
Synopsis
Synopsis
0
CEP/COS
0
EU WC
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Europe

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
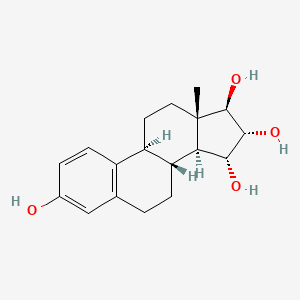

1. 15 Alpha Hydroxy Estriol
2. 15 Alpha Hydroxyestriol
3. 15 Alpha-hydroxyestriol
4. 15-alpha-hydroxy-estriol
1. 15183-37-6
2. Estetrol Anhydrous
3. 15.alpha.-hydroxyestriol
4. Enb39r14vf
5. (8r,9s,13s,14s,15r,16r,17r)-13-methyl-6,7,8,9,11,12,14,15,16,17-decahydrocyclopenta[a]phenanthrene-3,15,16,17-tetrol
6. Estetrol (usan)
7. Estetrol [usan]
8. (8r,9s,13s,14s,15r,16r,17r)-13-methyl-7,8,9,11,12,13,14,15,16,17-decahydro-6h-cyclopenta[a]phenanthrene-3,15,16,17-tetraol
9. E4
10. (14beta,15alpha,16alpha,17alpha)-estra-1,3,5(10)-triene-3,15,16,17-tetrol
11. E-4
12. Unii-enb39r14vf
13. Estell
14. Estetrolum
15. Oestetrol
16. Donesta
17. Estetrol (anhydrous)
18. 15alpha-hydroxyestriol
19. 15 Alpha-hydroxyestriol
20. Estetrol [inn]
21. Estetrol [who-dd]
22. Mls006010253
23. Schembl145580
24. Estetrol, >=98% (hplc)
25. Chembl1230314
26. Gtpl11591
27. Dtxsid50164888
28. Chebi:142773
29. Bdbm158505
30. Zinc5764481
31. Estetrol (anhydrous) [usan]
32. Who 10439
33. Akos030254521
34. At27982
35. Db12235
36. Ncgc00345819-03
37. Hy-15731
38. Smr004701329
39. Us9034854, E4
40. Cs-0008552
41. D11513
42. J-008852
43. Q5401078
44. Drovelis (ema); ; Estetrol Monohydrate + Drospirenone
45. Lydisilka (ema); ; Estetrol Monohydrate + Drospirenone
46. Nextstellis (us); Estetrol Monohydrate + Drospirenone
47. 1,3,5(10)-estratrien-3,15alpha,16alpha,17beta-tetrol
48. Estra-1(10),2,4-triene-3,15alpha,16alpha,17beta-tetrol
49. Estra-1,3,5(10)-triene-3,15alpha,16alpha,17beta-tetrol
50. 3,15alpha,16alpha,17beta-tetrahydroxyestra-1,3,5(10)-triene
51. Estra-1,3,5(10)-triene-3,15 Alpha,16alpha,17beta-tetrol
52. (15alpha,16alpha,17beta)-estra-1(10),2,4-triene-3,15,16,17-tetrol
53. Estra-1,3,5(10)-triene-3,15,16,17-tetrol, (15alpha,16alpha,17beta)-
54. Estra-1,3,5(10)-triene-3,15.alpha.,16.alpha.,17.beta.-tetrol
55. Estra-1,3,5(10)-triene-3,15,16,17-tetrol, (15.alpha.,16.alpha.,17.beta.)-
56. 4oh
| Molecular Weight | 304.4 g/mol |
|---|---|
| Molecular Formula | C18H24O4 |
| XLogP3 | 1.5 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 0 |
| Exact Mass | 304.16745924 g/mol |
| Monoisotopic Mass | 304.16745924 g/mol |
| Topological Polar Surface Area | 80.9 Ų |
| Heavy Atom Count | 22 |
| Formal Charge | 0 |
| Complexity | 441 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 7 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Estetrol is indicated in combination with drospirenone for the prevention of pregnancy.
Estetrol prevents pregnancy by suppressing ovulation.
Absorption
Estetrol is rapidly absorbed from the gastrointestinal tract. The Cmax of estetrol is 18 ng/mL according to the results of a pharmacokinetic study, with an AUC of 36.4 ngh/mL. When estetrol and drospirenone are taken in a single product, maximum serum concentrations of approximately 48.7 ng/mL are achieved within 1-3 h. Bioavailability of the combination ranges between 76 and 85%. The Tmax can range from 0.5 to 2 hours and time to steady state is approximately 4 days, according to the results of one clinical study.
Route of Elimination
Estrogens are generally excreted as sulfated and glucuronidated derivatives. Approximately 69% of a dose of estetrol is excreted in the urine, and about 22% is excreted in the feces as unchanged drug.
Volume of Distribution
Limited distribution of estetrol into red blood cells has been demonstrated.
Estretol is heavily metabolized after oral administration. Phase 2 metabolism of estrogen forms glucuronide and sulfate conjugates with negligible in-vitro estrogenic activity. In vitro metabolism studies demonstrate that UGT2B7 catalyzes the formation of E4-16-glucuronide. Estetrol is combined with [drospirenone] in a product. The hepatic cytochrome enzyme CYP3A4 metabolizes drospirenone to two primary metabolites: the acid form of drospirenone through the opening of the lactone ring and the 4,5 dihydrodrospirenone formed by reduction, followed by sulfation. Both metabolites are pharmacologically inactive.
The elimination half-life of estetrol is approximately 27 hours. Half-life may range between 19-40 hours.
Estetrol is a synthetic analogue of a naturally occurring estrogen present during pregnancy, demonstrating selectivity for both estrogen receptor- (ER-) and ER- and suppressing ovulation. Estetrol binds with a low to moderate affinity human estrogen receptor alpha (ER alpha) and ER beta with a preference for ER alpha. Estetrol demonstrates a unique mechanism of action via tissue selective activity, showing estrogen receptor agonist activity on the vagina, the uterus and the endometrium, and negative estrogenic activity on breast tissue.
 Transo-Pharm GmbH works globally to supply Active Pharmaceutical Ingredients adhering to the highest quality & GMP standards.
Transo-Pharm GmbH works globally to supply Active Pharmaceutical Ingredients adhering to the highest quality & GMP standards.

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 34214
Submission : 2019-11-27
Status : Active
Type : II
 Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.

NDC Package Code : 10577-918
Start Marketing Date : 2019-11-15
End Marketing Date : 2025-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 37136
Submission : 2022-06-10
Status : Active
Type : II

Registration Number : 305MF10111
Registrant's Address : BUDAPEST, GYOMROI UT 19-21, HUNGARY
Initial Date of Registration : 2023-09-26
Latest Date of Registration :


 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Market Place

Reply
27 Jul 2024
Reply
02 Sep 2022
Reply
25 Jun 2022
Reply
09 May 2022
Reply
03 Nov 2021
Reply
04 Sep 2021
Reply
29 May 2021
Reply
03 Sep 2020
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Patents & EXCLUSIVITIES
ABOUT THIS PAGE
98
PharmaCompass offers a list of Estetrol API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Estetrol manufacturer or Estetrol supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Estetrol manufacturer or Estetrol supplier.
PharmaCompass also assists you with knowing the Estetrol API Price utilized in the formulation of products. Estetrol API Price is not always fixed or binding as the Estetrol Price is obtained through a variety of data sources. The Estetrol Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Estetrol manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Estetrol, including repackagers and relabelers. The FDA regulates Estetrol manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Estetrol API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Estetrol manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Estetrol supplier is an individual or a company that provides Estetrol active pharmaceutical ingredient (API) or Estetrol finished formulations upon request. The Estetrol suppliers may include Estetrol API manufacturers, exporters, distributors and traders.
click here to find a list of Estetrol suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Estetrol DMF (Drug Master File) is a document detailing the whole manufacturing process of Estetrol active pharmaceutical ingredient (API) in detail. Different forms of Estetrol DMFs exist exist since differing nations have different regulations, such as Estetrol USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Estetrol DMF submitted to regulatory agencies in the US is known as a USDMF. Estetrol USDMF includes data on Estetrol's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Estetrol USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Estetrol suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Estetrol Drug Master File in Japan (Estetrol JDMF) empowers Estetrol API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Estetrol JDMF during the approval evaluation for pharmaceutical products. At the time of Estetrol JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Estetrol suppliers with JDMF on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Estetrol as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Estetrol API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Estetrol as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Estetrol and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Estetrol NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Estetrol suppliers with NDC on PharmaCompass.
Estetrol Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Estetrol GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Estetrol GMP manufacturer or Estetrol GMP API supplier for your needs.
A Estetrol CoA (Certificate of Analysis) is a formal document that attests to Estetrol's compliance with Estetrol specifications and serves as a tool for batch-level quality control.
Estetrol CoA mostly includes findings from lab analyses of a specific batch. For each Estetrol CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Estetrol may be tested according to a variety of international standards, such as European Pharmacopoeia (Estetrol EP), Estetrol JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Estetrol USP).
