The Covid-19 pandemic expedited the
growth of contract development and manufacturing organizations (CDMOs) as
pharma companies increasingly turned to them to accelerate drug development.
However, by the end of 2022, a decline in demand for
Covid vaccines hit some CDMOs, and they saw their supply deals come to an
abrupt end.The year 2022 also saw the steepest drop
in new drug approvals by the US Food and Drug Administration (FDA) since 2016.
Although the Center for Drug Evaluation and Research (CDER) only approved 37 new drugs, a 26 percent decline from the 50 drugs approved in 2021, there was a rise in new biologic drug approvals. Of these, 41 percent were biologics. Eight biologics were also approved by FDA’s Center for Biologics Evaluation and Research
(CBER) in 2022.Despite other challenges like high inflation, the ongoing Russia-Ukraine war and increasing economic uncertainties, CDMOs continued to grow through 2022 and the first quarter of 2023, particularly in biologics, cell and gene (C&G) therapies and highly potent active pharmaceutical ingredients (HPAPIs).CDMOs have been swift in altering their
production lines to meet the demands of smaller, more diverse projects. This
flexibility is likely to contribute to future growth. According
to Research and Markets, the global
CDMO market was valued at US$ 172.7 billion in 2022, and is projected to reach US$ 246.6 billion by 2026, growing
at a CAGR of 9.3 percent.View CDMO Activity Tracker of Q1 2023 (Free Excel Available)Large
CDMOs post double-digit growth; Sartorius buys Polyplus for US$ 2.6 billionSeveral big CDMOs such as the Lonza Group, Samsung Biologics and Siegfried AG reported impressive revenue
growth in 2022. Switzerland-based Lonza, the biggest CDMO player, saw its
revenue grow 15 percent to reach CHF 6.2 billion (US$ 6.7 billion) in 2022.Samsung Biologics’ CDMO revenue skyrocketed 49 percent in 2022, as compared to 2021, touching KRW 2.34 trillion (US$ 1.85 billion). Siegfried generated CHF 1,229 million (US$ 1.33 billion) in sales, a growth of 15.6
percent. And EUROAPI’s CDMO business grew 18.3 percent to reach € 267.5 million (US$ 0.28 billion). Meanwhile, Pfizer CentreOne generated US$ 1.34 billion in sales in 2022. As demand for Covid jabs dropped, Catalent announced structural changes and
cost-reduction measures, including more than 210 layoffs at
its Texas and Maryland facilities. It also laid off up to 400 employees at its Bloomington, Indiana site. Overall, Catalent’s revenues grew by 1 percent — at US$ 2.17 billion — in the last six months of 2022. Similarly, UK-based CDMO Abzena also laid off 66 employees in
San Diego.So far, the biggest CDMO deal of 2023 has
been the acquisition of France-based Polyplus, a provider of innovative upstream technologies for advanced biologic and C&G therapy production, by Germany-based CDMO Sartorius Stedim Biotech for €2.4 billion (US$ 2.6 billion). The deal gives Sartorius the know-how needed in the production of viral vectors for building C&G therapies.View CDMO Activity Tracker of Q1 2023 (Free Excel Available) Catalent
expands into cell therapies, Charles River Labs signs deal with RznomicsThe development of C&G therapies has emerged as a high growth
area for CDMOs. In 2022, CSL’s hemophilia B treatment Hemgenix emerged as the world’s most expensive medicine,
beating Bluebird bio’s C&G therapies — Skysona and Zynteglo. This year, around 13 C&G therapies are expected to receive approval in the US and Europe, offering big opportunities to CDMOs worldwide. The US$ 3.2 billion CDMO market for C&G therapies is estimated to be growing at 18.1 percent CAGR, and is poised to reach US$ 18.6 billion by 2032.In January, Catalent opened a new plasmid
DNA manufacturing facility in Belgium for cell therapies and expanded its partnership
with Sarepta for manufacturing its gene therapy candidate — SRP-9001. If approved, this could be the first gene therapy for Duchenne muscular dystrophy
(DMD). Catalent has also entered into a licensing agreement with Bhami Research Laboratory to access the latter’s formulation technology to help enable the subcutaneous delivery of high-concentration biologic therapies.Charles River Laboratories has signed a deal with
South Korean biopharma Rznomics to develop and manufacture viral
vectors for a gene therapy to treat liver cancer. It also inked another deal
with Purespring Therapeutics for plasmid DNAs and acquired SAMDI Tech for US$ 50 million. In recent years, the company has expanded its C&G portfolio through the acquisitions of Cobra Biologics, Vigene Biosciences and Cognate BioServices.View CDMO Activity Tracker of Q1 2023 (Free Excel Available) Samsung
Biologics, LOTTE, Lonza expand facilities for ADCsThe other emerging growth area for CDMOs
is biologics. The global CDMO market for biologics was valued at US$ 11.27 billion in 2021 and is expected to grow at 11.51 percent CAGR to reach US$ 21.90 billion by 2027. Technological advancements and approval of new biologic drugs is contributing to this growth.Biologics are triggering deals. After Provention Bio received FDA approval for its
type 1 diabetes drug Tzield last year, the US-based company
signed a deal with AGC Biologics in January this year to produce
the drug at its Seattle biologics plant. Last month, Sanofi announced it is acquiring
Provention Bio for US$ 2.9 billion.Several CDMOs are also vying for a share
of the fast growing antibody-drug conjugates (ADC) market, which is projected
to reach US$ 13.1 billion by
2030, up from US$ 3.51 billion in 2020. Samsung Biologics’ CEO, John Rim, announced in
January that ADCs would be a major focus area for the company. It is investing US$ 1.46 billion to construct its plant 5 for
biopharmaceuticals and plans to start operations in 2025. It has also entered
into a manufacturing deal with Pfizer for US$ 183 million.Newly-established CDMO LOTTE Biologics is also expanding its
facility for
ADCs in order to get a foothold in North America. LOTTE Biologics plans to invest US$ 3 billion
over the next seven years to build three mega plants. It has also acquired BMS’ manufacturing facility in New
York for US$ 160 million. Similarly, Lonza completed the expansion of
its facility for ADCs in Visp, Switzerland.View CDMO Activity Tracker of Q1 2023 (Free Excel Available)Growing
demand for HPAPIs, controlled substances draw investments in CDMOsThe global market for HPAPIs is growing
at 8.5 percent CAGR and is expected to reach US$ 34 billion in
2026. Several companies have announced expansion plans to meet this rising demand.Piramal Pharma Solutions has started production of APIs and HPAPIs at its
facility in Michigan, US. German CDMO ChemCon provides services for APIs,
HPAPIs and controlled substances, including amphetamines, cannabinoids, fentanyl derivatives and steroids. Additionally, it offers high-quality fine chemicals, isotopically labeled compounds, organic, inorganic and polymer chemistry. Its products are used for important specialty applications such as oncology, orphan diseases, critical care, dermatology, etc. Veranova’s subsidiary Macfarlan Smith has
completed a US$ 10 million expansion
of its mid-scale API manufacturing capabilities in Edinburgh, UK. Siegfried has opened a development center for
drug products in Barcelona, Spain, with dedicated facilities for HPAPI
products. Last November, Polpharma announced investment at its HPAPI facility in Poland. It expects to open a new R&D and production facility in the first quarter of 2024. Meanwhile, Eurofins CDMO Alphora Inc has also expanded its API development laboratories, including those for HPAPI development.CDMOs are also expanding their services
for controlled substances, particularly in therapeutic areas such as
depression, PTSD, addiction and pain management. Due to stringent regulations
on the development and manufacture of controlled substances, pharma companies
are turning to CDMOs that are experienced and licensed to handle them. These
include Cambrex Corporation, Curia, Patheon, Pfizer CentreOne, Veranova, ChemCon, Evonik, Minakem and Seqens.View CDMO Activity Tracker of Q1 2023 (Free Excel Available)Our
viewOver the last few years, we have noticed
how CDMOs have altered their business models with changes in the business
environment. Of late, CDMOs have benefitted from the trend of smaller biotechs outsourcing manufacturing of novel modalities, such as C&G and RNA therapies. Of the 37 drugs approved by the FDA’s CDER last year, 24 (or 65 percent)
were developed by mid or small-sized biopharma companies.We also know how the approval of new drugs benefits
the CDMO industry. For instance, FDA approved Rigel Pharma’s Rezlidhia (olutasidenib) in December as a treatment for relapsed and refractory acute myeloid leukemia. The drug’s dose manufacturing has been outsourced to Catalent. Therefore, a rise in the approval of novel drugs in 2023 should benefit the industry. And rapid advancements in biotechnology will ensure healthy growth of CDMOs.
Impressions: 2796
Acquisitions and spin-offs dominated headlines in 2019 and the tone was set very early with Bristol-Myers Squibb acquiring
New Jersey-based cancer drug company Celgene in a US$ 74 billion deal announced on
January 3, 2019. After factoring
in debt, the deal value ballooned to about US$ 95 billion, which according
to data compiled by Refinitiv, made it the largest healthcare deal on
record.
In the summer, AbbVie Inc,
which sells the world’s best-selling drug Humira, announced its acquisition of Allergan Plc, known for Botox and other cosmetic
treatments, for US$ 63 billion. While the companies are still awaiting
regulatory approval for their deal, with US$ 49 billion in combined 2019
revenues, the merged entity would rank amongst the biggest in the industry.
View Our Interactive Dashboard on Top drugs by sales in 2019 (Free Excel Available)
The big five by pharmaceutical sales — Pfizer,
Roche, J&J, Novartis and Merck
Pfizer
continued
to lead companies by pharmaceutical sales by reporting annual 2019 revenues of
US$ 51.8 billion, a decrease of US$ 1.9 billion, or 4 percent, compared to
2018. The decline was primarily attributed to the loss of exclusivity of Lyrica in 2019,
which witnessed its sales drop from US$ 5 billion in 2018 to US$ 3.3 billion in
2019.
In 2018, Pfizer’s then incoming CEO Albert Bourla had mentioned that the company did not see the need for any large-scale M&A activity as Pfizer had “the best pipeline” in its history, which needed the company to focus on deploying its capital to keep its pipeline flowing and execute on its drug launches.
Bourla stayed true to his word and barring the acquisition of Array Biopharma for US$ 11.4 billion and a spin-off to merge Upjohn, Pfizer’s off-patent branded and generic established medicines business with
Mylan, there weren’t any other big ticket deals which were announced.
The
Upjohn-Mylan merged entity will be called Viatris and is expected to have 2020
revenues between US$ 19 and US$ 20 billion
and could outpace Teva to
become the largest generic company in the world, in term of revenues.
Novartis, which had
followed Pfizer with the second largest revenues in the pharmaceutical industry
in 2018, reported its first full year earnings after spinning off its Alcon eye
care devices business division that
had US$ 7.15 billion in 2018 sales.
In 2019,
Novartis slipped two spots in the ranking after reporting total sales of US$
47.4 billion and its CEO Vas Narasimhan continued his deal-making spree by buying New
Jersey-headquartered The Medicines Company (MedCo) for US$ 9.7
billion to acquire a late-stage cholesterol-lowering
therapy named inclisiran.
As Takeda Pharmaceutical Co was
busy in 2019 on working to reduce its debt burden incurred due to its US$ 62
billion purchase of Shire Plc, which was announced in 2018, Novartis also purchased
the eye-disease medicine, Xiidra, from the Japanese drugmaker for US$ 5.3 billion.
Novartis’ management also spent a considerable part of 2019 dealing with data-integrity concerns which emerged from its 2018 buyout of AveXis, the
gene-therapy maker Novartis had acquired for US$ 8.7 billion.
The deal gave Novartis rights to Zolgensma,
a novel treatment intended for children less than two years of age with the
most severe form of spinal muscular atrophy (SMA). Priced at US$ 2.1 million,
Zolgensma is currently the world’s most expensive drug.
However,
in a shocking announcement, a month after approving the drug, the US Food and
Drug Administration (FDA) issued a press release on
data accuracy issues as the agency was informed by AveXis that
its personnel had manipulated data which
the FDA used to evaluate product comparability and nonclinical (animal)
pharmacology as part of the biologics license application (BLA), which was
submitted and reviewed by the FDA.
With US$
50.0 billion (CHF 48.5 billion) in annual pharmaceutical sales, Swiss drugmaker
Roche came in at number two position in 2019
as its sales grew 11 percent driven by
its multiple sclerosis medicine Ocrevus, haemophilia drug Hemlibra and cancer medicines Tecentriq and Perjeta.
Roche’s newly introduced medicines generated US$ 5.53 billion (CHF 5.4 billion) in growth, helping offset the impact of the competition from biosimilars for its three best-selling drugs MabThera/Rituxan, Herceptin and Avastin.
In late 2019, after months of increased
antitrust scrutiny, Roche completed
its US$ 5.1 billion acquisition of Spark Therapeutics to strengthen its presence in
gene therapy.
Last year, J&J reported almost flat worldwide sales of US$ 82.1 billion. J&J’s pharmaceutical division generated US$ 42.20 billion and its medical devices and consumer health divisions brought in US$ 25.96 billion and US$ 13.89 billion respectively.
Since J&J’s consumer health division sells analgesics, digestive health along with beauty and oral care products, the US$ 5.43 billion in consumer health sales from over-the-counter drugs and women’s health products was only used in our assessment of J&J’s total pharmaceutical revenues. With combined pharmaceutical sales of US$ 47.63 billion, J&J made it to number three on our list.
While the sales of products like Stelara, Darzalex, Imbruvica, Invega Sustenna drove J&J’s pharmaceutical business to grow by 4 percent over 2018, the firm had to contend with generic competition against key revenue contributors Remicade and Zytiga.
US-headquartered Merck, which is known as
MSD (short for Merck Sharp & Dohme) outside the United States and
Canada, is set to significantly move up the rankings next year fueled by its
cancer drug Keytruda, which witnessed a 55
percent increase in sales to US$ 11.1 billion.
Merck reported total revenues of US$ 41.75 billion and also
announced it will spin off its women’s health drugs,
biosimilar drugs and older products to create a new pharmaceutical
company with US$ 6.5 billion in annual revenues.
The firm had anticipated 2020 sales between US$ 48.8 billion and US$ 50.3 billion however this week it announced that the coronavirus pandemic will reduce 2020 sales by more than $2 billion.
View Our Interactive Dashboard on Top drugs by sales in 2019 (Free Excel Available)
Humira holds on to remain world’s best-selling drug
AbbVie’s acquisition of Allergan comes as the firm faces the expiration of patent protection for Humira, which brought in a staggering US$ 19.2 billion in sales last year for
the company. AbbVie has failed to successfully acquire or develop a major new
product to replace the sales generated by its flagship drug.
In 2019, Humira’s US revenues increased 8.6 percent to US$ 14.86 billion while internationally, due
to biosimilar competition, the sales dropped 31.1 percent to US$ 4.30 billion.
Bristol Myers Squibb’s Eliquis, which is also marketed by Pfizer, maintained its number two position
and posted total sales of US$ 12.1 billion, a 23 percent increase over 2018.
While Bristol Myers Squibb’s immunotherapy treatment Opdivo, sold in partnership with Ono in Japan, saw sales increase from US$ 7.57 billion to US$ 8.0 billion, the growth paled in comparison to the US$ 3.9
billion revenue increase of Opdivo’s key immunotherapy competitor Merck’s Keytruda.
Keytruda took the number three spot in drug sales that
previously belonged to Celgene’s Revlimid, which witnessed a sales decline from US$ 9.69 billion to US$ 9.4 billion.
Cancer treatment Imbruvica, which is marketed
by J&J and AbbVie, witnessed a 30 percent increase in sales. With US$ 8.1
billion in 2019 revenues, it took the number five position.
View Our Interactive Dashboard on Top drugs by sales in 2019 (Free Excel Available)
Vaccines – Covid-19 turns competitors into partners
This year has been dominated by the single biggest health emergency in years — the novel coronavirus (Covid-19) pandemic. As drugs continue to fail to meet expectations, vaccine development has received a lot of attention.
GSK reported the highest vaccine sales of all drugmakers with
total sales of US$ 8.4 billion (GBP 7.16 billion), a significant portion of its
total sales of US$ 41.8 billion (GBP 33.754 billion).
US-based Merck’s vaccine division also reported a significant increase in sales to US$ 8.0 billion and in 2019 received FDA and EU approval to market its Ebola vaccine Ervebo.
This is the first FDA-authorized vaccine against the deadly virus which causes
hemorrhagic fever and spreads from person to person through direct contact with
body fluids.
Pfizer and Sanofi also reported an increase in their vaccine sales to US$ 6.4
billion and US$ 6.2 billion respectively and the Covid-19 pandemic has recently
pushed drugmakers to move faster than ever before and has also converted
competitors into partners.
In a rare move, drug behemoths — Sanofi and GlaxoSmithKline (GSK) —joined hands to develop a vaccine for the novel coronavirus.
The two companies plan to start human trials
in the second half of this year, and if things go right, they will file
for potential approvals by the second half of 2021.
View Our Interactive Dashboard on Top drugs by sales in 2019 (Free Excel Available)
Our view
Covid-19 has brought the world economy to a grinding halt and shifted the global attention to the pharmaceutical industry’s capability to deliver solutions to address this pandemic.
Our compilation shows that vaccines and drugs
for infectious diseases currently form a tiny fraction of the total sales of
pharmaceutical companies and few drugs against infectious diseases rank high on
the sales list.
This could well explain the limited range of
options currently available to fight Covid-19. With the pandemic currently infecting
over 3 million people spread across more than 200 countries, we can safely
conclude that the scenario in 2020 will change substantially. And so should our
compilation of top drugs for the year.
View Our Interactive Dashboard on Top drugs by sales in 2019 (Free Excel Available)
Impressions: 54752
This week in Phispers, we look at how Mylan’s president Rajiv Malik and Emcure’s CEO Satish Mehta, along with 12 major generic drug players, got added to an expanded investigation into the drug price-fixing case in the US. While Trump declared the opioid crisis a health emergency, pharma companies Celgene and Gilead saw their third quarter results and guidance get impacted by a drug trial failure and competition. Meanwhile, there was news on Novartis buying AAA, CVS Health showing interest in buying health insurer Aetna and Merck cutting jobs and withdrawing its application for Keytruda as treatment for advanced lung cancer.
Mylan’s president, Emcure’s CEO, 12 firms to be probed in price-fixing case
This week, attorneys general from 45 US states named Mylan’s president Rajiv Malik and Emcure’s billionaire CEO Satish Mehta as they expanded their three-year old
investigation into price-fixing in the generic pharmaceuticals industry.
In addition to the two individuals, 12 major generic drug companies and 13 drugs were added to the expanded investigation. This week’s filing alleges collusion of 18 defendant companies, as well as two individual executives, involving a total of 15 drugs.
(To view the complete list of companies and
drugs, click here)
The states allege multiple conspiracies that restrained trade, artificially inflated and/or maintained prices and reduced competition in the generic drug industry. According to the expanded complaint, Mehta and Malik were “directly involved in conceiving an illegal agreement and taking affirmative steps to ensure it was executed by their subordinates.”
Earlier this year, a federal criminal investigation resulted
in the convictions of former Heritage Pharmaceuticals CEO Jeffrey Glazer and former Heritage President Jason
Malek. Heritage is a wholly-owned subsidiary of Emcure. Both Glazer and Malek pleaded guilty and have been cooperating in the anti-trust investigations being conducted by
federal and state prosecutors.
PharmaCompass had covered the illegal practices at
Heritage Pharmaceuticals that had come to light when Emcure had filed a lawsuit
against Glazer. It now remains to be seen if Glazer acted on his own or
colluded with his bosses in India to fix generic drug prices in the United
States.
As Trump declares opioid crisis an emergency, Insys founder
gets arrested
Last week, the US President Donald Trump hardened his stand on the
opioid crisis, blaming it on factors like criminals, drug companies, Mexico and
China. In the US, the crisis claimed over 30,000 lives in 2015.
He termed abuse of opioids a public health emergency and outlined steps to fight it — such as new efforts to curb prescription abuse, reduction in import of certain drugs and educating children on the risks.
“We must stop the flow of all types of illegal drugs into our communities,” Trump said. Blaming pharmaceutical companies for over-prescribing pain medications, he said the US Food and Drug Administration (FDA) will provide more training to doctors who prescribe drugs to prevent abuse. Moreover, he has requested that one opioid drug, Opana ER, no longer be sold.
According to Trump, a border wall with Mexico will “greatly help” reduce this problem. He also talked about China’s role in the production of fentanyl.
Meanwhile, John Kapoor, the founder of Insys Therapeutics, was arrested last week on
charges of participating in a scheme to bribe doctors to prescribe a
fentanyl-based cancer pain drug. Kapoor has resigned from the company’s board of directors, and his arrest is being seen as a step towards fighting the opioid epidemic.
The charges against Kapoor marked a major
escalation of probes into Subsys, an under-the-tongue spray that contains
fentanyl, an addictive synthetic opioid.
Celgene’s Crohn’s disease drug fails trial; Gilead reports weak Hep C sales
Two companies have not had a good going — Celgene and Gilead. The former announced that its drug mongersen — a potential treatment for Crohn’s disease — had failed. Celgene had purchased the drug four years ago for US$ 710 million from a private company in Dublin — Nogra Pharma Limited. The New Jersey-headquartered biotech firm had promised that
if mongersen gets approved, Nogra would get another US$ 815 million. But that
was not to be.
What was worse, Celgene’s third-quarter financial announced last week spooked its investors. The
company missed analysts’ sales projections for the third quarter by 4 percent, cut its guidance for 2020 by 5 to 10 percent, and announced that sales of a key (plaque psoriasis) drug — Otezla — had missed forecasts by US$ 100 million.
In a note, Geoffrey Porges, a biotech analyst at Leerink, said: “Investors are likely to ask whether the company’s good fortune has run out, with disappointments (mongersen) and negative revisions (Otezla) left and right.”
Gilead, on the other hand, is trying hard to move away from hepatitis C. Its hepatitis C drugs generated US$ 2 billion during the last quarter — just US$ 300 million short of Wall Street’s projections. However,
investors were cautioned about AbbVie’s new launch —Mavyret — which is expected to have a “big impact” on Gilead’s fourth-quarter results.
Gilead’s hepatitis C drugs have been facing competition from new rivals who have eaten into its market share and driven down prices over the last few quarters.
However, we could expect some M&A action soon. “Gilead noted that it is in a ‘constant state of valuation and opportunities’ and that it is very, very active,” said an analyst.
Novartis buys radiopharmaceutical firm AAA for
US$ 3.9 billion
In July, PharmaCompass had covered news on how Lutathera — a nuclear medicine targeted at the type of cancer that killed former Apple Inc co-founder and CEO Steve Jobs — got a nod from the European Medicines Agency (EMA), boosting prospects for its developer Advanced Accelerator Applications (AAA).
Well, last week the French biotech — AAA — got bought over by Novartis for
US$ 3.9 billion. Novartis is paying a 44 percent premium over AAA’s market valuation from September 28.
The deal further strengthens Novartis’ oncology business, which had got a shot in the arm in 2015, when it
acquired GlaxoSmithKline’s marketed cancer drugs. And in August, its gene-modifying leukemia treatment (a chimeric antigen receptor T-cell therapy or CAR-T therapy) — Kymriah — got approved. This was the first CAR-T therapy to bag an FDA approval.
With AAA, Novartis will now have a
technology that deploys trace amounts of radioactive compounds to not only
diagnose a disease (by creating images of organs and lesions), but also to
fight cancer.
Lutathera is a radiopharmaceutical — it uses radioisotopes to target neuroendocrine tumors. Along with Lutathera, Novartis also gets a pipeline of drugs from AAA with some near-term potential.
US Merck to cut 1,800 jobs, withdraws EU
application of cancer wonder drug
Late last week, Merck announced it had withdrawn its application for
Keytruda (pembrolizumab) as a combination treatment for an advanced stage of
non-small cell lung cancer (NSCLC) from the European Medicines Agency
(EMA).
An analyst with Evercore ISI, Umer Raffat, said
in a note that EMA was reluctant to approve drugs based on Phase II data, even
though the FDA regularly does so in oncology.
The company is also moving to a new sales team structure in the
US, and plans to cut 1,800 sales positions, while adding 960 jobs to a new
chronic care salesforce.
According to a Fox Business report, three of Merck’s US sales teams will be cut: primary care, disease-focused endocrinology and hospital chronic care. This is “to better support changes in our business in the United States,” Merck’s spokeswoman said.
Merck’s new chronic care team will focus on Januvia (a diabetes drug), other primary care products such as sleep medication Belsomra, and products for respiratory conditions and women's health, she added.
Releasing its third quarter numbers in the
US, Merck was also forced to concede that the NotPetya cyber attack had cost the company US$ 135 million in lost sales
along with US$ 175 million in related costs. That extra US$ 310 million in
costs will be repeated in Q4 as overall damages inch up to the US$ 1 billion
mark, the company said.
As Amazon threat looms over US pharmacy
biz, CVS eyes health insurer Aetna
Amazon has been trying to get into the US$ 412 billion pharmacy
business in the US. Every year, pharmacies in the US dispense about 4.5 billion prescriptions. Patients pick up about 9 out of 10 prescriptions at a retail pharmacy. Amazon’s entry into online
prescription drug sales, therefore, poses an existential threat to brick-and-mortar
pharmacies.
And that’s the reason why news about US pharmacy operator CVS Health Corp reportedly evincing interest in buying health insurer Aetna Inc is important. This potential acquisition could trigger another round of deal making in an industry that is fearing Amazon’s arrival.
According to news reports, CVS Health is expected to buy Aetna for US$ 66 billion
in what would be the biggest deal of the year.
If the deal goes through, it would also be
the biggest of its kind ever in healthcare. “A potential combination would diversify CVS profit streams ahead of an Amazon entry and set the stage for a new healthcare-retail delivery model,” Morgan Stanley analysts wrote
in a note.
A deal would make CVS-Aetna a one-stop shop for customers’ healthcare needs — which could range from employer healthcare and government plans to managing benefits and running drug stores.
Impressions: 3102
This
week, Phispers brings you news about Teva, which finally found a CEO in Kare
Schultz. Allergan transferred the patent of its best-selling eye drug Restasis
to the Mohawk tribe in order to protect it from patent dispute. There was more
news on EpiPen, as Pfizer received an FDA warning
letter for Mylan’s flagship product. And, a leaked audio revealed how Insys lied to a patient who didn’t have cancer to sell a prescription opioid.
Teva nabs Lundbeck’s CEO to revive its fortunes; to pay over US$ 44 million
After
months of bad news, this week there was finally some good news from the
much-troubled Teva. The Israeli generic
drug giant finally managed to get a new chief executive officer for itself, after Erez Vigodman’s resignation in February this year.
Teva
poached Lundbeck’s Kare Schultz as its new CEO. Schultz, 56, will move to Israel and be based at Teva’s headquarters in Petach Tikva. In an SEC filing, Teva said Schultz is starting with a base pay of US$ 2 million. Along
with several bonuses (including a signing bonus of US$ 20 million), his initial
package adds up to over US $44 million. Schultz has 30 years of
experience in global pharma business.
After
the acquisition of Allergan’s generic business — Actavis — last year, Teva has been saddled with huge debts. And the key question before the new CEO is whether he will maintain Teva as both a generics and specialty drugmaker, or split it in two or get out of the highly competitive generics business.
During
the last quarter (April to June), Teva posted revenues of US$ 5.7 billion, out
of which US$ 3.1 billion came from generics and US$ 2.1 billion from its own
branded drugs. About half the sales of its own medicines come from a
blockbuster multiple sclerosis drug which is now facing competition from generics.
Pfizer
gets FDA warning letter for supplying faulty EpiPens to Mylan
The
US Food and Drug Administration (FDA) has told Meridian
Medical Technologies,
a division of Pfizer
that makes the EpiPen injector device (sold
by Mylan NV), that serious component and product failures have been associated with patient deaths.
In
a warning letter issued on September 5, the FDA said the Pfizer unit failed to
adequately investigate problems at its manufacturing facility in Brentwood,
Missouri. It also did not take appropriate corrective actions before an FDA
inspection earlier this year.
Throughout last year,
Meridian received hundreds of complaints that the EpiPen device, which is used
to combat serious allergic reactions, failed to operate during life-threatening
emergencies.
In
March this year, tens of thousands of EpiPens were recalled worldwide after two
reports of the shot failing to work in emergencies.
In
a note to Stat News, Pfizer said: “Between 2015 and now, we have shipped more than 30 million EpiPen Auto-Injectors globally. It’s not unusual to receive product complaints, especially when the product is frequently administered by non-medically trained individuals.”
“We currently have no information to indicate that there was any causal connection between these product complaints and any patient deaths,” it added.
In
a statement, Mylan said it is confident of the safety
and efficacy of EpiPen products being produced at the Brentwood site.
According to the FDA warning letter, in April 2016, a customer complained that an EpiPen failed to activate. Though Meridian confirmed the problem, in June 2016 it decided the defect was infrequent. Moreover, between 2014 and 2017, Meridian received 171 devices from patients who complained their devices failed to activate even when they followed the instructions. But Meridian only examined some of the devices and told the FDA inspectors they could not examine the rest without “approval by management”.
The warning letter highlights that Meridian “failed to thoroughly investigate multiple serious component and product failures for your EpiPen products, including failures associated with patient deaths and severe illness. You also failed to expand the scope of your investigations into these serious and life-threatening failures or take appropriate corrective actions, until FDA's inspection.”
Allergan tries to protect eye drug by selling its patent
rights to Mohawk tribe
Allergan
played an unusual gambit last week to protect its best-selling eye drug — Restasis — from a patent dispute. It did that by transferring the patent of the drug to the Saint Regis Mohawk Tribe, a Mohawk Indian reservation in Franklin County, New York, United States.
Under
the deal, Allergan will pay the tribe US$ 13.75 million. In exchange, the tribe
will claim sovereign immunity as grounds to dismiss a patent challenge through
a unit of the United States Patent and Trademark Office. The tribe will lease
the patents back to Allergan, and will receive US$ 15 million in annual royalties
as long as the patents remain valid.
For
the Mohawk tribe, the deal could generate a new revenue stream. Bob Bailey, Allergan’s chief legal officer told FiercePharma: “I have spent time with the chiefs and their primary motivation for doing this… is to diversify the stream of incomes that this tribe enjoys away from casinos and cigarettes, into longer term, more viable streams…”
If
successful, the move will represent a new way for drug companies to circumvent
the inter partes review, a procedure for challenging the validity of a
United States patent before the United States Patent and Trademark Office.
The
move sent ripples across the pharmaceutical industry. Surely, if this strategy
pays off, there will be other drug companies following suit. Many observers
ridiculed the company on social media by calling the deal unscrupulous and sure
to bring more criticism to the industry. The tribe has reportedly also
taken ownership of patents owned by a technology company.
Interestingly,
last year, Allergan CEO Brent Saunders had won accolades for
condemning predatory drug pricing by the likes of Martin Shkreli and emphasized the company’s commitment to research and development.
This week, in a Federal District Court in
Marshall, Texas, Mylan challenged Allergan’s move. Netherland headquartered Mylan, in its court filing stated that Ireland headquartered Allergan is “attempting to misuse Native American sovereignty to shield invalid patents from cancellation.”
Biocon’s Malaysia facility gets EU nod; Dr Reddy’s gets six major observations
Biocon, India’s leading biopharmaceutical company, had some good news to share, after its recent setbacks related to its biosimilar program. Its insulin facility in Malaysia — Biocon Sdn Bhd — received an EU GMP compliance certificate
from the
HPRA (Ireland), the representative European inspection authority.
Biocon Malaysia is “one of Asia’s largest state-of-the-art integrated insulins manufacturing facilities, set up with an investment of about US$ 275 million, at the BioXcell Biotech Park in Johor, Malaysia,” Biocon said in a statement.
Dr Reddy’s Vizag plant:
Indian pharma major Dr Reddy’s said its plant in
Vishakapatnam (India) has been issued
six major observations related to good manufacturing practices (GMP) by the regulatory authority of Germany — Regierung von Oberbayern.
In a statement issued to the Bombay Stock Exchange last week, the company said: “This is to inform you that on 7th September, 2017, the Regulatory Authority of Germany, concluded an audit for our formulations manufacturing facility in Duvvada, Vishakapatnam, with zero critical and six major observations.” Products manufactured at this facility are not currently exported to the EU.
The
company will be submitting a Corrective and Preventive Action plan (CAPA) to
the authorities, the company added. The Duvvada facility’s compliance with the CAPA and other applicable regulations will be reviewed again by the regulator by November 2018 for continuation of its
EU-GMP certification, the
statement added.
Meanwhile,
Cadila Healthcare received zero observations from the USFDA for its Moraiya plant, situated in Gujarat
(India). The FDA has issued zero observations for the unit after its
re-inspection this year.
Opioid crisis: Leaked audio reveals how Insys lied to a patient who didn’t have cancer
In
the United States, an explosive audio revealed how opioids were being given by a drug company to a patient who didn’t have cancer. The audio was released by Senator Claire McCaskill’s office last week. It explains how many in America had been victims of the opioid crisis.
According
to Business Insider, the audio is a phone call between an employee from Insys, the Arizona-based maker of Subsys (an opioid pain medication given to cancer patients), and a pharmacy benefit manager (PBM).
In
the US, PBMs are supposed to ensure that everyone who gets Subsys has cancer.
But the patient in question, Sarah Fuller, didn't have cancer.
An
Insys employee tried to convince Fuller she had cancer, and succeeded. Fuller
eventually died of a Subsys overdose on March 25, 2016.
The audio accompanies a report from McCaskill’s office on the marketing and business practices of Insys. Since December 2016, several Insys executives, including a former CEO, have been arrested over the opioid issue.
German Merck hires JP Morgan to divest its consumer health
business
Germany’s Merck KGaA has hired JP Morgan to sell its consumer health business, which includes brands such as Seven Seas vitamins, the company said last week. The annual sales of Merck’s over-the-counter medicines and vitamin supplements are around US$ 1 billion.
The
divestment would help fund research into higher-margin prescription drugs,
Merck said. The business could be worth around US$ 4.5 billion.
According to news reports, Merck has already sounded out prospective buyers including Swiss food giant Nestle. Merck had held preliminary talks with Nestle in summer this year. Nestle reportedly favored a joint venture deal and no agreement could be reached.
According
to Euromonitor International, the global market for consumer health products is
worth around US$ 233 billion. Merck ranks 32nd in this sector, with a 0.4
percent share.
J&J
dumps hepatitis C drug development; Roche suffers R&D setback
Janssen Sciences Ireland UC, a unit of Johnson & Johnson, said it would discontinue further development of its hepatitis C
research, citing increased availability of a number of effective hepatitis C
therapies.
Its investigational hepatitis C treatment regimen — JNJ-4178 — is a combination of three direct acting antivirals. “The ongoing phase II studies with JNJ-4178 will be completed as planned, with no additional development thereafter,” Janssen said in a statement.
Roche
drug fails endpoints:
Roche said its drug for geographic atrophy — lampalizumab — failed in first of two late-stage studies.
Lampalizumab was once considered a potential mega-blockbuster that could bring
in US$ 6 billion a year.
Lampalizumab did not reduce mean change in geographic atrophy lesion area compared to a placebo at one year (48 weeks). Given the lack of efficacy, “further dosing in patients will be interrupted until the results from the second Phase III study are evaluated,” Roche said.
Impressions: 3144
In Phispers this week, we bring you news on two new bills in the US that seek to bring in cheaper medicines from Canada, and allow FDA to recall unsafe drugs. We also bring you the real reason why Sanofi, even after offering US$ 30 billion, could not bag Actelion. And, there is news on gene-editing technology CRISPR and Mylan and Biocon’s application for Pegfilgrastim, a proposed biosimilar. Read on.
US introduces bill to allow FDA to recall
unsafe drugs
In
the United States, Republican Rosa DeLauro introduced a new bill
last week that will allow the US Food and Drug Administration (FDA) to do what
many assume it already can require pharmaceutical or over-the-counter drug
(OTC) companies to stop marketing unsafe products. The law will also cover
homeopathic products.
The bill, once passed, will be known as the
Recall Unsafe Drugs Act. Once enacted, this law will give FDA mandatory recall
authority over drugs and homeopathic products.
“Most people would be shocked to learn that while the FDA has the authority to order a recall of medical devices and biologic products, such as vaccines and blood products, the agency cannot order recalls of prescription and over-the-counter medications,” DeLauro said.
This loophole is problematic, as some companies can refuse FDA’s calls to remove their pharmaceutical products from the market because they are unsafe.
New bill in US pushes for drug imports from
Canada, while industry resists it
The Trump administration appears committed to providing cheaper medicines. A new bill the Safe and Affordable Drugs from Canada Act would require the FDA to set up a “personal importation program” to allow individuals to import 90-day supplies of their medicines from Canadian pharmacies. The bill has been drafted by Senators Amy Klobuchar, Chuck Grassley and John McCain.
However,
the pharmaceutical industry is pushing back.
In a statement to FiercePharma, the Pharmaceutical Research and
Manufacturers of America (PhRMA) pointed to a Drug
Enforcement Administration report last year that mentioned to how
counterfeit fentanyl from China made
its way to the US through Canada and Mexico, worsening the current opioid
crisis.
Imports “would exacerbate these threats,” a PhRMA spokesperson said.
Imported drugs wouldn’t “be subject to the US FDA’s robust safety requirements, and there would be no way to trace the country of origin for the imported products. Even Canada has said it does not and would not be able to guarantee that US citizens would receive products that are safe, effective and of high quality,” the spokesperson added. “Guaranteeing patient safety is crucial, and we must have policies that ensure patients safely have access to the medicines they need.”
A
fortnight back, PharmaCompass carried a news on Marathon Pharmaceuticals, which received FDA approval for Emflaza (deflazacort) to treat rare
genetic disorder Duchenne muscular dystrophy (DMD). The drug carries a list price of US$ 89,000 for a year’s supply, when patients in the US are importing it from Canada and the UK for around US$ 1,000 to US$ 2,000.
Prospectus highlights Sanofi’s struggles in J&J’s US$ 30 billion acquisition of Actelion
Sanofi made headlines recently, not for the acquisitions it made, but for the one it wasn’t able to close. Late last year, the French pharma giant was widely identified as the big player that managed to push Johnson & Johnson away from negotiations with Actelion, which had started back in January 2016.
The Actelion-J&J deal prospectus offers insights into why Sanofi (identified as Company A) failed in its US$ 30 billion acquisition even though it would have delivered “approximately equivalent value to Actelion’s shareholders”.
According to the deal prospectus, even though Sanofi had bid a higher indicative price, the board of directors of Actelion perceived ‘significant uncertainty’ posed by a potential transaction with Sanofi.
The uncertainty was created when, “Sanofi indicated that it would only be willing to proceed with a transaction on the basis of a price lower than its previously communicated offer price and on different terms”.
While the deliberations have been on since January 2016, last month the board of Actelion felt “J&J’s proposal offered significantly greater transaction certainty because the transaction documentation was nearly final and because J&J had already completed the required due diligence”.
Sarepta sells priority review voucher to
Gilead for US$ 125 million
Sarepta Therapeutics has sold its priority review voucher (PRV) to Gilead
for US$ 125 million. The voucher is awarded under the rare pediatric PRV program. It speeds up the US FDA approval
process for any future drug or biologic from 10 months to six months.
For Gilead, this is the third PRV it has purchased. Last year, it bought PaxVax’s PRV for about US$ 200 million and in 2014 it bought Knight Therapeutics’ PRV for US$ 125 million.
The voucher was awarded to Sarepta after it won FDA approval for its DMD drug Exondys 51 (eteplirsen)
in September 2016.
Sarepta’s pricing is a far-cry from the most expensive PRV ever sold by United Therapeutics to AbbVie
for US$ 350 million in August 2015.
It will be interesting
to see what Marathon Pharmaceuticals who also received the most recently awarded PRV
for its DMD drug Emflaza (deflazacort) uses their voucher for after an “outrageous” pricing controversy paused the sales of their product.
With the passage of the 21st Century Cures
law, the number of PRVs on the market is likely to increase. This legislation reauthorizes the rare pediatric PRV program until October 2020.
Broad Institute to keep valuable patents on gene-editing technology — CRISPR
A biological and genomic
research center affiliated with MIT and Harvard known as the Broad Institute will
keep valuable patents on a
revolutionary gene-editing technology known as CRISPR. A US patent agency Patent
Trial and Appeal Board in Alexandria, Virginia rejected a
claim by a rival team associated
with the University of California at Berkeley and University of Vienna in
Austria that they invented the technology first.
CRISPR is a biotechnology innovation
that makes it easier for scientists to edit DNA in living cells. It works as a
type of molecular scissors that can trim away unwanted pieces of genetic
material, and replace them with new ones.
The patent rights for Broad
Institute could be worth billions of dollars, as the technology could
revolutionize treatment of genetic diseases, crop engineering and other areas.
In 2012, a research team led by Berkeley’s Jennifer Doudna and Vienna’s Emmanuelle Charpentier was first to apply for a CRISPR patent.
Months later, a team at Broad Institute, led by MIT’s Feng Zhang, applied for a patent by opting for a fast-track review process. It obtained a CRISPR patent in 2014, and has since obtained additional patents. Meanwhile, Berkeley is not the least bit happy
with the ruling and may appeal.
USFDA accepts Mylan and Biocon’s second biosimilar application of Amgen’s Neulasta
Last week, Mylan and Biocon announced that the US FDA has accepted Mylan’s Biologics License Application (BLA) for Pegfilgrastim,
a proposed biosimilar.
This product is a biosimilar version of Amgen Inc’s branded drug Neulasta, which is used to reduce the chance of
infection due to low white blood cell count and incidence of fever in patients
treated with chemotherapy in certain types of cancer.
Mylan and Biocon are exclusive partners on a
broad portfolio of biosimilars and generic insulin analogs. Pegfilgrastim is
one of the six biologic products co-developed by the companies.
The FDA goal date set under
the Biosimilar User Fee Act is October 9, 2017. The two companies have already
filed biosimilar pegfilgrastim with regulators in Europe, Australia and Canada.
Mylan President Rajiv Malik commented: “We’re proud of the FDA acceptance of our BLA for proposed biosimilar pegfilgrastim. This is the second BLA accepted for review by FDA as part of the Mylan and Biocon partnership within the past two months.”
Impressions: 2589
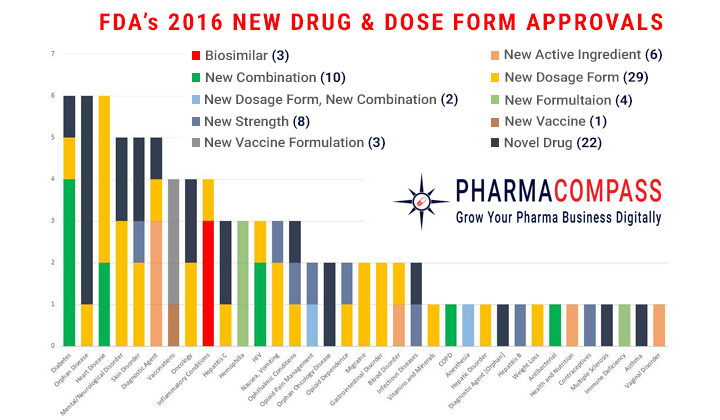
After 2 years of sky-high approval numbers, the US Food and Drug Administration’s (FDA) Center for Drug Evaluation and Research approved 22 novel drugs in 2016, down from the 19-year high of 45 in 2015. The FDA also approved many new dose forms, formulations, combination products and vaccines.
This
week, PharmaCompass, shares its analysis of the new drug approvals by the FDA
in 2016.
Reasons behind the low approvals in 2016
Of the 22 novel drugs approved by the FDA, the FDA approved 9 products with orphan designation, in line with the industry’s recent focus on rare diseases. However, as the industry shifts its focus towards biotechnology, only 7 of the novel products approved were biologic applications.
The 9
orphan designees approved (41% of all new drug approvals) were significantly lower than the 21 (47%) orphan designees approved in 2015 and 17 (41%) in 2014. FDA’s approval of 4 (18%) oncology drugs in 2016, was also down from the 14 (31%) approvals in 2015, 9 (22%) in 2014 and 9 (33%) in 2013.
The
reasons for the low 2016 approval count, have been attributed to the agency
approving five drugs in 2015 that actually had approval action dates in 2016
and an increase in the number of drugs that the agency rejected.
A key reason for the rejections was the sponsors’ failure to comply with good manufacturing practice regulations.
Click here to access our list of all New Drug Approvals (Excel version available) for FREE!
Gilead’s Epclusa, the first
all-oral, pan-genotypic, single tablet regimen for the treatment of adults with
chronic hepatitis C virus (HCV) infection, is expected to become the most
profitable approval of 2016 with expected sales of $ 8.4 billion by 2022.
2016 also saw the FDA’s contentious approval of Exondys 51™ (eteplirsen) under its accelerated approval process. Exondys 51™ treats Duchenne muscular dystrophy (DMD), a rare genetic disease which causes progressive muscle wasting that affects around 20,000 boys and young men in the United States.
Approvals of three biosimilars which
target $ 33 billion in brand drug sales
In a
giant leap for the generic pharmaceutical industry, three biosimilar
applications were approved in 2016 that target brand drugs which generated more
than $ 33 billion in sales last year.
All the three biosimilars approved were tumor-necrosis factor alpha (TNF α) inhibitors used to manage inflammatory conditions.
Pfizer and Celltrion’s Inflectra, a
biosimilar to Janssen’s Remicade® (2016 sales - $ 8.8 billion), was the first to get approval for all Remicade-approved indications, except pediatric ulcerative colitis. Inflectra was launched at risk in November 2016.
After Inflectra’s approval, the FDA approved Sandoz’s Erelzi, a biosimilar to Enbrel® (etanercept – Amgen, 2016 sales - $
9.1 billion).
On Sept. 23, 2016, Amgen’s Amjevita™ (adalimumab – atto), a biosimilar of the world’s best-selling drug by revenue, AbbVie’s Humira®(2016 sales - $ 16.4 billion), was also approved for treating adults with a variety of medical conditions ranging from rheumatoid arthritis, plaque psoriasis, to ulcerative colitis.
Click here to access our list of all New Drug Approvals (Excel version available) for FREE!
Kim Kardashian’s pregnancy drug is now available in a new strength
Diclegis,
a combination of an antihistamine (doxylamine succinate) and a form of vitamin B6 (pyridoxine HCl), made headlines
when the FDA issued a warning letter for the social media promotion of the drug by Kim Kardashain, star of the reality show ‘Keeping Up With The Kardashians’
Duchesnay,
the company which produces Diclegis, got approval for Bonjesta which combines twice
the amount of doxylamine and pyridoxine when compared with Diclegis which contains
10mg of each ingredient.
Indian generic companies get approvals for
differentiated products
As the
generic industry in the U.S. continues to face severe pricing pressure, Indian
generic companies are attempting to overcome these challenges by trying to move
up the value chain by supplying differentiated generic products.
Consistent
with this strategy, the applications of Sun Pharmaceuticals
for a new ophthalmic version of Bromfenac (Bromsite) and Dr. Reddy’s Laboratory
for an injectable version of Sumatriptan (Zembrace™ SymTouch) to treat migraines were also approved.
Click here to access our list of all New Drug Approvals (Excel version available) for FREE!
Pfizer launches an opioid treatment with
abuse-deterrent properties
The abuse
of opioids, including prescription painkillers and drugs like heroin, is
something the United States has struggled with since before the 1900s. Last year, the FDA announced that immediate-release
opioid painkillers such as oxycodone and fentanyl will have to
carry a "black box" warning
about the risk of abuse, addiction, overdose and death.
Pfizer
got approval last year for Troxyca ER a combination containing oxycodone and naltrexone. Troxyca ER has properties that are
expected to reduce abuse when crushed and administered by the oral and
intranasal routes.
In it,
the oxycodone releases slowly over several hours. If the capsules are crushed,
the encased naltrexone mixes with oxycodone, essentially cancelling any euphoric
effects.
Pfizer
also received approval for an extended release form of Tofacitinib (Xeljanz® XR) to treat adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response or intolerance to methotrexate.
Click here to access our list of all New Drug Approvals (Excel version available) for FREE!
A dietary supplement gets approved along with new forms of commonly used drugs
In 2016, Endoceutics
got Intarosa, a once-daily vaginal insert, approved which is the first FDA
approved product to contain prasterone, also known as dehydroepiandrosterone (DHEA).
Although
DHEA is included in some dietary supplements, the efficacy and safety of those
products have not been established for diagnosing, curing, mitigating, treating
or preventing any disease.
Lisinopril, the most commonly used drug
by U.S. Medicare patients, was approved as an Oral Solution for the first time.
Diabetes
treatments, one of the most commonly prescribed drug categories in the U.S., saw
the addition of Sanofi and Novo Nordisk’s fixed-dose,
long-acting insulin and glucagon-like peptide 1
(GLP-1) agonist combinations to the list of options available to treat adult
type-2 diabetes.
Our View
The approval
of new drugs ensures that the world has access to improved healthcare solutions
and breakthrough medical therapies.
With Donald Trump “focused on accelerating the FDA” and an on-going argument that drugs should not have to be proven effective before getting approved, this year looks like the one where the new drug
approval scenario will be shaken up for time to come.
Click here to access our list of all New Drug Approvals (Excel version available) for FREE!
Impressions: 14454
This week, Phispers delves into Pfizer’s acquisition of Medivation and its impact on Sanofi. There is also news on antipsychotic drug Abilify, besides snippets on a lawsuit against Valeant, Mylan’s price hikes and more quality snags that were unveiled at Pfizer’s plant near Chennai. Read on.
Rough week for Sanofi as FDA
delays its diabetes drugs, while Pfizer walks away with Medivation
Pfizer
agreed to pay US $14 billion in cash for Medivation in a deal that adds the
prostate cancer drug Xtandi
to its product portfolio.
Medivation was one of the few independent companies with a cancer treatment
that is selling well. Xtandi currently generates about US $ 2 billion a year in
annual sales.
The acquisition has come as a setback
for French drug maker Sanofi that spent five months pursuing Medivation. At one point, Sanofi even attempted to replace Medivation’s board and force a deal.
There
was another setback for Sanofi,
as it saw the launch of its diabetes combination medicine get delayed until
November this year. Sanofi spent US $ 245 million on a priority
review voucher to beat Danish drug maker Novo
Nordisk to market with a diabetes drug that pairs its best-selling
medicine Lantus
(a basal insulin) with lixisenatide. However, the FDA’s fast-track review pushed out the launch of the combination till at least November.
The FDA asked Sanofi for more data on the dual-drug delivery pen – a device that triggered debate during an FDA advisory panel review in May this year.
The FDA decision on Novo’s product is due next month. With this unexpected delay, Novo could get more than a two months’ head start over Sanofi.
FDA warns against the
use of antipsychotic drug Abilify
In the US, patients taking the antipsychotic drug Abilify have
reported uncontrollable urges to gamble, binge eat, shop, and have sex, according
to the FDA. The regulator issued a warning this week
on the drug, which is one of the top-selling prescription medications in the
United States.
Other serious side effects of the drug include a higher chance
of developing diabetes and hyperglycemia, and increased risk of suicide among
patients under the age of 24.
Also known as aripiprazole,
the drug is used to
treat schizophrenia, and can be used in combination with other drugs to
treat depression. In the US, 1.6 million patients received Abilify
prescriptions last year.
The warning comes amid pending class-action lawsuits against
the manufacturer of Abilify – Otsuka America Pharmaceutical. The class-action suits allege the company didn’t properly warn patients about the possibility of impulse-control issues.
However, these problems are rare. In the 13 years since the
drug was approved, there have been only 167 reports of patients experiencing
significant impulse-control problems, according to the FDA.
Counterfeit pills may
have killed the late singer Prince
A little over a year ago, the Drug Supply Chain Security
Act (or Drug Quality and Security Act) became effective in the United States. The law was introduced to secure the supply chain of medicines and restrict counterfeit drugs – an industry estimated to be bigger than Pfizer and GSK put
together.
It seems that regulatory agencies still have a lot of work to do in this
area. A mis-labelled bottle of
pills found in the home of the late singer Prince contained the powerful
painkiller fentanyl, a synthetic opioid 50 times more powerful than heroin. Prince died on April 21 this year.
According
to sources close to the investigation, the pills were found in a bottle
of Aleve, an over-the-counter medication sold in the US that contains
the painkiller naproxen. Two dozen pills, found in one bottle, were falsely labelled as ‘Watson 385’ – an identifier for a mix of acetaminophen
(paracetamol)
and hydrocodone.
Valeant mired in trouble
with a lawsuit and faltering sales of female sexual dysfunction drug
According to a lawsuit
filed last week, Valeant
Pharmaceuticals refilled
patient prescriptions without their permission and directed them to buy expensive drugs in
order to boost sales. The lawsuit provides insight into how a mail-order pharmacy – Philidor Rx Services – assisted Valeant in directing prescriptions to its brand-name medicines over cheaper generic versions.
Meanwhile,
Valeant hired Paul Herendeen as its new chief financial officer, luring the
executive away from Zoetis,
the animal health products maker.
While
Valeant is busy battling
challenges on various fronts, its US $
1 billion acquisition of Sprout Pharmaceuticals of Addyi – the first medicine to combat female sexual dysfunction – is turning
out of be a bust, as it has reported meagre sales.
Pfizer’s plant in Chennai faces more compliance issues
Earlier this month, Pfizer had to halt production at a plant near Chennai in India, after a Pharmaceutical
Inspection Convention and Pharmaceutical Inspection Co-operation Scheme (PIC/S)
joint inspection highlighted quality concerns. The other regulators in the
PIC/S inspection were the Medicines and Healthcare products Regulatory Agency
of the UK (MHRA), the United States Food and Drug Administration (USFDA),
Therapeutic Goods Administration of Australia (TGA) and Health Canada.
Three
years ago, the plant was first cited with an FDA warning letter. Last year, when
Pfizer acquired the plant from Hospira, it was well aware of the quality issues.
But Pfizer was probably not aware of the extent of troubles that awaited it. Last
week, a GMP non-compliance report posted by European Medicines Agency (EMA) listed
that an inspection by the MHRA uncovered
a variety of critical issues, raising doubts on whether
the injectable products coming out of the facility are sterile or not.
MHRA inspectors found that employees were using aseptic processes that could allow for microbial contamination. Pfizer’s investigations into issues were not getting to root causes of problems, they said. All of the plant’s shortcomings were linked to employees who lacked the “scientific knowledge” to know what to do.
The MHRA withdrew the plant’s GMP certificate and has halted imports to the European Union of six injected antibiotics until the problems get addressed.
Mylan’s outrageous drug price hikes for EpiPen come under scrutiny
The EpiPen auto-injector, which reverses life-threatening
allergic reactions, is under scrutiny. In 2015, the drug had generated US $ 1.2
billion in sales for Mylan.
The EpiPen has been around since 1977, but Mylan acquired
the auto-injector in 2007. The EpiPen precisely calibrates the dosage of epinephrine.
The patient now pays
about 400 percent more for this advantage to receive a dollar’s worth of the life-saving drug. EpiPens were sold for about US $ 57 when Mylan acquired it. Today, it is being
sold at US $ 500 or more in the US.
Meanwhile, Senator Amy Klobuchar (District Minnesota) has
asked the US Federal Trade Commission and the Senate Judiciary Committee to investigate
price hikes undertaken by Mylan. Klobuchar is the ranking member of Senate
Judiciary Antitrust Subcommittee. And Senator Richard Blumenthal (District Connecticut)
wrote to the company for data about assistance programs for
patients and first responders. He also
demanded that Mylan lower its price.
Last year, Mylan raised the price on EpiPen — its biggest-selling product — twice by 15 percent (each time). Due to lack of competition, the
price hikes were easy.
Korea’s Celltrion ships first batch of biosimilar Remicade to US
Last week, a day after winning a lawsuit in the US, Korean pharmaceutical firm Celltrion shipped
the first batch of its biosimilar medicine -- Remsima – to the country. The lawsuit was on the sale of Remsima – an autoimmune disorder drug – in the US, the world’s largest pharmaceutical market.
Celltrion said that the move will accelerate the US launch of Remsima, a
biosimilar version of Janssen's
Remicade. Remsima has been on sale in Europe since 2013.
Pfizer will take charge of sales of Remsima in the US. The drug will soon be
available to patients in the US suffering from rheumatoid arthritis and
ulcerative colitis under the brand name of Inflectra.
Remicaid’s sales were in excess of US $
8 billion in 2015. Celltrion
CEO Kim Hyoung-ki has projected that the company will earn more than US $ 1.7 billion in the US market next year, assuming a
double-digit market share.
Four healthcare CEOs on the world’s top 20 severance packages list
Even though the Pfizer-Allergan US $ 160 billion merger did not go through, Allergan CEO’s Brent Saunders has little
to complain. In a recent Bloomberg
compilation, his severance package of US $ 140 million ranks in the top
20 of all S&P 500 CEOs.
Joining him in the top 20 are other CEOs of healthcare
companies such as McKesson (with a severance package of US $ 198 million),
Aetna (US $ 91 million) and Regeneron (US $ 90 million).
McKesson Corporation is an American company distributing
pharmaceuticals at a retail sale level and providing health information
technology, medical supplies, and care management tools.
Aetna is an American managed health care company, which sells
traditional and consumer directed health care
insurance plans and related services, such as medical, pharmaceutical, dental,
behavioral health, long-term care, and disability plans.
Regeneron is a US-based biotechnology company with four FDA approved products and over US $ 4 billion in revenues in 2015.
Impressions: 5522
GSK, Google form first bioelectronics firm; 11 generic companies benefit from the Teva Allergan deal
This week,
Phispers brings to you the details of the bioelectronics firm formed by GSK and
Google. There is also news on companies like Teva, Takeda, Jinan Jinda and Eli
Lilly, besides two other news snippets pertaining to the FDA -- while the first
one pertains to generic approvals, the other one relates to an additional black
box warning on a few antibiotics. GSK and Google
join hands to form first bioelectronics startupGlaxoSmithKline and Google’s parent company – Alphabet – have joined hands to create a new company that is focused on fighting diseases by targeting electrical signals in the human body. This way, GSK and Alphabet’s life sciences unit – known as Verily Life Sciences – will be jump-starting a new field of medicine known as bioelectronics.Verily Life
Sciences and GSK will together contribute US $ 715.12
million
over seven years to the startup Galvani Bioelectronics. The startup will develop
miniature electronic implants for the treatment of asthma, diabetes and other
chronic conditions. The
implantable devices developed by Galvani, which is owned 55 percent by GSK and
45 percent by Verily, can modify electrical nerve signals. The aim is to
modulate irregular or altered impulses that occur in many illnesses.The
new company
will be based at GSK’s Stevenage research center north of London, with a second research hub in South San Francisco.The announcement comes just weeks after GSK had said it was going to use Apple’s HealthKit to conduct clinical trials.Three years ago, GSK had first unveiled its ambitions in bioelectronics in the journal – Nature. Bioelectronic remedies attach battery-powered implants the size of a grain of rice (or even smaller) to individual nerves to correct faulty electrical signals between the nervous system and the body’s organs.GSK believes altering these nerve signals could open up the airways of asthma patients, reduce inflammation in the gut from Crohn’s disease and treat patients with a range of other chronic ailments such as arthritis. So far, the implants have only been tested on animals but the aim is to produce treatments that will supplement or replace drugs that often come with side-effects.GSK
has been working on bioelectronic medicines since 2012 in a push to develop new
patentable treatments, since its Advair respiratory treatment faces competition
from generic versions. It has invested US $50 million in a venture capital fund
for bioelectronics and provided funding to scientists working in the field. Teva divests 79
products to 11 generic players to close Allergan dealTeva
Pharmaceutical Industries – the world’s largest generics drug company – won a US
anti-trust approval to purchase Allergan's generics
business, after agreeing to divest 79 generic drugs to rival firms. This was arrived
at to settle Federal
Trade Commission (FTC) charges that its proposed US $ 40.5 billion acquisition of Allergan’s generic pharmaceutical business would be anti-competitive. The remedy requires Teva to divest the drug portfolio to 11 firms, and marks the largest drug divestiture order in a FTC pharmaceutical merger case.The Teva-Allergan deal, which was announced in July 2015, solidifies Teva’s position as the world's largest maker of generics while freeing Allergan to focus on branded drugs.The
companies that
have acquired
the divested products are Mayne Pharma
Group, Impax
Laboratories, Dr Reddy’s Laboratories, Sagent
Pharmaceuticals, Cipla Limited, Zydus Worldwide
DMCC, Mikah Pharma, Perrigo Pharma
International, Aurobindo
Pharma USA,
Prasco and 3M Company. Eli Lilly CEO
steps down; company under probe by US Justice Department Eli Lilly CEO John
Lechleiter has stepped down after steering the pharma company through long R&D droughts. The company’s president David Ricks will move up to the top spot. And after a brief spell as executive chairman, Lechleiter will leave the company next spring.Lechleiter
has been the company's CEO since April 1, 2008, and the chairman of its board
of directors since January 1, 2009.The
announcement has come at a time when Eli Lilly has been asked by the
Justice Department to disclose information on relationships with pharmacy benefits
managers (PBMs), the companies that negotiate prices and set reimbursement
conditions.It
has not been clear what exactly the department of justice is looking for. In
the past, drug makers such as Novartis and AstraZeneca have agreed to
pay fines and penalties to settle allegations pertaining to PBMs. FDA continues
to race ahead with generic approvals The
American regulator has reduced its pile of ANDA (abbreviated new drug
applications) by about 500
applications in the first six months of 2016. The FDA has also approved 315 more ANDAs over the same time period and has sent 66 more complete response letters — or rejections — to drug makers.This
news comes after Bloomberg reported
last month that the FDA has become ‘something of a bogeyman’ for India’s stock markets by approving generic drug applications from India at a record place. Similarly, PharmaCompass
had reported last week that Indian
companies have been fixing compliance issues. China’s Jinan Jinda fails another EDQM inspection; compliance troubles in Denmark In
regulatory news from across the world, Jinan Jinda, a Chinese API
manufacturer that had failed an inspection by Italian regulators in June 2015,
had more bad news awaiting it a year on. In
a June 2016
re-inspection, this time by the Spanish Health Authority, the regulator maintained the ‘facilities non-compliance standing’ since two critical observations were made and the corrections from the previous inspection “were found as not having been implemented in a satisfactory way”. And critical deficiencies were found on raw data.In
the June 2015 inspection, the critical observation was related to an unofficial
and non-controlled storage area containing mainly raw materials and finished
products which had been made
inaccessible to inspectors as the door had been removed and replaced with a panel fixed with
screws to the wall.Meanwhile,
the FDA issued an untitled letter (dated July 15, 2016) to Danish allergy
immunotherapy company ALK-Abelló (ALK) over manufacturing and quality control issues at its Horsholm, Denmark facility. The letter comes after a 12-day inspection of the facility in March 2016. During the inspection, the FDA had cited ALK for four “significant deviations” from cGMP requirements. Another black
box warning added to antibiotics like Cipro and LevaquinThe
FDA has upgraded
warnings on
certain antibiotics, such as Johnson & Johnson’s Levaquin, Bayer’s Cipro
extended-release tablets and Merck’s Avelox. The FDA had
added a black box warning in 2008 about the increased risk of tendinitis in
which the tissue connecting muscle to bone becomes inflamed. In
May this year, the FDA had advised restricting the use of fluoroquinolone antibiotic for certain uncomplicated
infections and had warned about the disabling side-effects of the drug.The new warning talks about long-term risks to the drugs’ current black box warning. The agency also advised using the drugs only for serious infections. Manufacturers of fluoroquinolone have faced thousands of lawsuits from patients who claim that their injuries were caused by the drugs. J&J alone faced 3,400 lawsuits over Levaquin’s links to tendon problems and has also settled many of those cases. Takeda to
overhaul R&D, downsize operations in the UKTakeda Pharmaceutical of Japan has
said it plans to build a new pipeline of drugs. It plans to revamp its
research operations at the cost of around US $ 727 million.. The
company also plans to close some of its R&D operations in the UK. Takeda is
beginning the first ‘consultation stage’ of the layoff process in the UK, which hosts a pre-clinical R&D operation in Cambridge as well as a development center headquarter with facilities in the UK, Switzerland and Denmark.Under the revamp, Takeda’s R&D activities will be concentrated in Japan and the US, the 235-year old drug company said in a statement. Takeda plans to now focus on the three therapeutic areas of oncology, gastroenterology and the central nervous system.“We need to first build new capabilities and embrace new ways of working,” Andy Plump, Takeda’s chief medical and scientific officer, said in the statement.
Impressions: 2757
Teva Pharmaceutical Industries, Ltd., which acquired Cephalon in 2012, will make a total payment of $1.2 billion as part of a ‘pay-for-delay’ settlement reached with the Federal Trade Commission (FTC) last week.
What exactly did Cephalon, for which Teva paid $6.8 billion, do so wrong? Isn’t ‘pay-for-delay’ common practice in the pharmaceutical industry?
First of all what is a
pay-for-delay?
‘Pay for delay’ or reverse payment patent settlements, are agreements where the brand name drug manufacturer compensates generics, not to market the generic product for a specific period of time.
These settlements allow the brand manufacturers to extend their
patent monopolies and according to an FTC study, these deals
cost consumers and taxpayers $3.5 billion in higher drug costs every year.
What exactly happens and
why is it a big deal now?
Cephalon allegedly paid four generic drug companies (Teva, Ranbaxy Pharmaceuticals, Mylan Pharmaceuticals, and Barr Laboratories), over $300 million in total. In return the generics agreed to drop their patent challenges and forgo marketing of their generic versions of Cephalon’s blockbuster sleep-disorder drug Provigil, for six years, until April 2012.
An extended monopoly for Provigil, in the absence of generic competition, was “$4 billion in sales that no one expected”, the CEO of Cephalon reportedly said when the deal was struck.
While in Europe, regulators have been going after pay-for-delay cases for years, it was only as recently as 2013, in FTC v. Actavis, that the U.S. Supreme Court made clear that reverse payment patent settlements are subject to the same antitrust rules that govern general U.S. business conduct.
The payment made by Teva will compensate purchasers, including drug wholesalers, pharmacies, and insurers, who overpaid because of Cephalon’s illegal conduct, is the first positive outcome for the FTC after the Supreme Court ruling.
How common are ‘pay-for-delay’ settlements?
Based on data provided by the FTC, for the past few years, more
than 100 settlements are reached annually between brand and generic
pharmaceutical companies.
Over 30% of these settlements have the potential of being ‘pay-for-delay’ agreements.
Table// Potential
pay-for-delay settlements reached between brand and generic companies:
Financial Year
2004
2005
2006
2007
2008
2009
2010
2011
2012
2013
Final Settlements:
between brand and generic companies
14
11
28
33
66
68
113
156
140
145
Involving First Generic Filing
8
5
11
16
29
32
49
54
43
41
Potential Pay-for-Delay:
Involving First Generic Filing
2
9
11
13
15
26
18
23
13
Settlements
3
14
14
16
19
31
28
40
29
How severe are the penalties for ‘pay-for-delay’ settlements in Europe?
The European Commission has
fined Johnson
& Johnson (J&J) just under 10.8 million
euros and Novartis 5.49 million euros, after discovering a ‘pay-for-delay’ deal on the painkiller Duragesic (fentanyl).
The amount pales in comparison to
the whopping €428m fine on Servier and several other companies (Niche/ Unichem; Matrix, which is now part of Mylan; Teva; Krka and Lupin) for conspiring to delay generics of the widely-used blood pressure drug Coversyl/ Aceon (perindopril).
In yet another settlement, agreements which operated in 2002 and
2003 between the Danish originator Lundbeck, and other
generic companies, resulted in Euro
146 million in fines.
What should we expect in the future?
Based on an FTC
presentation made in September 2014, they highlighted 19 Cases to Watch, which has them targeting almost every
major brand and generic pharmaceutical company. However, with the complexities
involved, this list is continuously evolving:
The
cases (by name of the brand product) Actos,
Adderall,
Aggrenox,
AndroGel,
Cipro,
Effexor,
K-Dur, Lamictal, Lidoderm,
Lipitor,
Loestrin,
Nexium,
Niaspan,
Opana,
Provigil,
Skelaxin,
Solodyn,
Wellbutrin.The
brand companies involvedAbbvie,
Abbott,
AstraZeneca,
Bayer,
Besins,
Biovail,
Boehringer,
Cephalon,
Endo,
GlaxoSmithKline,
King,
Medicis,
Pfizer,
Shire,
Schering,
Takeda,
Warner
Chilcott, Wyeth.The
generic companies Actavis
, Barr,
Duramed,
Dr. Reddy’s, HMR, Impax,
Lupin,
Mutual,
Mylan, Par, Perrigo,
Ranbaxy,
Rugby, Sandoz,
Teva,
Upsher
Smith.
Our view:
Pharmaceutical companies,
lawyers and the FTC will be busy for the coming few years, since there are a
series of suits, which will be challenging settlements reached between brand
and generic pharmaceutical companies.
While patents
provide temporary monopolies to promote innovation, brand drug manufacturers
will need to resort to more innovative ways of sustaining their profits.
Click here and learn about the different strategies adopted in the United States to block generics?
Impressions: 3400