Acquisitions and spin-offs dominated headlines in 2019 and the tone was set very early with Bristol-Myers Squibb acquiring
New Jersey-based cancer drug company Celgene in a US$ 74 billion deal announced on
January 3, 2019. After factoring
in debt, the deal value ballooned to about US$ 95 billion, which according
to data compiled by Refinitiv, made it the largest healthcare deal on
record.
In the summer, AbbVie Inc,
which sells the world’s best-selling drug Humira, announced its acquisition of Allergan Plc, known for Botox and other cosmetic
treatments, for US$ 63 billion. While the companies are still awaiting
regulatory approval for their deal, with US$ 49 billion in combined 2019
revenues, the merged entity would rank amongst the biggest in the industry.
View Our Interactive Dashboard on Top drugs by sales in 2019 (Free Excel Available)
The big five by pharmaceutical sales — Pfizer,
Roche, J&J, Novartis and Merck
Pfizer
continued
to lead companies by pharmaceutical sales by reporting annual 2019 revenues of
US$ 51.8 billion, a decrease of US$ 1.9 billion, or 4 percent, compared to
2018. The decline was primarily attributed to the loss of exclusivity of Lyrica in 2019,
which witnessed its sales drop from US$ 5 billion in 2018 to US$ 3.3 billion in
2019.
In 2018, Pfizer’s then incoming CEO Albert Bourla had mentioned that the company did not see the need for any large-scale M&A activity as Pfizer had “the best pipeline” in its history, which needed the company to focus on deploying its capital to keep its pipeline flowing and execute on its drug launches.
Bourla stayed true to his word and barring the acquisition of Array Biopharma for US$ 11.4 billion and a spin-off to merge Upjohn, Pfizer’s off-patent branded and generic established medicines business with
Mylan, there weren’t any other big ticket deals which were announced.
The
Upjohn-Mylan merged entity will be called Viatris and is expected to have 2020
revenues between US$ 19 and US$ 20 billion
and could outpace Teva to
become the largest generic company in the world, in term of revenues.
Novartis, which had
followed Pfizer with the second largest revenues in the pharmaceutical industry
in 2018, reported its first full year earnings after spinning off its Alcon eye
care devices business division that
had US$ 7.15 billion in 2018 sales.
In 2019,
Novartis slipped two spots in the ranking after reporting total sales of US$
47.4 billion and its CEO Vas Narasimhan continued his deal-making spree by buying New
Jersey-headquartered The Medicines Company (MedCo) for US$ 9.7
billion to acquire a late-stage cholesterol-lowering
therapy named inclisiran.
As Takeda Pharmaceutical Co was
busy in 2019 on working to reduce its debt burden incurred due to its US$ 62
billion purchase of Shire Plc, which was announced in 2018, Novartis also purchased
the eye-disease medicine, Xiidra, from the Japanese drugmaker for US$ 5.3 billion.
Novartis’ management also spent a considerable part of 2019 dealing with data-integrity concerns which emerged from its 2018 buyout of AveXis, the
gene-therapy maker Novartis had acquired for US$ 8.7 billion.
The deal gave Novartis rights to Zolgensma,
a novel treatment intended for children less than two years of age with the
most severe form of spinal muscular atrophy (SMA). Priced at US$ 2.1 million,
Zolgensma is currently the world’s most expensive drug.
However,
in a shocking announcement, a month after approving the drug, the US Food and
Drug Administration (FDA) issued a press release on
data accuracy issues as the agency was informed by AveXis that
its personnel had manipulated data which
the FDA used to evaluate product comparability and nonclinical (animal)
pharmacology as part of the biologics license application (BLA), which was
submitted and reviewed by the FDA.
With US$
50.0 billion (CHF 48.5 billion) in annual pharmaceutical sales, Swiss drugmaker
Roche came in at number two position in 2019
as its sales grew 11 percent driven by
its multiple sclerosis medicine Ocrevus, haemophilia drug Hemlibra and cancer medicines Tecentriq and Perjeta.
Roche’s newly introduced medicines generated US$ 5.53 billion (CHF 5.4 billion) in growth, helping offset the impact of the competition from biosimilars for its three best-selling drugs MabThera/Rituxan, Herceptin and Avastin.
In late 2019, after months of increased
antitrust scrutiny, Roche completed
its US$ 5.1 billion acquisition of Spark Therapeutics to strengthen its presence in
gene therapy.
Last year, J&J reported almost flat worldwide sales of US$ 82.1 billion. J&J’s pharmaceutical division generated US$ 42.20 billion and its medical devices and consumer health divisions brought in US$ 25.96 billion and US$ 13.89 billion respectively.
Since J&J’s consumer health division sells analgesics, digestive health along with beauty and oral care products, the US$ 5.43 billion in consumer health sales from over-the-counter drugs and women’s health products was only used in our assessment of J&J’s total pharmaceutical revenues. With combined pharmaceutical sales of US$ 47.63 billion, J&J made it to number three on our list.
While the sales of products like Stelara, Darzalex, Imbruvica, Invega Sustenna drove J&J’s pharmaceutical business to grow by 4 percent over 2018, the firm had to contend with generic competition against key revenue contributors Remicade and Zytiga.
US-headquartered Merck, which is known as
MSD (short for Merck Sharp & Dohme) outside the United States and
Canada, is set to significantly move up the rankings next year fueled by its
cancer drug Keytruda, which witnessed a 55
percent increase in sales to US$ 11.1 billion.
Merck reported total revenues of US$ 41.75 billion and also
announced it will spin off its women’s health drugs,
biosimilar drugs and older products to create a new pharmaceutical
company with US$ 6.5 billion in annual revenues.
The firm had anticipated 2020 sales between US$ 48.8 billion and US$ 50.3 billion however this week it announced that the coronavirus pandemic will reduce 2020 sales by more than $2 billion.
View Our Interactive Dashboard on Top drugs by sales in 2019 (Free Excel Available)
Humira holds on to remain world’s best-selling drug
AbbVie’s acquisition of Allergan comes as the firm faces the expiration of patent protection for Humira, which brought in a staggering US$ 19.2 billion in sales last year for
the company. AbbVie has failed to successfully acquire or develop a major new
product to replace the sales generated by its flagship drug.
In 2019, Humira’s US revenues increased 8.6 percent to US$ 14.86 billion while internationally, due
to biosimilar competition, the sales dropped 31.1 percent to US$ 4.30 billion.
Bristol Myers Squibb’s Eliquis, which is also marketed by Pfizer, maintained its number two position
and posted total sales of US$ 12.1 billion, a 23 percent increase over 2018.
While Bristol Myers Squibb’s immunotherapy treatment Opdivo, sold in partnership with Ono in Japan, saw sales increase from US$ 7.57 billion to US$ 8.0 billion, the growth paled in comparison to the US$ 3.9
billion revenue increase of Opdivo’s key immunotherapy competitor Merck’s Keytruda.
Keytruda took the number three spot in drug sales that
previously belonged to Celgene’s Revlimid, which witnessed a sales decline from US$ 9.69 billion to US$ 9.4 billion.
Cancer treatment Imbruvica, which is marketed
by J&J and AbbVie, witnessed a 30 percent increase in sales. With US$ 8.1
billion in 2019 revenues, it took the number five position.
View Our Interactive Dashboard on Top drugs by sales in 2019 (Free Excel Available)
Vaccines – Covid-19 turns competitors into partners
This year has been dominated by the single biggest health emergency in years — the novel coronavirus (Covid-19) pandemic. As drugs continue to fail to meet expectations, vaccine development has received a lot of attention.
GSK reported the highest vaccine sales of all drugmakers with
total sales of US$ 8.4 billion (GBP 7.16 billion), a significant portion of its
total sales of US$ 41.8 billion (GBP 33.754 billion).
US-based Merck’s vaccine division also reported a significant increase in sales to US$ 8.0 billion and in 2019 received FDA and EU approval to market its Ebola vaccine Ervebo.
This is the first FDA-authorized vaccine against the deadly virus which causes
hemorrhagic fever and spreads from person to person through direct contact with
body fluids.
Pfizer and Sanofi also reported an increase in their vaccine sales to US$ 6.4
billion and US$ 6.2 billion respectively and the Covid-19 pandemic has recently
pushed drugmakers to move faster than ever before and has also converted
competitors into partners.
In a rare move, drug behemoths — Sanofi and GlaxoSmithKline (GSK) —joined hands to develop a vaccine for the novel coronavirus.
The two companies plan to start human trials
in the second half of this year, and if things go right, they will file
for potential approvals by the second half of 2021.
View Our Interactive Dashboard on Top drugs by sales in 2019 (Free Excel Available)
Our view
Covid-19 has brought the world economy to a grinding halt and shifted the global attention to the pharmaceutical industry’s capability to deliver solutions to address this pandemic.
Our compilation shows that vaccines and drugs
for infectious diseases currently form a tiny fraction of the total sales of
pharmaceutical companies and few drugs against infectious diseases rank high on
the sales list.
This could well explain the limited range of
options currently available to fight Covid-19. With the pandemic currently infecting
over 3 million people spread across more than 200 countries, we can safely
conclude that the scenario in 2020 will change substantially. And so should our
compilation of top drugs for the year.
View Our Interactive Dashboard on Top drugs by sales in 2019 (Free Excel Available)
Impressions: 54754
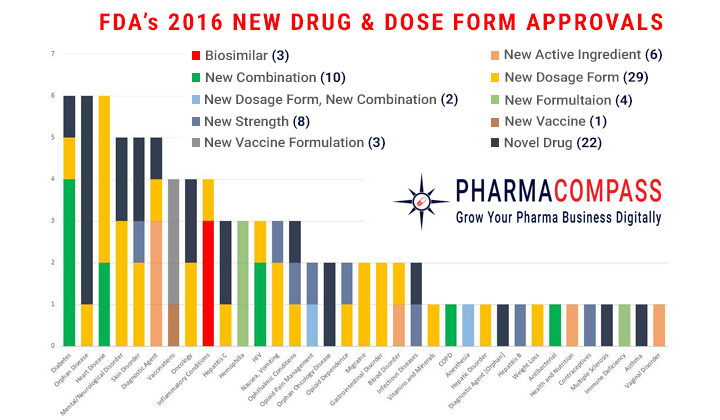
After 2 years of sky-high approval numbers, the US Food and Drug Administration’s (FDA) Center for Drug Evaluation and Research approved 22 novel drugs in 2016, down from the 19-year high of 45 in 2015. The FDA also approved many new dose forms, formulations, combination products and vaccines.
This
week, PharmaCompass, shares its analysis of the new drug approvals by the FDA
in 2016.
Reasons behind the low approvals in 2016
Of the 22 novel drugs approved by the FDA, the FDA approved 9 products with orphan designation, in line with the industry’s recent focus on rare diseases. However, as the industry shifts its focus towards biotechnology, only 7 of the novel products approved were biologic applications.
The 9
orphan designees approved (41% of all new drug approvals) were significantly lower than the 21 (47%) orphan designees approved in 2015 and 17 (41%) in 2014. FDA’s approval of 4 (18%) oncology drugs in 2016, was also down from the 14 (31%) approvals in 2015, 9 (22%) in 2014 and 9 (33%) in 2013.
The
reasons for the low 2016 approval count, have been attributed to the agency
approving five drugs in 2015 that actually had approval action dates in 2016
and an increase in the number of drugs that the agency rejected.
A key reason for the rejections was the sponsors’ failure to comply with good manufacturing practice regulations.
Click here to access our list of all New Drug Approvals (Excel version available) for FREE!
Gilead’s Epclusa, the first
all-oral, pan-genotypic, single tablet regimen for the treatment of adults with
chronic hepatitis C virus (HCV) infection, is expected to become the most
profitable approval of 2016 with expected sales of $ 8.4 billion by 2022.
2016 also saw the FDA’s contentious approval of Exondys 51™ (eteplirsen) under its accelerated approval process. Exondys 51™ treats Duchenne muscular dystrophy (DMD), a rare genetic disease which causes progressive muscle wasting that affects around 20,000 boys and young men in the United States.
Approvals of three biosimilars which
target $ 33 billion in brand drug sales
In a
giant leap for the generic pharmaceutical industry, three biosimilar
applications were approved in 2016 that target brand drugs which generated more
than $ 33 billion in sales last year.
All the three biosimilars approved were tumor-necrosis factor alpha (TNF α) inhibitors used to manage inflammatory conditions.
Pfizer and Celltrion’s Inflectra, a
biosimilar to Janssen’s Remicade® (2016 sales - $ 8.8 billion), was the first to get approval for all Remicade-approved indications, except pediatric ulcerative colitis. Inflectra was launched at risk in November 2016.
After Inflectra’s approval, the FDA approved Sandoz’s Erelzi, a biosimilar to Enbrel® (etanercept – Amgen, 2016 sales - $
9.1 billion).
On Sept. 23, 2016, Amgen’s Amjevita™ (adalimumab – atto), a biosimilar of the world’s best-selling drug by revenue, AbbVie’s Humira®(2016 sales - $ 16.4 billion), was also approved for treating adults with a variety of medical conditions ranging from rheumatoid arthritis, plaque psoriasis, to ulcerative colitis.
Click here to access our list of all New Drug Approvals (Excel version available) for FREE!
Kim Kardashian’s pregnancy drug is now available in a new strength
Diclegis,
a combination of an antihistamine (doxylamine succinate) and a form of vitamin B6 (pyridoxine HCl), made headlines
when the FDA issued a warning letter for the social media promotion of the drug by Kim Kardashain, star of the reality show ‘Keeping Up With The Kardashians’
Duchesnay,
the company which produces Diclegis, got approval for Bonjesta which combines twice
the amount of doxylamine and pyridoxine when compared with Diclegis which contains
10mg of each ingredient.
Indian generic companies get approvals for
differentiated products
As the
generic industry in the U.S. continues to face severe pricing pressure, Indian
generic companies are attempting to overcome these challenges by trying to move
up the value chain by supplying differentiated generic products.
Consistent
with this strategy, the applications of Sun Pharmaceuticals
for a new ophthalmic version of Bromfenac (Bromsite) and Dr. Reddy’s Laboratory
for an injectable version of Sumatriptan (Zembrace™ SymTouch) to treat migraines were also approved.
Click here to access our list of all New Drug Approvals (Excel version available) for FREE!
Pfizer launches an opioid treatment with
abuse-deterrent properties
The abuse
of opioids, including prescription painkillers and drugs like heroin, is
something the United States has struggled with since before the 1900s. Last year, the FDA announced that immediate-release
opioid painkillers such as oxycodone and fentanyl will have to
carry a "black box" warning
about the risk of abuse, addiction, overdose and death.
Pfizer
got approval last year for Troxyca ER a combination containing oxycodone and naltrexone. Troxyca ER has properties that are
expected to reduce abuse when crushed and administered by the oral and
intranasal routes.
In it,
the oxycodone releases slowly over several hours. If the capsules are crushed,
the encased naltrexone mixes with oxycodone, essentially cancelling any euphoric
effects.
Pfizer
also received approval for an extended release form of Tofacitinib (Xeljanz® XR) to treat adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response or intolerance to methotrexate.
Click here to access our list of all New Drug Approvals (Excel version available) for FREE!
A dietary supplement gets approved along with new forms of commonly used drugs
In 2016, Endoceutics
got Intarosa, a once-daily vaginal insert, approved which is the first FDA
approved product to contain prasterone, also known as dehydroepiandrosterone (DHEA).
Although
DHEA is included in some dietary supplements, the efficacy and safety of those
products have not been established for diagnosing, curing, mitigating, treating
or preventing any disease.
Lisinopril, the most commonly used drug
by U.S. Medicare patients, was approved as an Oral Solution for the first time.
Diabetes
treatments, one of the most commonly prescribed drug categories in the U.S., saw
the addition of Sanofi and Novo Nordisk’s fixed-dose,
long-acting insulin and glucagon-like peptide 1
(GLP-1) agonist combinations to the list of options available to treat adult
type-2 diabetes.
Our View
The approval
of new drugs ensures that the world has access to improved healthcare solutions
and breakthrough medical therapies.
With Donald Trump “focused on accelerating the FDA” and an on-going argument that drugs should not have to be proven effective before getting approved, this year looks like the one where the new drug
approval scenario will be shaken up for time to come.
Click here to access our list of all New Drug Approvals (Excel version available) for FREE!
Impressions: 14459