
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Europe
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
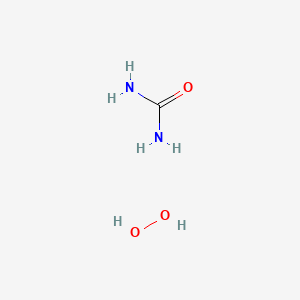

1. Hydrogen Peroxide, Urea
2. Perhydrol Urea
3. Urea Hydrogen Peroxide
4. Urea Peroxide
1. Urea Hydrogen Peroxide
2. 124-43-6
3. Percarbamide
4. Urea Peroxide
5. Urea Dioxide
6. Urea Hydroperoxide
7. Hydroperit
8. Hydroperite
9. Percarbamid
10. Perhydrit
11. Thenardol
12. Hyperol
13. Ortizon
14. Perhydrol-urea
15. Hydrogen Peroxide Carbamide
16. Hydrogen Peroxide Urea
17. Murine Ear Drops
18. Urea Compound With Hydrogen Peroxide (1:1)
19. Carbamide Peroxide, Solution
20. Hydrogen Peroxide;urea
21. Hydrogen Peroxide-urea Adduct
22. Urea Hydrogen Peroxide Adduct
23. Urea, Compd. With Hydrogen Peroxide (1:1)
24. Carbamide Peroxide [usp]
25. Urea, Compd. With Hydrogen Peroxide (h2o2) (1:1)
26. 31pz2vau81
27. Chebi:75178
28. Nsc-24852
29. Carbamide Peroxide (usp)
30. 14479-85-7
31. Hydrogen Peroxide; Urea
32. Proxigel
33. Debrox
34. Gly-oxide
35. Thera-ear
36. Ear Wax Treatment
37. Ureahydrogenperoxide
38. Auro Ear Wax Remover
39. Einecs 204-701-4
40. Nsc 24852
41. Un1511
42. Unii-31pz2vau81
43. Per Carbamide
44. Hydrogen Peroxide, Compd. With Urea (1:1)
45. Carbamide Peroxide Otic Solution
46. Mfcd00013119
47. Urea-hydrogen Peroxide
48. Urea.h2o2
49. Hydrogen Peroxide.urea
50. H2o2 Urea
51. Murine Ear Drops (tn)
52. Dsstox_cid_4726
53. Wln: Zvz & Qq
54. Dsstox_rid_77512
55. Dsstox_gsid_24726
56. Hydrogen Peroxide Urea Adduct
57. Urea Peroxide [inci]
58. Urea Hydrogen Peroxide, 97%
59. Carbamide Peroxide [mi]
60. Chembl3184026
61. Dtxsid9024726
62. Carbamide Peroxide [vandf]
63. Hydrogen Peroxide - Urea (1:1)
64. Urea, Compd. With Peroxide (1:)
65. Nsc24852
66. Tox21_302451
67. Urea Compound With Hydrogen Peroxide
68. Hydrogen Peroxide-urea Adduct, Tablet
69. Akos015904087
70. Db11129
71. Hydrogen Peroxide Urea ;perhydrol-urea
72. Hydrogen Peroxide-urea Compound (1:1)
73. Urea Hydrogen Peroxide (1:1)
74. Urea Hydrogen Peroxide [mart.]
75. Urea Hydrogen Peroxide [who-dd]
76. Ncgc00256660-01
77. (h2 N)2 C O (h2 O2)
78. Carbamide Peroxide [usp Monograph]
79. Cas-124-43-6
80. Hydrogen Peroxide, Compd. With Urea(1:1)
81. Urea, Compd. With Hydrogen Peroxide(1:1)
82. D03383
83. Urea Hydrogen Peroxide [un1511] [oxidizer]
84. A805233
85. J-005078
86. J-525152
87. Q-200793
88. Q2633879
89. Hydrogen Peroxide-urea Adduct, Usp, 96.0-102.0%
90. Hydrogen Peroxide-urea Adduct, Powder, 15-17% Active Oxygen Basis
91. Hydrogen Peroxide-urea Adduct, Purum P.a., "rapid-soluble", Tablet (1 G Each)
92. Uhp
| Molecular Weight | 94.07 g/mol |
|---|---|
| Molecular Formula | CH6N2O3 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 0 |
| Exact Mass | 94.03784206 g/mol |
| Monoisotopic Mass | 94.03784206 g/mol |
| Topological Polar Surface Area | 110 Ų |
| Heavy Atom Count | 6 |
| Formal Charge | 0 |
| Complexity | 29 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Indicated as a dental bleaching agent. Indicated as an oral wound healing agent in oral mucosal injuries. Indicated as an aid in the removal of hardened ear wax.
Carbamide peroxide releases hydrogen peroxide and free radicals upon contact with water or outer surfaces of ear and tooth. Hydrogen peroxide exerts cerumenolytic, enamel-bleaching and antiseptic actions. _In vitro_, the chemical stability of ceramics against bleaching agents was observed after treatment with 15% carbamide peroxide for 56 h, 16% carbamide peroxide for 126 h, 10% or 15% carbamide peroxide and 38% hydrogen peroxide for 30 minutes or 45 minutes, respectively. According to _in vitro_ studies, high (37%) or low (10 or 16%) concentrated carbamide peroxide agents were similarly effective as oral bleaching agents. Treatment with carbamide peroxide may lead to demineralization which involves decreased mineral content of enamel calcium, phosphate, and fluoride, and alteration of the chemical, structural, and mechanical properties. Carbamide peroxide may affect the organic components of the enamel and lead to increased susceptibility to erosion, fracture stability or decreased abrasion resistance of the treated area.
Anti-Infective Agents, Local
Substances used on humans and other animals that destroy harmful microorganisms or inhibit their activity. They are distinguished from DISINFECTANTS, which are used on inanimate objects. (See all compounds classified as Anti-Infective Agents, Local.)
Tooth Bleaching Agents
Chemicals that are used to oxidize pigments in TEETH and thus effect whitening. (See all compounds classified as Tooth Bleaching Agents.)
Absorption
Upon treatment into the external auditory canal or the dental cavity, exposure to carbamide peroxide is limited to the intimate contact with the treated area without any systemic absorption.
Route of Elimination
No established pharmacokinetic data.
Volume of Distribution
No established pharmacokinetic data.
Clearance
No established pharmacokinetic data.
No established pharmacokinetic data.
No established pharmacokinetic data.
Carbamide peroxide release hydrogen peroxide upon contact with teeth, which is a strong oxidizing and bleaching agent. It also release free radicals such as H+ or H3O+. Hydrogen peroxide also acts as an antiseptic, especially in sites with relative anaerobiosis. Following otic administration, carbamide peroxide complex releases hydrogen peroxide that breaks up the hardened wax. The hydrogen peroxide component, which further breaks down into water, is also a cerumenolytic that hydrates the desquamated sheets of corneocytes, which are the major constituent of cerumen plugs. The glycerol and urea facilitates softening of the cerumen, either with or without syringing. Both hydrogen peroxide and urea mildly induce keratolysis with disintegration of the ear wax to help reduce the keratin-load in the ear debris and allow other active components to reach the skin under the debris.
 Apex Healthcare Limited: ISO 9001:2008, WHO-GMP, US FDA-audited manufacturer & exporter of APIs, bulk drugs, and formulations.
Apex Healthcare Limited: ISO 9001:2008, WHO-GMP, US FDA-audited manufacturer & exporter of APIs, bulk drugs, and formulations.

NDC Package Code : 69617-015
Start Marketing Date : 2025-09-17
End Marketing Date : 2027-12-31
Dosage Form (Strength) : POWDER (25kg/25kg)
Marketing Category : BULK INGREDIENT

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 37188
Submission : 2022-06-17
Status : Active
Type : IV

Date of Issue : 2026-01-12
Valid Till : 2028-02-02
Written Confirmation Number : WC-0518
Address of the Firm :


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39868
Submission : 2024-04-17
Status : Active
Type : II

USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others
USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others

NDC Package Code : 67614-0034
Start Marketing Date : 2024-01-22
End Marketing Date : 2027-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF



 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
About the Company : Hoventa Pharmachem is an India-based pharmaceutical company engaged in the manufacturing, sourcing, and global supply of Active Pharmaceutical Ingredients (APIs), intermediates, an...

About the Company : Nano Chemazone is a cutting-edge company dedicated to advancing Nanomaterials & Nanotechnology through innovative research and development. Specializing in high-quality nanotechn...

About the Company : Roussel Delma Pharmaceuticals, a well-regarded and rapidly expanding pharmaceutical manufacturing company, has been a prominent presence in the Pharmaceutical & Healthcare Industry...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Related Excipient Companies

Excipients by Applications
Market Place

ABOUT THIS PAGE
98
PharmaCompass offers a list of Urea Hydrogen Peroxide API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Urea Hydrogen Peroxide manufacturer or Urea Hydrogen Peroxide supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Urea Hydrogen Peroxide manufacturer or Urea Hydrogen Peroxide supplier.
A Urea Hydrogen Peroxide manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Urea Hydrogen Peroxide, including repackagers and relabelers. The FDA regulates Urea Hydrogen Peroxide manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Urea Hydrogen Peroxide API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Urea Hydrogen Peroxide manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Urea Hydrogen Peroxide supplier is an individual or a company that provides Urea Hydrogen Peroxide active pharmaceutical ingredient (API) or Urea Hydrogen Peroxide finished formulations upon request. The Urea Hydrogen Peroxide suppliers may include Urea Hydrogen Peroxide API manufacturers, exporters, distributors and traders.
click here to find a list of Urea Hydrogen Peroxide suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Urea Hydrogen Peroxide DMF (Drug Master File) is a document detailing the whole manufacturing process of Urea Hydrogen Peroxide active pharmaceutical ingredient (API) in detail. Different forms of Urea Hydrogen Peroxide DMFs exist exist since differing nations have different regulations, such as Urea Hydrogen Peroxide USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Urea Hydrogen Peroxide DMF submitted to regulatory agencies in the US is known as a USDMF. Urea Hydrogen Peroxide USDMF includes data on Urea Hydrogen Peroxide's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Urea Hydrogen Peroxide USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Urea Hydrogen Peroxide suppliers with USDMF on PharmaCompass.
A Urea Hydrogen Peroxide written confirmation (Urea Hydrogen Peroxide WC) is an official document issued by a regulatory agency to a Urea Hydrogen Peroxide manufacturer, verifying that the manufacturing facility of a Urea Hydrogen Peroxide active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Urea Hydrogen Peroxide APIs or Urea Hydrogen Peroxide finished pharmaceutical products to another nation, regulatory agencies frequently require a Urea Hydrogen Peroxide WC (written confirmation) as part of the regulatory process.
click here to find a list of Urea Hydrogen Peroxide suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Urea Hydrogen Peroxide as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Urea Hydrogen Peroxide API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Urea Hydrogen Peroxide as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Urea Hydrogen Peroxide and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Urea Hydrogen Peroxide NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Urea Hydrogen Peroxide suppliers with NDC on PharmaCompass.
Urea Hydrogen Peroxide Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Urea Hydrogen Peroxide GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Urea Hydrogen Peroxide GMP manufacturer or Urea Hydrogen Peroxide GMP API supplier for your needs.
A Urea Hydrogen Peroxide CoA (Certificate of Analysis) is a formal document that attests to Urea Hydrogen Peroxide's compliance with Urea Hydrogen Peroxide specifications and serves as a tool for batch-level quality control.
Urea Hydrogen Peroxide CoA mostly includes findings from lab analyses of a specific batch. For each Urea Hydrogen Peroxide CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Urea Hydrogen Peroxide may be tested according to a variety of international standards, such as European Pharmacopoeia (Urea Hydrogen Peroxide EP), Urea Hydrogen Peroxide JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Urea Hydrogen Peroxide USP).

