
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
KDMF
0
VMF
0
FDA Orange Book

0
Europe

0
Australia

0
South Africa

DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
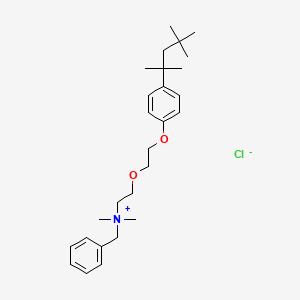

1. Bencetonium Chloride
2. Benzethonium
3. Chloride, Bencetonium
4. Chloride, Benzethonium
5. Formula Magic
6. Hyamine 1622
7. Magic, Formula
8. Orchid Fresh Ii
9. Phemeride
10. Phemerol
11. Phemethryn
12. Puri Clens
13. Puri-clens
14. Puriclens
15. Quatrachlor
16. Solamin
1. 121-54-0
2. Hyamine
3. Phemeride
4. Phemerol Chloride
5. Quatrachlor
6. Benzethoniumchloride
7. Hyamine 1622
8. Banagerm
9. Phemerol
10. Phemithyn
11. Disilyn
12. Kylacol
13. Diapp
14. Polymine D
15. Benzetonium Chloride
16. Anti-germ 77
17. Benzethonii Chloridum
18. Antiseptol
19. Cloruro De Benzetonio
20. Inactisol
21. Neostelin Green
22. Chlorure De Benzethonium
23. Sanizol
24. Benzethonium Chloride 1622
25. Microklenz
26. P-tert-octylphenoxyethoxyethyldimethylbenzylammonium Chloride
27. Nsc-20200
28. 5929-09-9
29. Benzyl-dimethyl-[2-[2-[4-(2,4,4-trimethylpentan-2-yl)phenoxy]ethoxy]ethyl]azanium;chloride
30. Diisobutylphenoxyethoxyethyl Dimethyl Benzyl Ammonium Chloride
31. Diisobutylphenoxyethoxyethyldimethyl Benzyl Ammonium Chloride
32. Chebi:31264
33. Phemerol Chloride Monohydrate
34. N-benzyl-n,n-dimethyl-2-(2-(4-(2,4,4-trimethylpentan-2-yl)phenoxy)ethoxy)ethanaminium Chloride
35. Ph41d05744
36. 1313-08-2
37. Benzenemethanaminium, N,n-dimethyl-n-(2-(2-(4-(1,1,3,3-tetramethylbutyl)phenoxy)ethoxy)ethyl)-, Chloride
38. P-diisobutyl Phenoxyethoxyethyl Dimethyl Benzylammonium Chloride
39. (2-(2-(4-diisobutylphenoxy)ethoxy)ethyl)dimethylbenzylammonium Chloride
40. Dsstox_cid_3810
41. 121-54-0 (cl-)
42. Benzyldimethyl-p-(1,1,3,3-tetramethylbutyl)phenoxyethoxy-ethylammonium Chloride
43. Benzetonio Cloruro
44. Benzyldimethyl(2-(2-(p-(1,1,3,3-tetramethylbutyl)phenoxy)ethoxy)ethyl)ammonium Chloride
45. Dsstox_rid_77195
46. Dsstox_gsid_23810
47. Benzethoni Chloridum
48. N,n-dimethyl-n-(2-(2-(4-(1,1,3,3-tetramethylbutyl)phenoxy)ethoxy)ethyl)-benzenemethanaminium Chloride
49. N-benzyl-n,n-dimethyl-2-(2-(4-(2,4,4-trimethylpentan-2-yl)phenoxy)ethoxy)ethan-1-aminium Chloride
50. Qac
51. P-diisobutyl(phenoxyethoxy)ethyl]dimethylbenzylammonium Chloride
52. Chembl221753
53. Rs89wu8v92
54. Benzyldimethyl(2-(2-(4-(1,1,3,3-tetramethylbutyl)phenoxy)ethoxy)ethyl)ammonium Chloride
55. Benzenemethanaminium, N,n-dimethyl-n-(2-(2-(4-(1,1,3,3-tetramethylbutyl)phenoxy)ethoxy)ethyl)-, Chloride (1:1)
56. Formula 144
57. Nsc20200
58. Caswell No. 614b
59. Sr-05000001572
60. Ncgc00016373-03
61. Ammonium, Benzyldimethyl(2-(2-(p-(1,1,3,3-tetramethylbutyl)phenoxy)ethoxy)ethyl)-, Chloride, Monohydrate
62. Cas-121-54-0
63. Mfcd00011742
64. Bzt (van)
65. Benzetonio Cloruro [dcit]
66. Salanine
67. Ccris 4748
68. Hsdb 567
69. [2-[2-(4-diisobutylphenoxy)ethoxy]ethyl]dimethylbenzylammonium Chloride
70. {2-[2-(4-diisobutylphenoxy)ethoxy]ethyl}dimethylbenzylammonium Chloride
71. Unii-ph41d05744
72. Antiseptol (quarternary Compound)
73. Phemersol Chloride
74. Benzyldimethyl(2-{2-[4-(1,1,3,3-tetramethylbutyl)phenoxy]ethoxy}ethyl)ammonium Chloride
75. Benzyldimethyl(2-{2-[p-(1,1,3,3-tetramethylbutyl)phenoxy]ethoxy}ethyl)ammonium Chloride
76. Microklenz (tn)
77. Benzethonii Chloridum [inn-latin]
78. Nci-c61494
79. Benzenemethanaminium, N,n-dimethyl-n-[2-[2-[4-(1,1,3,3-tetramethylbutyl)phenoxy]ethoxy]ethyl]-, Chloride (1:1)
80. N,n-dimethyl-n-(2-{2-[4-(1,1,3,3-tetramethylbutyl)phenoxy]ethoxy}ethyl)-benzenemethanaminium Chloride
81. N-benzyl-n,n-dimethyl-2-{2-[4-(2,4,4-trimethylpentan-2-yl)phenoxy]ethoxy}ethanaminium Chloride
82. Prestwick_995
83. Einecs 204-479-9
84. Hyamine (tn)
85. Cloruro De Benzetonio [inn-spanish]
86. Nsc 20200
87. Chlorure De Benzethonium [inn-french]
88. Epa Pesticide Chemical Code 069154
89. Neostelin Green (tn)
90. Benzethonium Chloride [usp:inn:ban:jan]
91. Ec 204-479-9
92. Benzethonium Chloride, 97%
93. Benzyldimethyl(2-{2-[4-(2,4,4-trimethylpentan-2-yl)phenoxy]ethoxy}ethyl)azanium Chloride
94. Unii-rs89wu8v92
95. Benzethonium Chloride Hydrate
96. Schembl21713
97. Mls002153968
98. Spectrum1500138
99. Dtxsid6023810
100. Hms502g17
101. Benzethonium Chloride [ii]
102. Benzethonium Chloride [mi]
103. Hms1570m17
104. Hms1920g07
105. Hms2091m09
106. Hms2097m17
107. Hms2230m22
108. Hms3373h09
109. Hms3652d03
110. Hms3714m17
111. Hms3885p07
112. Pharmakon1600-01500138
113. Benzethonium Chloride [inn]
114. Benzethonium Chloride [jan]
115. Diisobutyl Phenoxy Ethoxy Ethyl Dimethyl Benzyl Ammonium Chloride
116. Hy-b0942
117. Benzethonium Chloride [hsdb]
118. Benzethonium Chloride [inci]
119. Tox21_110403
120. Tox21_202488
121. Benzethonium Chloride [vandf]
122. Ccg-39713
123. Nsc755908
124. P-diisobutylphenoxyethoxyethyldimethylbenzylammonium Chloride Monohydrate
125. S4162
126. Benzethonium Chloride [mart.]
127. 2-(2-(p-(diisobutyl)phenoxy)ethoxy)ethyl Dimethyl Benzyl Ammonium Chloride
128. Akos005287417
129. Benzethonium Chloride [usp-rs]
130. Benzethonium Chloride [who-dd]
131. Tox21_110403_1
132. Nsc-755908
133. Ammonium, Benzyldimethyl(2-(2-(p-(1,1,3,3-tetramethylbutyl)phenoxy)ethoxy)ethyl)-, Chloride
134. Benzethonium Chloride (jp17/usp/inn)
135. Ncgc00016373-08
136. Ncgc00091528-01
137. Ncgc00091528-02
138. Ncgc00094597-01
139. Ncgc00094597-02
140. Ncgc00094597-03
141. Ncgc00094597-04
142. Ncgc00260037-01
143. As-14646
144. Smr001233307
145. Benzethonium Chloride [ep Impurity]
146. Benzethonium Chloride [ep Monograph]
147. Benzethonium Chloride [usp Impurity]
148. Db-053360
149. B0044
150. Ft-0622657
151. Ft-0635910
152. Hyamine(r) 1622 Solution, 4 Mm In H2o
153. Sw197077-3
154. A13653
155. Benzethonium Chloride, Tested According To Usp
156. D01140
157. N11901
158. Benzethonium Chloride 1000 Microg/ml In Methanol
159. Benzethonium Chloride, Bioultra, >=99.0% (at)
160. Q425119
161. J-200196
162. Sr-05000001572-1
163. Sr-05000001572-3
164. Benzethonium Chloride Concentrate [usp Monograph]
165. F2173-1223
166. Z1317839149
167. Benzethonium Chloride, >=97% (titration), >=98% (hplc)
168. [diisobutyl(phenoxyethoxy)ethyl]dimethylbenzylammonium Chloride
169. P-diisobutylphenoxyethoxyethyl Dimethyl Benzyl Ammonium Chloride
170. [[(diisobutyl)phenoxyethoxyethyl]dimethylbenzyl]ammonium Chloride
171. [[(diisobutylphenoxy)ethoxy]ethyl] Dimethyl Benzyl Ammonium Chloride
172. Ammonium,1,3,3-tetramethylbutyl)phenoxy]ethoxy]ethyl]-, Chloride
173. Ammonium,1,3,3-tetramethylbutyl)phenoxy]ethoxy]ethyl]-,chloride
174. Benzethonium Chloride, European Pharmacopoeia (ep) Reference Standard
175. Benzethonium Chloride, United States Pharmacopeia (usp) Reference Standard
176. Benzethonium Chloride, Pharmaceutical Secondary Standard; Certified Reference Material
177. Benzyldimethyl-[[[p-(1,3,3-tetramethylbutyl)phenoxy]ethoxy]ethyl]ammonium Chloride
178. Benzyldimethyl[2-[2-[4-(1,3,3-tetramethylbutyl)phenoxy]ethoxy]ethyl]ammonium Chloride
179. Benzyldimethyl[2-[2-[p-(1,3,3-tetramethylbutyl)phenoxy]ethoxy]ethyl]ammonium Chloride
180. Wln: 1x1 & 1 & 1x1 & 1 & R Do2o2k1 & 1 & 1r & Q & G
181. Benzenemethanaminium, N,n-dimethyl-n-(2-(2-(4-(1,1,3,3-tetramethylbutyl)phenoxy)ethoxy)- Ethyl)-, Chloride
182. Benzenemethanaminium, N,n-dimethyl-n-(2-(2-(4-(1,1,3,3-tetramethylbutyl)phenoxy)ethoxy)ethyl)-, Chloride, Monohydrate
183. Benzenemethanaminium,n-dimethyl-n-[2-[2-[4-(1,1,3,3-tetramethylbutyl)phenoxy]ethoxy]ethyl]-, Chloride
184. N,n-dimethyl-n-(2-(2-(4-(1,1,3,3-tetramethylbutyl)phenoxy)ethoxy)- Ethyl)benzenemethanaminium Chloride
185. N-benzyl-n,n-dimethyl-2-(2-(4-(2,4,4-trimethylpentan-2-yl)phenoxy)ethoxy)ethanaminiumchloride
| Molecular Weight | 448.1 g/mol |
|---|---|
| Molecular Formula | C27H42ClNO2 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 12 |
| Exact Mass | 447.2904073 g/mol |
| Monoisotopic Mass | 447.2904073 g/mol |
| Topological Polar Surface Area | 18.5 Ų |
| Heavy Atom Count | 31 |
| Formal Charge | 0 |
| Complexity | 466 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Anti-Infective Agents, Local
Substances used on humans and other animals that destroy harmful microorganisms or inhibit their activity. They are distinguished from DISINFECTANTS, which are used on inanimate objects. (See all compounds classified as Anti-Infective Agents, Local.)
D - Dermatologicals
D08 - Antiseptics and disinfectants
D08A - Antiseptics and disinfectants
D08AJ - Quaternary ammonium compounds
D08AJ08 - Benzethonium chloride
PERCUTANEOUS ABSORPTION IS PROBABLY INSIGNIFICANT. /BENZALKONIUM CHLORIDE/
Gosselin, R.E., R.P. Smith, H.C. Hodge. Clinical Toxicology of Commercial Products. 5th ed. Baltimore: Williams and Wilkins, 1984., p. II-280

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Related Excipient Companies

Excipients by Applications
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
82
PharmaCompass offers a list of Benzethonium Chloride API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Benzethonium Chloride manufacturer or Benzethonium Chloride supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Benzethonium Chloride manufacturer or Benzethonium Chloride supplier.
PharmaCompass also assists you with knowing the Benzethonium Chloride API Price utilized in the formulation of products. Benzethonium Chloride API Price is not always fixed or binding as the Benzethonium Chloride Price is obtained through a variety of data sources. The Benzethonium Chloride Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Benzethonium manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Benzethonium, including repackagers and relabelers. The FDA regulates Benzethonium manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Benzethonium API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Benzethonium manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Benzethonium supplier is an individual or a company that provides Benzethonium active pharmaceutical ingredient (API) or Benzethonium finished formulations upon request. The Benzethonium suppliers may include Benzethonium API manufacturers, exporters, distributors and traders.
click here to find a list of Benzethonium suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Benzethonium Drug Master File in Japan (Benzethonium JDMF) empowers Benzethonium API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Benzethonium JDMF during the approval evaluation for pharmaceutical products. At the time of Benzethonium JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Benzethonium suppliers with JDMF on PharmaCompass.
A Benzethonium written confirmation (Benzethonium WC) is an official document issued by a regulatory agency to a Benzethonium manufacturer, verifying that the manufacturing facility of a Benzethonium active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Benzethonium APIs or Benzethonium finished pharmaceutical products to another nation, regulatory agencies frequently require a Benzethonium WC (written confirmation) as part of the regulatory process.
click here to find a list of Benzethonium suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Benzethonium as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Benzethonium API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Benzethonium as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Benzethonium and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Benzethonium NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Benzethonium suppliers with NDC on PharmaCompass.
Benzethonium Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Benzethonium GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Benzethonium GMP manufacturer or Benzethonium GMP API supplier for your needs.
A Benzethonium CoA (Certificate of Analysis) is a formal document that attests to Benzethonium's compliance with Benzethonium specifications and serves as a tool for batch-level quality control.
Benzethonium CoA mostly includes findings from lab analyses of a specific batch. For each Benzethonium CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Benzethonium may be tested according to a variety of international standards, such as European Pharmacopoeia (Benzethonium EP), Benzethonium JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Benzethonium USP).
