

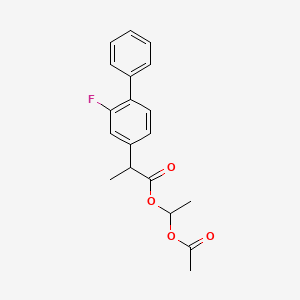

1. Fp 83
2. Fp-83
3. Fp83
1. 91503-79-6
2. Ropion
3. Lfp 83
4. Fp 83
5. Fp-83
6. I0ou31pui5
7. Lfp-83
8. Dtxsid8048600
9. Fp83
10. Dtxcid1028526
11. (1,1'-biphenyl)-4-acetic Acid, 2-fluoro-alpha-methyl-, 1-(acetyloxy)ethyl Ester
12. Refchem:141041
13. 1-(acetyloxy)ethyl 2-(2-fluoro-(1,1'-biphenyl)-4-yl)propanoate
14. 1-acetoxyethyl 2-(2-fluoro-[1,1'-biphenyl]-4-yl)propanoate
15. 1-acetyloxyethyl 2-(3-fluoro-4-phenylphenyl)propanoate
16. 1-acetoxyethyl 2-(2-fluoro-4-biphenylyl)propionate
17. Mfcd00900049
18. Lipfen
19. Lfp 83;ropiopn;lfp-83;lfp83
20. Flurbiprofen Axetil (jan)
21. Flurbiprofen Axetil [jan]
22. Lipo-flurbiprofen Axetil
23. Liposomal Flurbiprofen Axetil
24. Cas-91503-79-6
25. C19h19fo4
26. Unii-i0ou31pui5
27. Lfp83
28. Ncgc00182710-01
29. Ncgc00182710-02
30. 2-fluoro-a-methyl-[1,1'-biphenyl]-4-acetic Acid 1-(acetyloxy)ethyl Ester; Flurbiprofen Axetil
31. 1-acetoxyethyl-2-(2-fluoro-4-biphenylyl)propionate
32. Ropion (tn)
33. 4-biphenylacetic Acid, 2-fluoro-alpha-methyl-, 1-acetoxyethyl Ester
34. 2-fluoro-alpha-methyl-(1,1'-biphenyl)-4-acetic Acid 1-(acetyloxy)ethyl Ester
35. Orb1304142
36. Schembl1649401
37. Chembl3183067
38. Schembl29370747
39. Chebi:31627
40. Hms5085j04
41. Msk10984
42. Tox21_113023
43. Ac-455
44. Flurbiprofen Axetil [who-dd]
45. S6457
46. Akos024464989
47. Tox21_113023_1
48. Db14938
49. Ds-8257
50. Fa23508
51. Da-33622
52. Hy-101481
53. Cs-0021519
54. F1176
55. D01475
56. F17835
57. Sbi-0654123.0001
58. 1-acetoxyethyl-2-(2-fluoro-4-biphenyl)propionate
59. 1-acetoxyethyl 2-(2-fluoro-4-biphenylyl)propanoate
60. Q27280207
61. 1-(acetyloxy)ethyl 2-(2-fluorobiphenyl-4-yl)propanoate
62. Acetoxyethyl 2-(2-fluoro-[1,1'-biphenyl]-4-yl)propanoate
63. 1-(acetyloxy)ethyl 2-(2-fluoro[1,1'-biphenyl]-4-yl)propanoate
64. 2-fluoro-a-methyl-[1,1'-biphenyl]-4-acetic Acid 1-(acetyloxy)ethyl Ester
65. (1,1'-biphenyl)-4-acetic Acid, 2-fluoro-.alpha.-methyl-, 1-(acetyloxy)ethyl Ester
66. Lfp 83; Ropiopn; 1-acetoxyethyl 2-(2-fluoro-4-biphenylyl)propionate; 1-acetoxyethyl 2-(2-fluoro-[1,1'-biphenyl]-4-yl)propanoate
| Molecular Weight | 330.3 g/mol |
|---|---|
| Molecular Formula | C19H19FO4 |
| XLogP3 | 4.9 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 7 |
| Exact Mass | Da |
| Monoisotopic Mass | Da |
| Topological Polar Surface Area | 52.6 |
| Heavy Atom Count | 24 |
| Formal Charge | 0 |
| Complexity | 433 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 2 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Anti-Inflammatory Agents, Non-Steroidal
Anti-inflammatory agents that are non-steroidal in nature. In addition to anti-inflammatory actions, they have analgesic, antipyretic, and platelet-inhibitory actions. They act by blocking the synthesis of prostaglandins by inhibiting cyclooxygenase, which converts arachidonic acid to cyclic endoperoxides, precursors of prostaglandins. Inhibition of prostaglandin synthesis accounts for their analgesic, antipyretic, and platelet-inhibitory actions; other mechanisms may contribute to their anti-inflammatory effects.
