
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
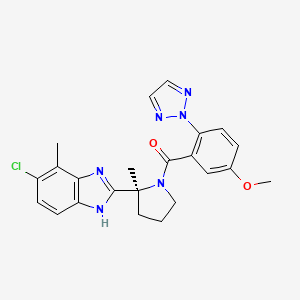

1. ((s)-(2-(5-chloro-4-methyl-1h-benzo(d)imidazol-2-yl)-2-methylpyrrolidin-1-yl) (5 Methoxy-2-(2h-1,2,3-triazol-2-yl)phenyl)methanone)
2. Act-541468
3. Daridorexant
4. Methanone, ((2s)-2-(6-chloro-7-methyl-1h-benzimidazol-2-yl)-2-methyl-1-pyrrolidinyl)(5-methoxy-2-(2h-1,2,3-traizol-2-yl)phenyl)-
1. Daridorexant
2. 1505484-82-1
3. Act-541468
4. Quviviq
5. Daridorexant [usan]
6. Lmq24g57e9
7. Act541468
8. [(2~{s})-2-(6-chloranyl-7-methyl-1~{h}-benzimidazol-2-yl)-2-methyl-pyrrolidin-1-yl]-[5-methoxy-2-(1,2,3-triazol-2-yl)phenyl]methanone
9. [(2s)-2-(5-chloro-4-methyl-1h-benzimidazol-2-yl)-2-methylpyrrolidin-1-yl]-[5-methoxy-2-(triazol-2-yl)phenyl]methanone
10. [(2s)-2-(5-chloro-4-methyl-1h-benzimidazol-2-yl)-2-methylpyrrolidin-1-yl][5-methoxy-2-(2h-1,2,3-triazol-2-yl)phenyl]methanone
11. Methanone, ((2s)-2-(6-chloro-7-methyl-1h-benzimidazol-2-yl)-2-methyl-1-pyrrolidinyl)(5-methoxy-2-(2h-1,2,3-triazol-2-yl)phenyl)-
12. Nemorexant (former Name)
13. Daridorexant [inn]
14. Unii-lmq24g57e9
15. Daridorexant [who-dd]
16. Chembl4297590
17. Schembl16766318
18. Gtpl11648
19. Bdbm334973
20. Dtxsid701336356
21. Nemorexant; Act-541468
22. Who 10679
23. Us9732075, Example 5.36
24. At32805
25. Db15031
26. Hy-109095
27. Cs-0039396
28. Act 541468;act-541468
29. (2s)-2-(5-chloro-4-methyl-1h-benzimidazol-2-yl)-2-methylpyrrolidin-1-yl)(5-methoxy-2-(2h-1,2,3-triazol-2-yl)phenyl)methanone
30. (s)-(2-(5-chloro-4-methyl-1h-benzo[d]imidazol-2-yl)-2-methylpyrrolidin-1-yl)(5 Methoxy-2-(2h-1,2,3-triazol-2-yl)phenyl)methanone
31. (s)-2-(5-chloro-4-methyl-1h Benzo[d]imidazol-2-yl)-2-methylpyrrolidin-1-yl](5-methoxy-2-(2h-1,2,3-triazol-2-yl)phenyl)methanone
32. [(s)-2-(5-chloro-4-methyl-1h-benzoimidazol-2-yl)-2-methyl-pyrrolidin-1-yl]-(5-methoxy-2-[1,2,3]triazol-2-yl-phenyl)-methanone
| Molecular Weight | 450.9 g/mol |
|---|---|
| Molecular Formula | C23H23ClN6O2 |
| XLogP3 | 4.1 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Exact Mass | 450.1571017 g/mol |
| Monoisotopic Mass | 450.1571017 g/mol |
| Topological Polar Surface Area | 88.9 Ų |
| Heavy Atom Count | 32 |
| Formal Charge | 0 |
| Complexity | 693 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Quviviq is indicated for the treatment of adult patients with insomnia characterised by symptoms present for at least 3 months and considerable impact on daytime functioning.
N05
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 41227
Submission : 2025-01-20
Status : Active
Type : II
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]About the Company : Established in May 2012, Shandong Loncom Pharmaceutical is a wholly owned subsidiary of Shandong Bestcomm Pharmaceutical Co., Ltd. Located in the Qihe Economic Development Zone, Sh...
About the Company : Inke S.A., is focused since 1980 in the development and manufacture of the highest quality Active Pharmaceutical Ingredients (APIs) with complex synthesis processes for diverse the...
 Farmak works in the development, production and marketing of APIs, Intermediates & Specialties// FDA inspected.
Farmak works in the development, production and marketing of APIs, Intermediates & Specialties// FDA inspected.
About the Company : Farmak, A.S. is a privately held chemical and pharmaceutical company based in the Czech Republic. It specializes in researching, developing and producing active pharmaceutical ingr...
 Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
About the Company : Established in 2004, Metrochem API is one of the fastest-growing APIs, pellets & intermediates manufacturers. It has 6 dedicated manufacturing facilities for its 3 core product gro...
About the Company : Since 1962, MOEHS has produced high-quality Active Pharmaceutical Ingredients (APIs) for the global market. With decades of technical expertise, Moehs Group delivers pharmaceutical...
 Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
About the Company : Supriya Lifescience Limited, established in 1987 and headquartered in Mumbai, is a globally recognized, technology-driven manufacturer of APIs, CDMO, and formulations. Its faciliti...
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Details:
Daridorexant is a controlled substance drug, which is currently being evaluated in Phase IV clinical studies for the treatment of sleep initiation and maintenance disorders.
Lead Product(s): Daridorexant,Inapplicable
Therapeutic Area: Sleep Brand Name: Undisclosed
Study Phase: Phase IVProduct Type: Controlled Substance
Sponsor: Idorsia Pharmaceuticals
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable April 29, 2026

Lead Product(s) : Daridorexant,Inapplicable
Therapeutic Area : Sleep
Highest Development Status : Phase IV
Partner/Sponsor/Collaborator : Idorsia Pharmaceuticals
Deal Size : Inapplicable
Deal Type : Inapplicable
Efficacy and Safety of Daridorexant in Patients With Major Depressive Disorder and Insomnia
Details : Daridorexant is a controlled substance drug, which is currently being evaluated in Phase IV clinical studies for the treatment of sleep initiation and maintenance disorders.
Product Name : Undisclosed
Product Type : Controlled Substance
Upfront Cash : Inapplicable
April 29, 2026

Details:
Daridorexant is a controlled substance drug, which is currently being evaluated in Phase II clinical studies for the treatment of alcoholism.
Lead Product(s): Daridorexant,Inapplicable
Therapeutic Area: Psychiatry/Psychology Brand Name: Undisclosed
Study Phase: Phase IIProduct Type: Controlled Substance
Sponsor: National Institutes of Health
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable April 15, 2026

Lead Product(s) : Daridorexant,Inapplicable
Therapeutic Area : Psychiatry/Psychology
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : National Institutes of Health
Deal Size : Inapplicable
Deal Type : Inapplicable
Daridorexant for Alcohol Use Disorder
Details : Daridorexant is a controlled substance drug, which is currently being evaluated in Phase II clinical studies for the treatment of alcoholism.
Product Name : Undisclosed
Product Type : Controlled Substance
Upfront Cash : Inapplicable
April 15, 2026

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Through the licensing deal for Daridorexant, a controlled substance targeting the orexin receptor, the agreement aims to advance treatment options for insomnia.
Lead Product(s): Daridorexant,Inapplicable
Therapeutic Area: Sleep Brand Name: Quviviq
Study Phase: Approved FDFProduct Type: Controlled Substance
Sponsor: Pharmalink, S.L.
Deal Size: Undisclosed Upfront Cash: Undisclosed
Deal Type: Licensing Agreement March 10, 2026

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Daridorexant,Inapplicable
Therapeutic Area : Sleep
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Pharmalink, S.L.
Deal Size : Undisclosed
Deal Type : Licensing Agreement
Idorsia Expands QUVIVIQ Global Reach with Pharmalink Partnership
Details : Through the licensing deal for Daridorexant, a controlled substance targeting the orexin receptor, the agreement aims to advance treatment options for insomnia.
Product Name : Quviviq
Product Type : Controlled Substance
Upfront Cash : Undisclosed
March 10, 2026

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Through the licensing deal for Daridorexant, a controlled substance targeting the orexin receptor, the agreement aims to advance treatment options for insomnia.
Lead Product(s): Daridorexant,Inapplicable
Therapeutic Area: Sleep Brand Name: Quviviq
Study Phase: Approved FDFProduct Type: Controlled Substance
Sponsor: EMS
Deal Size: Undisclosed Upfront Cash: Undisclosed
Deal Type: Licensing Agreement January 28, 2026

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Daridorexant,Inapplicable
Therapeutic Area : Sleep
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : EMS
Deal Size : Undisclosed
Deal Type : Licensing Agreement
Idorsia's QUVIVIQ expands globally with EMS partnership in Latin America
Details : Through the licensing deal for Daridorexant, a controlled substance targeting the orexin receptor, the agreement aims to advance treatment options for insomnia.
Product Name : Quviviq
Product Type : Controlled Substance
Upfront Cash : Undisclosed
January 28, 2026

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Daridorexant is a Controlled Substance drug candidate, which is currently being evaluated in Phase III clinical studies for the treatment of Sleep Initiation and Maintenance Disorders.
Lead Product(s): Daridorexant,Inapplicable
Therapeutic Area: Sleep Brand Name: Undisclosed
Study Phase: Phase IIIProduct Type: Controlled Substance
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable June 19, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Daridorexant,Inapplicable
Therapeutic Area : Sleep
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Efficacy & Safety of Daridorexant in Participants with Insomnia
Details : Daridorexant is a Controlled Substance drug candidate, which is currently being evaluated in Phase III clinical studies for the treatment of Sleep Initiation and Maintenance Disorders.
Product Name : Undisclosed
Product Type : Controlled Substance
Upfront Cash : Inapplicable
June 19, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Daridorexant is a Controlled Substance drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of undefined medical condition.
Lead Product(s): Daridorexant,Inapplicable
Therapeutic Area: Undisclosed Brand Name: Undisclosed
Study Phase: Phase IProduct Type: Controlled Substance
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable March 22, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Daridorexant,Inapplicable
Therapeutic Area : Undisclosed
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Daridorexant is a Controlled Substance drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of undefined medical condition.
Product Name : Undisclosed
Product Type : Controlled Substance
Upfront Cash : Inapplicable
March 22, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Daridorexant is a Controlled Substance drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Sleep Initiation and Maintenance Disorders.
Lead Product(s): Daridorexant,Inapplicable
Therapeutic Area: Sleep Brand Name: Undisclosed
Study Phase: Phase IIIProduct Type: Controlled Substance
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable August 24, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Daridorexant,Inapplicable
Therapeutic Area : Sleep
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
A Study of Daridorexant in Chinese Patients With Insomnia Disorder
Details : Daridorexant is a Controlled Substance drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Sleep Initiation and Maintenance Disorders.
Product Name : Undisclosed
Product Type : Controlled Substance
Upfront Cash : Inapplicable
August 24, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Daridorexant is a Controlled Substance drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Stress Disorders, Post-Traumatic.
Lead Product(s): Daridorexant,Inapplicable
Therapeutic Area: Psychiatry/Psychology Brand Name: Undisclosed
Study Phase: Phase IIProduct Type: Controlled Substance
Sponsor: U.S. Army Medical Research and Development Command | PPD | Berry Consultants | Idorsia Pharmaceuticals | Cambridge Cognition | Citeline
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable July 17, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Daridorexant,Inapplicable
Therapeutic Area : Psychiatry/Psychology
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : U.S. Army Medical Research and Development Command | PPD | Berry Consultants | Idorsia Pharmaceuticals | Cambridge Cognition | Citeline
Deal Size : Inapplicable
Deal Type : Inapplicable
Department of Defense PTSD Adaptive Platform Trial - Intervention C - Daridorexant
Details : Daridorexant is a Controlled Substance drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Stress Disorders, Post-Traumatic.
Product Name : Undisclosed
Product Type : Controlled Substance
Upfront Cash : Inapplicable
July 17, 2023

Details:
Daridorexant is a Controlled Substance drug candidate, which is currently being evaluated in phase IV clinical studies for the treatment of Alzheimer Disease.
Lead Product(s): Daridorexant,Inapplicable
Therapeutic Area: Neurology Brand Name: Undisclosed
Study Phase: Phase IVProduct Type: Controlled Substance
Sponsor: Idorsia Pharmaceuticals
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable June 29, 2023

Lead Product(s) : Daridorexant,Inapplicable
Therapeutic Area : Neurology
Highest Development Status : Phase IV
Partner/Sponsor/Collaborator : Idorsia Pharmaceuticals
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Daridorexant is a Controlled Substance drug candidate, which is currently being evaluated in phase IV clinical studies for the treatment of Alzheimer Disease.
Product Name : Undisclosed
Product Type : Controlled Substance
Upfront Cash : Inapplicable
June 29, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Daridorexant is a Controlled Substance drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of undefined medical condition.
Lead Product(s): Daridorexant,Inapplicable
Therapeutic Area: Undisclosed Brand Name: Undisclosed
Study Phase: Phase IProduct Type: Controlled Substance
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable May 26, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Daridorexant,Inapplicable
Therapeutic Area : Undisclosed
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
A Phase 1 Trial to Investigate the Biological Equivalence of 5 × 10 mg Tablets and 2 × 25 ...
Details : Daridorexant is a Controlled Substance drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of undefined medical condition.
Product Name : Undisclosed
Product Type : Controlled Substance
Upfront Cash : Inapplicable
May 26, 2023

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]6-CHLORO-2,3-DIAMINO TOLUENE HYDROCHLORIDE
CAS Number : 103146-25-4
End Use API : Daridorexant
About The Company : Founded with a mission to transform strategic capital into specialty chemicals, Ami Group focuses on Agrochemicals, Cosmetics, and Polymers. Ami Organics Ltd. i...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Tablet
Dosage Strength : 25MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
 Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Tablet
Dosage Strength : 50MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Allowed
Registration Country : Switzerland
Brand Name : QUVIVIQ
Dosage Form : Film Coated Tablet
Dosage Strength : 25mg
Packaging :
Approval Date : 01/12/2022
Application Number : 68481
Regulatory Info : Allowed
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Allowed
Registration Country : Switzerland
Brand Name : QUVIVIQ
Dosage Form : Film Coated Tablet
Dosage Strength : 50mg
Packaging :
Approval Date : 01/12/2022
Application Number : 68481
Regulatory Info : Allowed
Registration Country : Switzerland

Regulatory Info : Prescription
Registration Country : Estonia
Brand Name : Quviviq
Dosage Form : Film-Coated Tablet
Dosage Strength : 25mg
Packaging :
Approval Date :
Application Number :
Regulatory Info : Prescription
Registration Country : Estonia

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info : Prescription
Registration Country : Estonia
Brand Name : Quviviq
Dosage Form : Film-Coated Tablet
Dosage Strength : 50mg
Packaging :
Approval Date :
Application Number :
Regulatory Info : Prescription
Registration Country : Estonia

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Allowed
Registration Country : Switzerland
Brand Name : QUVIVIQ
Dosage Form : Film Coated Tablet
Dosage Strength : 25mg
Packaging :
Approval Date : 01/12/2022
Application Number : 68481
Regulatory Info : Allowed
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Allowed
Registration Country : Switzerland
Brand Name : QUVIVIQ
Dosage Form : Film Coated Tablet
Dosage Strength : 50mg
Packaging :
Approval Date : 01/12/2022
Application Number : 68481
Regulatory Info : Allowed
Registration Country : Switzerland

Regulatory Info : Prescription
Registration Country : Estonia
Brand Name : Quviviq
Dosage Form : Film-Coated Tablet
Dosage Strength : 25mg
Packaging :
Approval Date :
Application Number :
Regulatory Info : Prescription
Registration Country : Estonia

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info : Prescription
Registration Country : Estonia
Brand Name : Quviviq
Dosage Form : Film-Coated Tablet
Dosage Strength : 50mg
Packaging :
Approval Date :
Application Number :
Regulatory Info : Prescription
Registration Country : Estonia

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]https://www.pharmacompass.com/radio-compass-blog/dmf-filings-surge-44-in-q1-2025-india-tops-list-with-51-rise-in-year-on-year-submissions
Global Sales Information

Market Place

Reply
26 Apr 2024
Reply
19 Jan 2023
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
ABOUT THIS PAGE
42
PharmaCompass offers a list of Daridorexant API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Daridorexant manufacturer or Daridorexant supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Daridorexant manufacturer or Daridorexant supplier.
A Daridorexant manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Daridorexant, including repackagers and relabelers. The FDA regulates Daridorexant manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Daridorexant API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Daridorexant manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Daridorexant supplier is an individual or a company that provides Daridorexant active pharmaceutical ingredient (API) or Daridorexant finished formulations upon request. The Daridorexant suppliers may include Daridorexant API manufacturers, exporters, distributors and traders.
click here to find a list of Daridorexant suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Daridorexant DMF (Drug Master File) is a document detailing the whole manufacturing process of Daridorexant active pharmaceutical ingredient (API) in detail. Different forms of Daridorexant DMFs exist exist since differing nations have different regulations, such as Daridorexant USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Daridorexant DMF submitted to regulatory agencies in the US is known as a USDMF. Daridorexant USDMF includes data on Daridorexant's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Daridorexant USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Daridorexant suppliers with USDMF on PharmaCompass.
Daridorexant Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Daridorexant GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Daridorexant GMP manufacturer or Daridorexant GMP API supplier for your needs.
A Daridorexant CoA (Certificate of Analysis) is a formal document that attests to Daridorexant's compliance with Daridorexant specifications and serves as a tool for batch-level quality control.
Daridorexant CoA mostly includes findings from lab analyses of a specific batch. For each Daridorexant CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Daridorexant may be tested according to a variety of international standards, such as European Pharmacopoeia (Daridorexant EP), Daridorexant JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Daridorexant USP).

