacid
201. N-acetyltryptophan Impurity A [ep Impurity]
202. Sr-01000075590-1
203. F0001-2364
204. Z1245635763
205. 1h-indole-3-propanoic Acid, .alpha.-amino-, (s)-
206. L-tryptophan, Certified Reference Material, Tracecert(r)
207. Tryptophan, European Pharmacopoeia (ep) Reference Standard
208. L-tryptophan, United States Pharmacopeia (usp) Reference Standard
209. L-tryptophan, From Non-animal Source, Meets Ep, Jp, Usp Testing Specifications, Suitable For Cell Culture, 99.0-101.0%
210. L-tryptophan, Pharmagrade, Ajinomoto, Ep, Jp, Usp, Manufactured Under Appropriate Gmp Controls For Pharma Or Biopharmaceutical Production, Suitable For Cell Culture
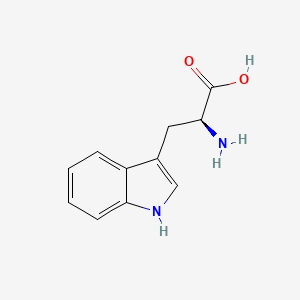
| Molecular Weight | 204.22 g/mol |
|---|---|
| Molecular Formula | C11H12N2O2 |
| XLogP3 | -1.1 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 3 |
| Exact Mass | 204.089877630 g/mol |
| Monoisotopic Mass | 204.089877630 g/mol |
| Topological Polar Surface Area | 79.1 Ų |
| Heavy Atom Count | 15 |
| Formal Charge | 0 |
| Complexity | 245 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Tryptophan is a precursor of serotonin. Because CNS depletion of serotonin is considered to be involved in depression, tryptophan has been used in its treatment. Although it has been given alone, evidence of effectiveness is scant and tryptophan has generally been used as adjunctive therapy in depression. It has sometimes been given with pyridoxine and ascorbic acid, which are involved in its metabolism to serotonin
Sweetman SC (ed), Martindale: The Complete Drug Reference. London: Pharmaceutical Press (2009), p.427.
/EXPTL USE/: Inhibition of Walker 256 intramuscular carcinoma in rats by admin of l-tryptophan.
PMID:5446186 Gold J; Oncology 24 (4): 291-303 (1970)
(L)-Tryptophan decreases sleep latency and slightly increases sleeping time without altering qualitative characteristics of polygraphic patterns during sleep in normal subjects. In insomniac patients, it increases sleeping time and decreases both sleep latency and number of awakenings.
PMID:4935665 Hartmann E et al; Psychopharmacologia 19 (2): 114 (1971)
Beneficial effects were observed when L-tryptophan was administered to 2 patients with myoclonus. In each case suspension of methylcellulose and water containing 1 g of (L)-tryptophan/15 mL was prepared and administered orally at a level of 10 g daily in 5 divided doses.
Delean J, Richardson JC; Lancet 2 (7940): 870-1 (1975)
For more Therapeutic Uses (Complete) data for (L)-Tryptophan (11 total), please visit the HSDB record page.
Since serotonin plays a role in inducing and maintaining sleep, l-tryptophan has been administered orally to increase brain levels of serotonin. Although a dose of 1 g significantly decreased sleep latency and total time awake without altering sleep patterns, the hypnotic action is observed only during the early part of the sleep cycle, is unpredictable, and is not characterized by a satisfactory dose-response relationship. Because the hypnotic action has not been confirmed in other studies, this use of l-tryptophan must be considered investigational and the drug is not recommended in routine clinical practice. In order to avoid central serotonergic toxicity, tryptophan should not be used in patients also receiving a monoamine oxidase inhibitor or the serotonin uptake inhibitor, fluoxetine (Prozac).
American Medical Association. AMA Drug Evaluations Annual 1991. Chicago, IL: American Medical Association, 1991., p. 215
Tryptophan-containing products have been associated with the eosinophilia-myalgia syndrome. Other adverse effects that have been reported include nausea, headache, lightheadedness, and drowsiness.
Sweetman SC (ed), Martindale: The Complete Drug Reference. London: Pharmaceutical Press (2009), p.427.
An increased incidence of bladder tumours has been reported in mice given l-tryptophan orally as well as in cholesterol pellets embedded in the bladder lumen. However, there was no increase in tumour incidence when only high-dose, oral tryptophan was given.
Sweetman SC (ed), Martindale: The Complete Drug Reference. London: Pharmaceutical Press (2009), p.427.
Tryptophan has been associated with eosinophilia-myalgia syndrome; caution is advised in patients receiving the drug who develop some, but not all, of the symptoms of this syndrome. It should not be used in those with a history of eosinophilia-myalgia syndrome associated with tryptophan treatment.
Sweetman SC (ed), Martindale: The Complete Drug Reference. London: Pharmaceutical Press (2009), p.427.
For more Drug Warnings (Complete) data for (L)-Tryptophan (7 total), please visit the HSDB record page.
Tryptophan may be useful in increasing serotonin production, promoting healthy sleep, managing depression by enhancing mental and emotional well-being, managing pain tolerance, and managing weight.
Tryptophan is critical for the production of the body's proteins, enzymes and muscle tissue. It is also essential for the production of niacin, the synthesis of the neurotransmitter serotonin and melatonin. Tryptophan supplements can be used as natural relaxants to help relieve insomnia. Tryptophan can also reduce anxiety and depression and has been shown to reduce the intensity of migraine headaches. Other promising indications include the relief of chronic pain, reduction of impulsivity or mania and the treatment of obsessive or compulsive disorders. Tryptophan also appears to help the immune system and can reduce the risk of cardiac spasms. Tryptophan deficiencies may lead to coronary artery spasms. Tryptophan is used as an essential nutrient in infant formulas and intravenous feeding. Tryptophan is marketed as a prescription drug (Tryptan) for those who do not seem to respond well to conventional antidepressants. It may also be used to treat those afflicted with seasonal affective disorder (a winter-onset depression). Tryptopan serves as the precursor for the synthesis of serotonin (5-hydroxytryptamine, 5-HT) and melatonin (N-acetyl-5-methoxytryptamine).
Antidepressive Agents, Second-Generation
A structurally and mechanistically diverse group of drugs that are not tricyclics or monoamine oxidase inhibitors. The most clinically important appear to act selectively on serotonergic systems, especially by inhibiting serotonin reuptake. (See all compounds classified as Antidepressive Agents, Second-Generation.)
N - Nervous system
N06 - Psychoanaleptics
N06A - Antidepressants
N06AX - Other antidepressants
N06AX02 - Tryptophan
(L)-Tryptophan with plant oils in soft gelatin capsules permitted lower dosage than with usual dosage form. Max of free tryptophan in serum was achieved in 1st hr whereas 4-5 times as much would be required with tablets or hard gelatin capsules.
Klosa J; Ger Offen Patent NO 2824362 12/13/79
Absorption and Fate. Tryptophan is readily absorbed from the gastro-intestinal tract. Tryptophan is extensively bound to serum albumin. It is metabolized to serotonin and other metabolites, incl kynurenine derivatives, and excreted in the urine. Pyridoxine and ascorbic acid appear to be concerned in its metabolism.
Reynolds, J.E.F., Prasad, A.B. (eds.) Martindale-The Extra Pharmacopoeia. 28th ed. London: The Pharmaceutical Press, 1982., p. 61
Although the free amino acids dissolved in the body fluids are only a very small proportion of the body's total mass of amino acids, they are very important for the nutritional and metabolic control of the body's proteins. ... Although the plasma compartment is most easily sampled, the concentration of most amino acids is higher in tissue intracellular pools. Typically, large neutral amino acids, such as leucine and phenylalanine, are essentially in equilibrium with the plasma. Others, notably glutamine, glutamic acid, and glycine, are 10- to 50-fold more concentrated in the intracellular pool. Dietary variations or pathological conditions can result in substantial changes in the concentrations of the individual free amino acids in both the plasma and tissue pools. /Amino acids/
NAS, Food and Nutrition Board, Institute of Medicine; Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids (Macronutrients). National Academy Press, Washington, D.C., pg. 596, 2009. Available from, as of March 10, 2010: https://www.nap.edu/catalog/10490.html
After ingestion, proteins are denatured by the acid in the stomach, where they are also cleaved into smaller peptides by the enzyme pepsin, which is activated by the increase in stomach acidity that occurs on feeding. The proteins and peptides then pass into the small intestine, where the peptide bonds are hydrolyzed by a variety of enzymes. These bond-specific enzymes originate in the pancreas and include trypsin, chymotrypsins, elastase, and carboxypeptidases. The resultant mixture of free amino acids and small peptides is then transported into the mucosal cells by a number of carrier systems for specific amino acids and for di- and tri-peptides, each specific for a limited range of peptide substrates. After intracellular hydrolysis of the absorbed peptides, the free amino acids are then secreted into the portal blood by other specific carrier systems in the mucosal cell or are further metabolized within the cell itself. Absorbed amino acids pass into the liver, where a portion of the amino acids are taken up and used; the remainder pass through into the systemic circulation and are utilized by the peripheral tissues. /Amino acids/
NAS, Food and Nutrition Board, Institute of Medicine; Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids (Macronutrients). National Academy Press, Washington, D.C., pg. 599, 2009. Available from, as of March 10, 2010: https://www.nap.edu/catalog/10490.html
For more Absorption, Distribution and Excretion (Complete) data for (L)-Tryptophan (9 total), please visit the HSDB record page.
Hepatic.
In Hartnup disease ... tryptophane appear/s/ in urine due to defective renal and intestinal absorption of tryptophane ... It is an intermediary metabolite in the synthesis of serotonin (5-hydroxytryptamine) and 5-hydroxyindole acetic acid (HIAA).
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 590
Patients with bladder cancer excreted significantly more kynurenic acid, acetylkynurenine, kynurenine, and 3-hydroxykynurenine after ingesting a loading dose of L-tryptophan than did control subjects with no known disease.
Searle, C. E. (ed.). Chemical Carcinogens. ACS Monograph 173. Washington, DC: American Chemical Society, 1976., p. 443
Tryptophan is metabolized in the liver by tryptophan pyrrolase and tryptophan hydroxylase. Metabolites include hydroxytryptophan, which is then converted to serotonin, and kynurenine derivatives. Some tryptophan is converted to nicotinic acid and nicotinamide. Pyridoxine and ascorbic acid are cofactors in the decarboxylation and hydroxylation, respectively, of tryptophan; pyridoxine apparently prevents the accumulation of the kynurenine metabolites.
Sweetman SC (ed), Martindale: The Complete Drug Reference. London: Pharmaceutical Press (2009), p.427.
Yields indole-3-pyruvic acid in man ... and in rats; yields tryptamine in guinea pigs. /From table/
Goodwin, B.L. Handbook of Intermediary Metabolism of Aromatic Compounds. New York: Wiley, 1976., p. T-38
For more Metabolism/Metabolites (Complete) data for (L)-Tryptophan (21 total), please visit the HSDB record page.
The biological half-life of tryptophan was reported to be 15.8 hr.
Reynolds, J.E.F., Prasad, A.B. (eds.) Martindale-The Extra Pharmacopoeia. 28th ed. London: The Pharmaceutical Press, 1982., p. 61
A number of important side reactions occur during the catabolism of tryptophan on the pathway to acetoacetate. The first enzyme of the catabolic pathway is an iron porphyrin oxygenase that opens the indole ring. The latter enzyme is highly inducible, its concentration rising almost 10-fold on a diet high in tryptophan. Kynurenine is the first key branch point intermediate in the pathway. Kynurenine undergoes deamniation in a standard transamination reaction yielding kynurenic acid. Kynurenic acid and metabolites have been shown to act as antiexcitotoxics and anticonvulsives. A second side branch reaction produces anthranilic acid plus alanine. Another equivalent of alanine is produced further along the main catabolic pathway, and it is the production of these alanine residues that allows tryptophan to be classified among the glucogenic and ketogenic amino acids. The second important branch point converts kynurenine into 2-amino-3-carboxymuconic semialdehyde, which has two fates. The main flow of carbon elements from this intermediate is to glutarate. An important side reaction in liver is a transamination and several rearrangements to produce limited amounts of nicotinic acid, which leads to production of a small amount of NAD+ and NADP+.
Findings indicate that enhanced rates of serotonin turnover produced by (L)-tryptophan and physical restraint are associated with inhibition of thyroid-stimulating hormone (TSH) and stimulation of prolactin release from anterior pituitary in rats.
PMID:1083471 Mueller GP et al; Life Sci 18 (7): 715-24 (1976)
L-Tryptophan, an indispensable amino acid, serves as a precursor for several small molecules of functional significance including the vitamin niacin, the neurotransmitter serotonin, the metabolite tryptamine, and the pineal hormone melatonin. Increases in tryptophan have been shown to increase synthesis of the neurotransmitters in brain, blood, and other body organs.
NAS, Food and Nutrition Board, Institute of Medicine; Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids (Macronutrients). National Academy Press, Washington, D.C., pg. 731, 2009. Available from, as of March 10, 2010: https://www.nap.edu/catalog/10490.html
Amino acids are selected for protein synthesis by binding with transfer RNA (tRNA) in the cell cytoplasm. The information on the amino acid sequence of each individual protein is contained in the sequence of nucleotides in the messenger RNA (mRNA) molecules, which are synthesized in the nucleus from regions of DNA by the process of transcription. The mRNA molecules then interact with various tRNA molecules attached to specific amino acids in the cytoplasm to synthesize the specific protein by linking together individual amino acids; this process, known as translation, is regulated by amino acids (e.g., leucine), and hormones. Which specific proteins are expressed in any particular cell and the relative rates at which the different cellular proteins are synthesized, are determined by the relative abundances of the different mRNAs and the availability of specific tRNA-amino acid combinations, and hence by the rate of transcription and the stability of the messages. From a nutritional and metabolic point of view, it is important to recognize that protein synthesis is a continuing process that takes place in most cells of the body. In a steady state, when neither net growth nor protein loss is occurring, protein synthesis is balanced by an equal amount of protein degradation. The major consequence of inadequate protein intakes, or diets low or lacking in specific indispensable amino acids relative to other amino acids (often termed limiting amino acids), is a shift in this balance so that rates of synthesis of some body proteins decrease while protein degradation continues, thus providing an endogenous source of those amino acids most in need. /Amino acids/
NAS, Food and Nutrition Board, Institute of Medicine; Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids (Macronutrients). National Academy Press, Washington, D.C., pg. 601-602, 2009. Available from, as of March 10, 2010: https://www.nap.edu/catalog/10490.html
The mechanism of intracellular protein degradation, by which protein is hydrolyzed to free amino acids, is more complex and is not as well characterized at the mechanistic level as that of synthesis. A wide variety of different enzymes that are capable of splitting peptide bonds are present in cells. However, the bulk of cellular proteolysis seems to be shared between two multienzyme systems: the lysosomal and proteasomal systems. The lysosome is a membrane-enclosed vesicle inside the cell that contains a variety of proteolytic enzymes and operates mostly at acid pH. Volumes of the cytoplasm are engulfed (autophagy) and are then subjected to the action of the protease enzymes at high concentration. This system is thought to be relatively unselective in most cases, although it can also degrade specific intracellular proteins. The system is highly regulated by hormones such as insulin and glucocorticoids, and by amino acids. The second system is the ATP-dependent ubiquitin-proteasome system, which is present in the cytoplasm. The first step is to join molecules of ubiquitin, a basic 76-amino acid peptide, to lysine residues in the target protein. Several enzymes are involved in this process, which selectively targets proteins for degradation by a second component, the proteasome.
NAS, Food and Nutrition Board, Institute of Medicine; Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids (Macronutrients). National Academy Press, Washington, D.C., pg. 602, 2009. Available from, as of March 10, 2010: https://www.nap.edu/catalog/10490.html
For more Mechanism of Action (Complete) data for (L)-Tryptophan (7 total), please visit the HSDB record page.

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Related Excipient Companies

Excipients by Applications
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
39
PharmaCompass offers a list of Tryptophan API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Tryptophan manufacturer or Tryptophan supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Tryptophan manufacturer or Tryptophan supplier.
PharmaCompass also assists you with knowing the Tryptophan API Price utilized in the formulation of products. Tryptophan API Price is not always fixed or binding as the Tryptophan Price is obtained through a variety of data sources. The Tryptophan Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A MolPort-001-794-499 manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of MolPort-001-794-499, including repackagers and relabelers. The FDA regulates MolPort-001-794-499 manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. MolPort-001-794-499 API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of MolPort-001-794-499 manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A MolPort-001-794-499 supplier is an individual or a company that provides MolPort-001-794-499 active pharmaceutical ingredient (API) or MolPort-001-794-499 finished formulations upon request. The MolPort-001-794-499 suppliers may include MolPort-001-794-499 API manufacturers, exporters, distributors and traders.
click here to find a list of MolPort-001-794-499 suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A MolPort-001-794-499 DMF (Drug Master File) is a document detailing the whole manufacturing process of MolPort-001-794-499 active pharmaceutical ingredient (API) in detail. Different forms of MolPort-001-794-499 DMFs exist exist since differing nations have different regulations, such as MolPort-001-794-499 USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A MolPort-001-794-499 DMF submitted to regulatory agencies in the US is known as a USDMF. MolPort-001-794-499 USDMF includes data on MolPort-001-794-499's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The MolPort-001-794-499 USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of MolPort-001-794-499 suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The MolPort-001-794-499 Drug Master File in Japan (MolPort-001-794-499 JDMF) empowers MolPort-001-794-499 API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the MolPort-001-794-499 JDMF during the approval evaluation for pharmaceutical products. At the time of MolPort-001-794-499 JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of MolPort-001-794-499 suppliers with JDMF on PharmaCompass.
A MolPort-001-794-499 CEP of the European Pharmacopoeia monograph is often referred to as a MolPort-001-794-499 Certificate of Suitability (COS). The purpose of a MolPort-001-794-499 CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of MolPort-001-794-499 EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of MolPort-001-794-499 to their clients by showing that a MolPort-001-794-499 CEP has been issued for it. The manufacturer submits a MolPort-001-794-499 CEP (COS) as part of the market authorization procedure, and it takes on the role of a MolPort-001-794-499 CEP holder for the record. Additionally, the data presented in the MolPort-001-794-499 CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the MolPort-001-794-499 DMF.
A MolPort-001-794-499 CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. MolPort-001-794-499 CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of MolPort-001-794-499 suppliers with CEP (COS) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing MolPort-001-794-499 as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for MolPort-001-794-499 API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture MolPort-001-794-499 as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain MolPort-001-794-499 and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a MolPort-001-794-499 NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of MolPort-001-794-499 suppliers with NDC on PharmaCompass.
MolPort-001-794-499 Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of MolPort-001-794-499 GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right MolPort-001-794-499 GMP manufacturer or MolPort-001-794-499 GMP API supplier for your needs.
A MolPort-001-794-499 CoA (Certificate of Analysis) is a formal document that attests to MolPort-001-794-499's compliance with MolPort-001-794-499 specifications and serves as a tool for batch-level quality control.
MolPort-001-794-499 CoA mostly includes findings from lab analyses of a specific batch. For each MolPort-001-794-499 CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
MolPort-001-794-499 may be tested according to a variety of international standards, such as European Pharmacopoeia (MolPort-001-794-499 EP), MolPort-001-794-499 JP (Japanese Pharmacopeia) and the US Pharmacopoeia (MolPort-001-794-499 USP).
