
Synopsis
Synopsis
0
CEP/COS
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
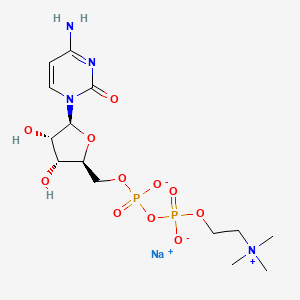

1. Citicoline Sodium
2. 33818-15-4
3. Citicoline Sodium Salt
4. Cytidine 5'-diphosphocholine Sodium
5. Cdp-choline Sodium
6. Citicoline, Sodium
7. Cdp-choline, Sodium
8. Cytidine 5'-diphosphocholine Sodium Salt
9. Cytidine 5'-diphosphocholine Sodium Salt Dihydrate
10. Cytidine-5'-diphosphocholine Monosodium Salt
11. A821952
12. Sodium [[(2s,3r,4s,5s)-5-(4-amino-2-oxo-pyrimidin-1-yl)-3,4-dihydroxy-tetrahydrofuran-2-yl]methoxy-oxido-phosphoryl] 2-(trimethylammonio)ethyl Phosphate;citicoline Sodium Salt
| Molecular Weight | 510.31 g/mol |
|---|---|
| Molecular Formula | C14H25N4NaO11P2 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 10 |
| Exact Mass | 510.08927590 g/mol |
| Monoisotopic Mass | 510.08927590 g/mol |
| Topological Polar Surface Area | 216 Ų |
| Heavy Atom Count | 32 |
| Formal Charge | 0 |
| Complexity | 813 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 4 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
 Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
 SNJ Labs: WHO-GMP certified API leader in India, specializing in bulk drugs & intermediates with unmatched expertise.
SNJ Labs: WHO-GMP certified API leader in India, specializing in bulk drugs & intermediates with unmatched expertise.

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39545
Submission : 2024-02-16
Status : Active
Type : II

Date of Issue : 2025-08-08
Valid Till : 2028-08-07
Written Confirmation Number : WC-0617
Address of the Firm :
 PI Health Sciences: Biotech for Hire providing end-to-end discovery and development powered by chemistry, biology, and AI.
PI Health Sciences: Biotech for Hire providing end-to-end discovery and development powered by chemistry, biology, and AI.
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 27708
Submission : 2013-11-11
Status : Inactive
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 12724
Submission : 1997-10-20
Status : Inactive
Type : II

Registration Number : 307MF10110
Registrant's Address : 1-6-1 Otemachi, Chiyoda-ku, Tokyo
Initial Date of Registration : 2025-08-28
Latest Date of Registration :



Date of Issue : 2025-07-11
Valid Till : 2028-07-02
Written Confirmation Number : WC-0097
Address of the Firm :


Date of Issue : 2024-09-20
Valid Till : 2027-09-19
Written Confirmation Number : WC-592
Address of the Firm :

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
 SNJ Labs: WHO-GMP certified API leader in India, specializing in bulk drugs & intermediates with unmatched expertise.
SNJ Labs: WHO-GMP certified API leader in India, specializing in bulk drugs & intermediates with unmatched expertise.
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39545
Submission : 2024-02-16
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 12724
Submission : 1997-10-20
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 27708
Submission : 2013-11-11
Status : Inactive
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Japanese Pharmacopoeia Citicoline (for manufacturing purposes only)
Registration Number : 307MF10110
Registrant's Address : 1-6-1 Otemachi, Chiyoda-ku, Tokyo
Initial Date of Registration : 2025-08-28
Latest Date of Registration : 2025-08-28

Japanese Pharmacopoeia Citicoline (for manufacturing purposes only)
Registration Number : 217MF10245
Registrant's Address : 1-6-1 Otemachi, Chiyoda-ku, Tokyo
Initial Date of Registration : 2005-06-15
Latest Date of Registration : 2017-04-14

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] SNJ Labs: WHO-GMP certified API leader in India, specializing in bulk drugs & intermediates with unmatched expertise.
SNJ Labs: WHO-GMP certified API leader in India, specializing in bulk drugs & intermediates with unmatched expertise.
Date of Issue : 2025-08-08
Valid Till : 2028-08-07
Written Confirmation Number : WC-0617
Address of the Firm : Survey No. 247/1, Village: Hadala, Taluka: Rajkot,\r\nDistrict. -Rajkot, Gujarat...
Date of Issue : 2024-06-10
Valid Till : 2027-06-09
Written Confirmation Number : WC-481
Address of the Firm : Vill.Jaula Khurad, Tehsil Derabassi, Distt.Sahibzada Ajit Singh Nagar, Punjab

Date of Issue : 2024-06-10
Valid Till : 2027-06-09
Written Confirmation Number : WC-0481
Address of the Firm : Vill.Jaula Khurad, Tehsil Derabassi, Distt.Sahibzada Ajit Singh Nagar, Punjab

Date of Issue : 2024-09-20
Valid Till : 2027-09-19
Written Confirmation Number : WC-592
Address of the Firm : Plot No. 411/1, L.I.C. Sector, Silvassa Road, G.I.D.C., Vapi, Dist -Valsad -3961...

Date of Issue : 2025-07-11
Valid Till : 2028-07-02
Written Confirmation Number : WC-0097
Address of the Firm : Block No. 251/B, 252/B, 253, 254, 255, 256, 258/B, 276, 277, 278/P, 279, 280, 28...

Date of Issue : 2025-07-11
Valid Till : 2028-07-02
Written Confirmation Number : WC-0097
Address of the Firm : Block No. 251/B, 252/B, 253, 254, 255, 256, 258/B, 276, 277, 278/P, 279, 280, 28...

Date of Issue : 2022-12-30
Valid Till : 2025-12-29
Written Confirmation Number : WC-05450
Address of the Firm : PlotNo.:SP4-4,Industrial Area,Keshwana Rajput, Shahpura, Distt.Jaipur-303108, Ra...

Date of Issue : 2022-12-30
Valid Till : 2025-12-29
Written Confirmation Number : WC-0545
Address of the Firm : Plot No.: SP4-4, Industrial Area, Keshwana Rajput, Shahpura, Distt. Jaipur-30310...

Date of Issue : 2024-05-29
Valid Till : 2027-03-04
Written Confirmation Number : WC-0294
Address of the Firm : 120 A & B, 120P and 121, Industrial Area, Baikampady, New Mangalore -575011, Ind...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
About the Company : LGM Pharma is a global leader in sourcing APIs, including hard-to-find drug substances, for pharmaceutical and biotech industries. LGM also operates as a full-service drug product ...
About the Company : Jai Radhe Sales, founded in 1999, is a global distributor specializing in high-quality pharmaceutical ingredients from India. It offers complete sourcing solutions, technical and r...
 Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
About the Company : Tenatra International was established as a proprietorship firm in 1999. It got off to a very good start, supporting clients in the United States, Mexico and Europe. As business opp...
 SNJ Labs: WHO-GMP certified API leader in India, specializing in bulk drugs & intermediates with unmatched expertise.
SNJ Labs: WHO-GMP certified API leader in India, specializing in bulk drugs & intermediates with unmatched expertise.
About the Company : SNJ Labs Pvt. Ltd., established in 2013, is India’s largest manufacturer of Iron Sucrose, achieving a production capacity of 120 MT per year within five years. The company specia...
 PI Health Sciences: Biotech for Hire providing end-to-end discovery and development powered by chemistry, biology, and AI.
PI Health Sciences: Biotech for Hire providing end-to-end discovery and development powered by chemistry, biology, and AI.
About the Company : PI Health Sciences offers end-to-end drug discovery and development, integrating medicinal chemistry, synthetic chemistry, biology, and AI-driven technologies. Its co-located teams...
About the Company : Enaltec, founded in 2006, specializes in developing and manufacturing complex, small-volume, technology-driven APIs at competitive prices, leveraging the India advantage for global...
About the Company : Aspire group was established in the year 2000 with a motto of quality drug for better health of all. In its journey till date, aspire has developed enormous reputation, consistent ...

About the Company : As an EU-GMP certified global company and an established hallmark for pharmaceutical standards, Global Calcium has stood the test of time since its inception in 1979 as Calcium Ind...

About the Company : MANA PHARMA, S.L. is a Spanish Pharmaceutical Company, with Authorization as a Wholesale Pharmaceutical Distribution Entity of Medicines for Human Use as well as a Certificate of G...

About the Company : Sudarshan Pharma operates in the pharmaceutical and specialty chemicals industries. Their specialty chemicals and intermediates are used in pharma, paint, food, and adhesive sector...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Market Place

Reply
21 Apr 2026
Reply
07 Apr 2026
Reply
16 Oct 2025
Reply
18 Mar 2025
Reply
27 Mar 2024
Reply
05 Mar 2024
Reply
09 Nov 2023
Reply
31 Oct 2023
Reply
19 Oct 2023
Reply
09 Oct 2023
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
39
PharmaCompass offers a list of Citicoline Sodium API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Citicoline Sodium manufacturer or Citicoline Sodium supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Citicoline Sodium manufacturer or Citicoline Sodium supplier.
A Cytidine Diphosphate Choline manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Cytidine Diphosphate Choline, including repackagers and relabelers. The FDA regulates Cytidine Diphosphate Choline manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Cytidine Diphosphate Choline API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Cytidine Diphosphate Choline manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Cytidine Diphosphate Choline supplier is an individual or a company that provides Cytidine Diphosphate Choline active pharmaceutical ingredient (API) or Cytidine Diphosphate Choline finished formulations upon request. The Cytidine Diphosphate Choline suppliers may include Cytidine Diphosphate Choline API manufacturers, exporters, distributors and traders.
click here to find a list of Cytidine Diphosphate Choline suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Cytidine Diphosphate Choline DMF (Drug Master File) is a document detailing the whole manufacturing process of Cytidine Diphosphate Choline active pharmaceutical ingredient (API) in detail. Different forms of Cytidine Diphosphate Choline DMFs exist exist since differing nations have different regulations, such as Cytidine Diphosphate Choline USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Cytidine Diphosphate Choline DMF submitted to regulatory agencies in the US is known as a USDMF. Cytidine Diphosphate Choline USDMF includes data on Cytidine Diphosphate Choline's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Cytidine Diphosphate Choline USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Cytidine Diphosphate Choline suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Cytidine Diphosphate Choline Drug Master File in Japan (Cytidine Diphosphate Choline JDMF) empowers Cytidine Diphosphate Choline API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Cytidine Diphosphate Choline JDMF during the approval evaluation for pharmaceutical products. At the time of Cytidine Diphosphate Choline JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Cytidine Diphosphate Choline suppliers with JDMF on PharmaCompass.
A Cytidine Diphosphate Choline written confirmation (Cytidine Diphosphate Choline WC) is an official document issued by a regulatory agency to a Cytidine Diphosphate Choline manufacturer, verifying that the manufacturing facility of a Cytidine Diphosphate Choline active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Cytidine Diphosphate Choline APIs or Cytidine Diphosphate Choline finished pharmaceutical products to another nation, regulatory agencies frequently require a Cytidine Diphosphate Choline WC (written confirmation) as part of the regulatory process.
click here to find a list of Cytidine Diphosphate Choline suppliers with Written Confirmation (WC) on PharmaCompass.
Cytidine Diphosphate Choline Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Cytidine Diphosphate Choline GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Cytidine Diphosphate Choline GMP manufacturer or Cytidine Diphosphate Choline GMP API supplier for your needs.
A Cytidine Diphosphate Choline CoA (Certificate of Analysis) is a formal document that attests to Cytidine Diphosphate Choline's compliance with Cytidine Diphosphate Choline specifications and serves as a tool for batch-level quality control.
Cytidine Diphosphate Choline CoA mostly includes findings from lab analyses of a specific batch. For each Cytidine Diphosphate Choline CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Cytidine Diphosphate Choline may be tested according to a variety of international standards, such as European Pharmacopoeia (Cytidine Diphosphate Choline EP), Cytidine Diphosphate Choline JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Cytidine Diphosphate Choline USP).

