
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Listed Dossiers
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Gsk 1120212
2. Gsk-1120212
3. Gsk1120212
4. Jtp 74057
5. Jtp-74057
6. Jtp74057
1. 871700-17-3
2. Gsk1120212
3. Mekinist
4. Gsk-1120212
5. Jtp 74057
6. Jtp-74057
7. Gsk 1120212
8. Trametinib (gsk1120212)
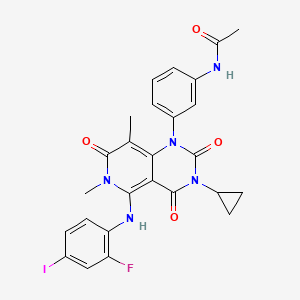
9. N-[3-[3-cyclopropyl-5-[(2-fluoro-4-iodophenyl)amino]-3,4,6,7-tetrahydro-6,8-dimethyl-2,4,7-trioxopyrido[4,3-d]pyrimidin-1(2h)-yl]phenyl]acetamide
10. Gsk212
11. Tmt212
12. Trametinib [usan]
13. Chebi:75998
14. Tmt-212
15. 33e86k87qn
16. Trametinib (usan)
17. N-(3-{3-cyclopropyl-5-[(2-fluoro-4-iodophenyl)amino]-6,8-dimethyl-2,4,7-trioxo-3,4,6,7-tetrahydropyrido[4,3-d]pyrimidin-1(2h)-yl}phenyl)acetamide
18. N-[3-[3-cyclopropyl-5-(2-fluoro-4-iodoanilino)-6,8-dimethyl-2,4,7-trioxopyrido[4,3-d]pyrimidin-1-yl]phenyl]acetamide
19. Acetamide, N-(3-(3-cyclopropyl-5-((2-fluoro-4-iodophenyl)amino)-3,4,6,7-tetrahydro-6,8- Dimethyl-2,4,7-trioxopyrido(4,3-d)pyrimidin-1(2h)-yl)phenyl)-
20. N-(3-(3-cyclopropyl-5-((2-fluoro-4-iodophenyl)amino)-6,8-dimethyl-2,4,7-trioxo-3,4,6,7-tetrahydropyrido[4,3-d]pyrimidin-1(2h)-yl)phenyl)acetamide
21. Unii-33e86k87qn
22. Trametinib [usan:inn]
23. Trametinibum
24. Jtp74057
25. N-(3-(3-cyclopropyl-5-((2-fluoro-4-iodophenyl)amino)-6,8-dimethyl-2,4,7-trioxo-3,4,6,7- Tetrahydropyrido(4,3-d)pyrimidin-1(2h)-yl)phenyl)acetamide
26. N-(3-(3-cyclopropyl-5-((2-fluoro-4-iodophenyl)amino)-6,8-dimethyl-2,4,7-trioxo-3,4,6,7-tetrahydropyrido(4,3-d)pyrimidin-1(2h)-yl)phenyl)acetamide
27. N-(3-{3-cyclopropyl-5-((2-fluoro-4-iodophenyl)amino)-6,8-dimethyl-2,4,7-trioxo-3,4,6,7- Tetrahydropyrido(4,3-d)pyrimidin-1(2h)-yl}phenyl)acetamide
28. Qom
29. Trametinib [mi]
30. Trametinib (gsk1120212jtp 74057)
31. Trametinib [inn]
32. Trametinib [vandf]
33. Trametinib [who-dd]
34. Schembl170938
35. Gtpl6495
36. Gsk1120212 (trametinib)
37. Chembl2103875
38. Ex-a022
39. Bcpp000218
40. Dtxsid901007381
41. Hms3295i05
42. Hms3656j11
43. Bcp02307
44. Bdbm50531540
45. Mfcd17215075
46. Nsc758246
47. Nsc800956
48. S2673
49. Zinc43100709
50. Akos015850628
51. Am90271
52. Ccg-264976
53. Cs-0060
54. Db08911
55. Ex-5957
56. Nsc-758246
57. Nsc-800956
58. Sb16553
59. Ncgc00263180-01
60. Ncgc00263180-07
61. Ncgc00263180-14
62. Ac-25891
63. As-19382
64. Hy-10999
65. N-[3-[3-cyclopropyl-5-(2-fluoro-4-iodo-anilino)-6,8-dimethyl-2,4,7-trioxo-pyrido[4,3-d]pyrimidin-1-yl]phenyl]acetamide
66. Ft-0688438
67. Sw218089-2
68. A25168
69. D10175
70. Gsk1120212 - Jtp-74057
71. Gsk1120212,jtp-74057, Gsk212
72. Sr-01000941589
73. A1-01871
74. J-523325
75. Q7833138
76. Sr-01000941589-1
77. Brd-k12343256-001-01-4
78. Acetamide, N-[3-[3-cyclopropyl-5-[(2-fluoro-4-iodophenyl)amino]-3,4,6,7-tetrahydro-6,8-dimethyl-2,4,7-trioxopyrido[4,3-d]pyrimidin-1(2h)-yl]phenyl]-
79. N-(3-(3-cyclopropyl-5-(2-fluoro-4-iodophenylamino)-6,8-dimethyl-2,4,7-trioxo-3,4,6,7-tetrahydropyrido[4,3-d]pyrimidin-1(2h)-yl)phenyl)acetamide
80. N-[3-[3-cyclopropyl-5-[(2-fluoro-4-iodophenyl)amino]-3,4,6,7-tetrahydro-6,8-dimethyl-2,4,7-trioxopyrido[4,3-d]pyrimidin-1(2h)-yl]phe Nyl]acetamide
81. N-[3-[3-cyclopropyl-5-[(2-fluoro-4-iodophenyl)amino]-6,8-dimethyl-2,4,7-trioxopyrido[3,4-e]pyrimidin-1-yl]phenyl]acetamide
82. N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodoanilino)-6,8-dimethyl-2,4,7-trioxo-3,4,6,7-tetrahydropyrido[4,3-d]pyrimidin-1(2h)-yl]phenyl}ethanimidic Acid
83. N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodophenylamino)-6,8-dimethyl-2,4,7-trioxo-3,4,6,7-tetrahydro-2h-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide
| Molecular Weight | 615.4 g/mol |
|---|---|
| Molecular Formula | C26H23FIN5O4 |
| XLogP3 | 3.4 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 5 |
| Exact Mass | 615.07788 g/mol |
| Monoisotopic Mass | 615.07788 g/mol |
| Topological Polar Surface Area | 102 Ų |
| Heavy Atom Count | 37 |
| Formal Charge | 0 |
| Complexity | 1090 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Mekinist |
| PubMed Health | Trametinib (By mouth) |
| Drug Classes | Antineoplastic Agent |
| Drug Label | Trametinib dimethyl sulfoxide is a kinase inhibitor. The chemical name is acetamide, N-[3-[3-cyclopropyl-5-[(2-fluoro-4- iodophenyl)amino]-3,4,6,7-tetrahydro-6,8-dimethyl- 2,4,7-trioxopyrido[4,3-d]pyrimidin-1(2H)-yl]phenyl]-, compound with 1,1-sul... |
| Active Ingredient | Trametinib dimethyl sulfoxide |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 1mg non-solvated parent; eq 0.5mg non-solvated parent; eq 2mg non-solvated parent |
| Market Status | Prescription |
| Company | Glaxosmithkline |
| 2 of 2 | |
|---|---|
| Drug Name | Mekinist |
| PubMed Health | Trametinib (By mouth) |
| Drug Classes | Antineoplastic Agent |
| Drug Label | Trametinib dimethyl sulfoxide is a kinase inhibitor. The chemical name is acetamide, N-[3-[3-cyclopropyl-5-[(2-fluoro-4- iodophenyl)amino]-3,4,6,7-tetrahydro-6,8-dimethyl- 2,4,7-trioxopyrido[4,3-d]pyrimidin-1(2H)-yl]phenyl]-, compound with 1,1-sul... |
| Active Ingredient | Trametinib dimethyl sulfoxide |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 1mg non-solvated parent; eq 0.5mg non-solvated parent; eq 2mg non-solvated parent |
| Market Status | Prescription |
| Company | Glaxosmithkline |
Trametinib is indicated for the treatment of unresectable or metastatic melanoma with BRAF V600E or V600K mutations, as detected by an FDA-approved test [FDA]. In May 2018, it was approved for use with [DB08912] for the treatment of treat anaplastic thyroid cancer caused by an abnormal BRAF V600E gene.
FDA Label
* Melanoma:
Trametinib as monotherapy or in combination with dabrafenib is indicated for the treatment of adult patients with unresectable or metastatic melanoma with a BRAF V600 mutation (see sections 4. 4 and 5. 1).
Trametinib monotherapy has not demonstrated clinical activity in patients who have progressed on a prior BRAF inhibitor therapy (see section 5. 1).
* Adjuvant treatment of melanoma:
Trametinib in combination with dabrafenib is indicated for the adjuvant treatment of adult patients with Stage III melanoma with a BRAF V600 mutation, following complete resection.
* Non-small cell lung cancer (NSCLC):
Trametinib in combination with dabrafenib is indicated for the treatment of adult patients with advanced non-small cell lung cancer with a BRAF V600 mutation.
Trametinib is an anticancer agent which causes apoptosis (or programmed cell death) and inhibits cell proliferation, which are both important in the treatment of malignancies.
Antineoplastic Agents
Substances that inhibit or prevent the proliferation of NEOPLASMS. (See all compounds classified as Antineoplastic Agents.)
Protein Kinase Inhibitors
Agents that inhibit PROTEIN KINASES. (See all compounds classified as Protein Kinase Inhibitors.)
L01EE01
L - Antineoplastic and immunomodulating agents
L01 - Antineoplastic agents
L01E - Protein kinase inhibitors
L01EE - Mitogen-activated protein kinase (mek) inhibitors
L01EE01 - Trametinib
Absorption
Trametinib is readily absorbed. When an oral administration of trametinib was given to patients with BRAF V600 mutation-positive melanoma, peak plasma concentration occurred 1.5 hours post-dose (Tmax). A single 2 mg oral dose has a bioavailability of 72%. When a dose of 2mg/day is given, the peak plasma concentration (Cmax) is 22.2 ng/mL.
Route of Elimination
80% of the dose is excreted in the feces. <20% of the dose is excreted in the urine with <0.1% of the excreted dose in the form of the parent compound.
Volume of Distribution
Apparent volume of distribution (Vd/F) = 214 L
Clearance
Apparent clearance = 4.9 L/h
Trametinib is metabolized predominantly via deacetylation alone or with mono-oxygenation or in combination with glucuronidation biotransformation pathways in vitro. Deacetylation is likely mediated by hydrolytic enzymes, such as carboxyl-esterases or amidases. The cytochrome P450 enzyme system is not involved with the metabolism of trametinib. The predominant circulating component in the plasma is the parent drug.
Elimination half-life = 3.9-4.8 days.
Trametinib is a reversible, allosteric inhibitor of mitogen-activated extracellular signal regulated kinase 1 _(MEK1)_ and _MEK2_ activation and of_ MEK1_ and _MEK2_ kinase activity. MEK proteins are upstream regulators of the extracellular signal-related kinase (ERK) pathway, which promotes cellular proliferation. Trametinib helps with melanoma with the BRAF V600E or V600K as the mutation results in the constitutive activation of the BRAF pathway which includes MEK1 and MEK2.
 European CDMO and Gx manufacturer with 75 years of experience in delivering premium APIs to pharmaceutical partners worldwide.
European CDMO and Gx manufacturer with 75 years of experience in delivering premium APIs to pharmaceutical partners worldwide.
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39561
Submission : 2024-06-19
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 40493
Submission : 2024-09-29
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2022-10-18
Pay. Date : 2022-09-12
DMF Number : 37504
Submission : 2022-09-13
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 43478
Submission : 2026-03-24
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2022-03-31
Pay. Date : 2022-01-06
DMF Number : 35525
Submission : 2020-12-31
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2026-04-09
Pay. Date : 2026-02-27
DMF Number : 31589
Submission : 2017-03-31
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 43552
Submission : 2026-03-21
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 43513
Submission : 2026-01-30
Status : Active
Type : II

USDMF
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 40250
Submission : 2024-08-23
Status : Active
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Details:
Trametinib is a small molecule drug, which is currently being evaluated in Phase II clinical studies for the treatment of vascular malformations.
Lead Product(s): Trametinib,Inapplicable
Therapeutic Area: Cardiology/Vascular Diseases Brand Name: Undisclosed
Study Phase: Phase IIProduct Type: Miscellaneous
Sponsor: Washington University School of Medicine
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable April 24, 2026

Lead Product(s) : Trametinib,Inapplicable
Therapeutic Area : Cardiology/Vascular Diseases
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : Washington University School of Medicine
Deal Size : Inapplicable
Deal Type : Inapplicable
24VA021; VATCH Trametinib for Ras/MAPK Pathway VAs
Details : Trametinib is a small molecule drug, which is currently being evaluated in Phase II clinical studies for the treatment of vascular malformations.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
April 24, 2026

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
JDQ443 (Opnurasib) is a small molecule drug, which is currently being evaluated in Phase I/ Phase II clinical studies for the treatment of carcinoma, non-small-cell lung.
Lead Product(s): JDQ443,TNO155,Trametinib,Cetuximab,Tislelizumab
Therapeutic Area: Oncology Brand Name: JDQ443
Study Phase: Phase I/ Phase IIProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable March 12, 2026

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : JDQ443,TNO155,Trametinib,Cetuximab,Tislelizumab
Therapeutic Area : Oncology
Highest Development Status : Phase I/ Phase II
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : JDQ443 (Opnurasib) is a small molecule drug, which is currently being evaluated in Phase I/ Phase II clinical studies for the treatment of carcinoma, non-small-cell lung.
Product Name : JDQ443
Product Type : Miscellaneous
Upfront Cash : Inapplicable
March 12, 2026

Details:
Tafinlar (Dabrafenib Mesylate) is a small molecule drug, which is currently being evaluated in Phase II/ Phase III clinical studies for the treatment of hematologic neoplasms.
Lead Product(s): Dabrafenib Mesylate,Trametinib
Therapeutic Area: Oncology Brand Name: Tafinlar
Study Phase: Phase II/ Phase IIIProduct Type: Miscellaneous
Sponsor: Novartis Pharmaceuticals Corporation
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable February 27, 2026

Lead Product(s) : Dabrafenib Mesylate,Trametinib
Therapeutic Area : Oncology
Highest Development Status : Phase II/ Phase III
Partner/Sponsor/Collaborator : Novartis Pharmaceuticals Corporation
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Tafinlar (Dabrafenib Mesylate) is a small molecule drug, which is currently being evaluated in Phase II/ Phase III clinical studies for the treatment of hematologic neoplasms.
Product Name : Tafinlar
Product Type : Miscellaneous
Upfront Cash : Inapplicable
February 27, 2026

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Through the acquisition, Hikma holds the rights of Mekinist-Generic (trametinib dimethyl sulfoxide) tablets from Novugen. It is being indicated for the treatment of BRAF-mutated melanoma.
Lead Product(s): Trametinib,Inapplicable
Therapeutic Area: Oncology Brand Name: Mekinist-Generic
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Hikma Pharmaceuticals
Deal Size: Undisclosed Upfront Cash: Undisclosed
Deal Type: Acquisition April 17, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Trametinib,Inapplicable
Therapeutic Area : Oncology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Hikma Pharmaceuticals
Deal Size : Undisclosed
Deal Type : Acquisition
Hikma acquires Novugen's FDA-approved ANDA for trametinib
Details : Through the acquisition, Hikma holds the rights of Mekinist-Generic (trametinib dimethyl sulfoxide) tablets from Novugen. It is being indicated for the treatment of BRAF-mutated melanoma.
Product Name : Mekinist-Generic
Product Type : Miscellaneous
Upfront Cash : Undisclosed
April 17, 2025

Details:
Trametinib is a Other Small Molecule drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Histiocytosis, Langerhans-Cell.
Lead Product(s): Trametinib,Inapplicable
Therapeutic Area: Rare Diseases and Disorders Brand Name: Undisclosed
Study Phase: Phase IIProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable September 03, 2024

Lead Product(s) : Trametinib,Inapplicable
Therapeutic Area : Rare Diseases and Disorders
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Targeted Approach to Langerhans Cell Histiocytosis (LCH) Using MEK Inhibitor, Trametinib
Details : Trametinib is a Other Small Molecule drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Histiocytosis, Langerhans-Cell.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
September 03, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
ERAS-254 (naporafenib) , an oral Pan-Raf inhibitor is being investigated with trametinib (MEK inhibitor) in patients with NRAS-mutant (NRASm) melanoma.
Lead Product(s): Naporafenib,Trametinib
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: Phase IIIProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable June 18, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Naporafenib,Trametinib
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Erasca Starts Phase 3 Trial for Naporafenib in Melanoma
Details : ERAS-254 (naporafenib) , an oral Pan-Raf inhibitor is being investigated with trametinib (MEK inhibitor) in patients with NRAS-mutant (NRASm) melanoma.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
June 18, 2024

Details:
Trametinib is a Other Small Molecule drug candidate, which is currently being evaluated in phase I/ phase II clinical studies for the treatment of Neoplasms.
Lead Product(s): Trametinib,Anlotinib Hydrochloride,Tislelizumab
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: Phase I/ Phase IIProduct Type: Miscellaneous
Sponsor: BeOne Medicines | Chia Tai Tianqing Pharmaceutical Group | Novartis Pharmaceuticals Corporation
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable June 13, 2024

Lead Product(s) : Trametinib,Anlotinib Hydrochloride,Tislelizumab
Therapeutic Area : Oncology
Highest Development Status : Phase I/ Phase II
Partner/Sponsor/Collaborator : BeOne Medicines | Chia Tai Tianqing Pharmaceutical Group | Novartis Pharmaceuticals Corporation
Deal Size : Inapplicable
Deal Type : Inapplicable
Trametinib Plus Anlotinib Combined With Tislelizumab in KRAS-mutant NSCLC
Details : Trametinib is a Other Small Molecule drug candidate, which is currently being evaluated in phase I/ phase II clinical studies for the treatment of Neoplasms.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
June 13, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
The net proceeds will be used to advance the clinical development of ERAS-254 (naporafenib), which is being evaluated in late-stage clinical trials for the treatment of RAS Q61X-mutated melanoma.
Lead Product(s): Naporafenib,Trametinib
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: Phase IProduct Type: Miscellaneous
Sponsor: J.P. Morgan
Deal Size: $184.0 million Upfront Cash: Undisclosed
Deal Type: Public Offering May 21, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Naporafenib,Trametinib
Therapeutic Area : Oncology
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : J.P. Morgan
Deal Size : $184.0 million
Deal Type : Public Offering
Erasca Announces Closing of Underwritten Offering of Common Stock and Additional Shares
Details : The net proceeds will be used to advance the clinical development of ERAS-254 (naporafenib), which is being evaluated in late-stage clinical trials for the treatment of RAS Q61X-mutated melanoma.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Undisclosed
May 21, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Naporafenib is a Other Small Molecule drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Melanoma.
Lead Product(s): Naporafenib,Trametinib
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: Phase IIIProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable April 03, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Naporafenib,Trametinib
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Naporafenib is a Other Small Molecule drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Melanoma.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
April 03, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Erasca will use the proceeds to fund R&D of its product candidates, including ERAS-254 (naporafenib) with trametinib for patients with RAS Q61X solid tumors, and other developmental programs.
Lead Product(s): Naporafenib,Trametinib
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: Phase IProduct Type: Miscellaneous
Sponsor: BofA Securities
Deal Size: $45.0 million Upfront Cash: Undisclosed
Deal Type: Private Placement March 27, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Naporafenib,Trametinib
Therapeutic Area : Oncology
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : BofA Securities
Deal Size : $45.0 million
Deal Type : Private Placement
Erasca Announces $45 Million Oversubscribed Private Placement Financing
Details : Erasca will use the proceeds to fund R&D of its product candidates, including ERAS-254 (naporafenib) with trametinib for patients with RAS Q61X solid tumors, and other developmental programs.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Undisclosed
March 27, 2024

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : DISCN
Registration Country : USA
Brand Name : TRAMETINIB DIMETHYL SULFOXIDE
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 0.5MG
Packaging :
Approval Date : 2024-08-06
Application Number : 219002
Regulatory Info : DISCN
Registration Country : USA

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : DISCN
Registration Country : USA
Brand Name : TRAMETINIB DIMETHYL SULFOXIDE
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 2MG
Packaging :
Approval Date : 2024-08-06
Application Number : 219002
Regulatory Info : DISCN
Registration Country : USA

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : RX
Registration Country : USA
Brand Name : MEKINIST
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 0.5MG
Packaging :
Approval Date : 2013-05-29
Application Number : 204114
Regulatory Info : RX
Registration Country : USA

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : DISCN
Registration Country : USA
Brand Name : MEKINIST
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 1MG **Federal Register determination that product was not discontinued or withdrawn for safety or effectiveness reasons**
Packaging :
Approval Date : 2013-05-29
Application Number : 204114
Regulatory Info : DISCN
Registration Country : USA

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : RX
Registration Country : USA
Brand Name : MEKINIST
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 2MG
Packaging :
Approval Date : 2013-05-29
Application Number : 204114
Regulatory Info : RX
Registration Country : USA

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : RX
Registration Country : USA
Brand Name : MEKINIST
Dosage Form : SOLUTION;ORAL
Dosage Strength : EQ 0.05MG BASE/ML
Packaging :
Approval Date : 2023-03-16
Application Number : 217513
Regulatory Info : RX
Registration Country : USA

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : USA
Brand Name : MEKINIST
Dosage Form : TABLET;ORAL
Dosage Strength : 2MG
Packaging :
Approval Date :
Application Number : 217513
Regulatory Info :
Registration Country : USA

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : USA
Brand Name : MEKINIST
Dosage Form : SOLUTION;ORAL
Dosage Strength : 4.7MG
Packaging :
Approval Date :
Application Number : 217513
Regulatory Info :
Registration Country : USA

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Allowed
Registration Country : Switzerland
Brand Name : Mekinist
Dosage Form : Film Coated Tablet
Dosage Strength : 0.5mg
Packaging :
Approval Date : 22/02/2016
Application Number : 65883
Regulatory Info : Allowed
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Allowed
Registration Country : Switzerland
Brand Name : Mekinist
Dosage Form : Film Coated Tablet
Dosage Strength : 2mg
Packaging :
Approval Date : 22/02/2016
Application Number : 65883
Regulatory Info : Allowed
Registration Country : Switzerland

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Allowed
Registration Country : Switzerland
Brand Name : Mekinist
Dosage Form : Film Coated Tablet
Dosage Strength : 0.5mg
Packaging :
Approval Date : 22/02/2016
Application Number : 65883
Regulatory Info : Allowed
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Allowed
Registration Country : Switzerland
Brand Name : Mekinist
Dosage Form : Film Coated Tablet
Dosage Strength : 2mg
Packaging :
Approval Date : 22/02/2016
Application Number : 65883
Regulatory Info : Allowed
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Marketed
Registration Country : Norway
Brand Name : Mekinist
Dosage Form : Film Coated Tablet
Dosage Strength : 2mg
Packaging :
Approval Date :
Application Number :
Regulatory Info : Marketed
Registration Country : Norway

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Marketed
Registration Country : Norway
Brand Name : Spexotras
Dosage Form : Powder For Oral Solution
Dosage Strength : 0.05mg/ml
Packaging :
Approval Date :
Application Number :
Regulatory Info : Marketed
Registration Country : Norway

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Prescription
Registration Country : Estonia
Brand Name : Mekinist
Dosage Form : Film-Coated Tablet
Dosage Strength : 0.5mg
Packaging :
Approval Date :
Application Number :
Regulatory Info : Prescription
Registration Country : Estonia

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Prescription
Registration Country : Estonia
Brand Name : Mekinist
Dosage Form : Film-Coated Tablet
Dosage Strength : 2mg
Packaging :
Approval Date :
Application Number :
Regulatory Info : Prescription
Registration Country : Estonia

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Authorized
Registration Country : Spain
Brand Name : Mekinist
Dosage Form : Film Coated Tablet
Dosage Strength : 2MG
Packaging :
Approval Date : 03-03-2025
Application Number : 114931006IP
Regulatory Info : Authorized
Registration Country : Spain

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Authorized
Registration Country : Spain
Brand Name : Mekinist
Dosage Form : Film Coated Tablet
Dosage Strength : 2MG
Packaging :
Approval Date : 24-09-2015
Application Number : 114931006
Regulatory Info : Authorized
Registration Country : Spain

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Prescription
Registration Country : Denmark
Brand Name : Mekinist
Dosage Form : Film Coated Tablet
Dosage Strength : 0.5mg
Packaging :
Approval Date : 30-06-2014
Application Number : 28105247413
Regulatory Info : Prescription
Registration Country : Denmark

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Approved
Registration Country : Sweden
Brand Name : Mekinist
Dosage Form : Film Coated Tablet
Dosage Strength : 0.5mg
Packaging :
Approval Date : 30-06-2014
Application Number : 2.01E+13
Regulatory Info : Approved
Registration Country : Sweden

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : PRESCRIPTION
Registration Country : Canada
Brand Name : MEKINIST
Dosage Form : TABLET
Dosage Strength : 0.5MG
Packaging : 30
Approval Date :
Application Number : 2409623
Regulatory Info : PRESCRIPTION
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : PRESCRIPTION
Registration Country : Canada
Brand Name : MEKINIST
Dosage Form : TABLET
Dosage Strength : 2MG
Packaging : 30
Approval Date :
Application Number : 2409658
Regulatory Info : PRESCRIPTION
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : PRESCRIPTION
Registration Country : Canada
Brand Name : MEKINIST
Dosage Form : POWDER FOR SOLUTION
Dosage Strength : 4.7MG
Packaging :
Approval Date :
Application Number : 2539993
Regulatory Info : PRESCRIPTION
Registration Country : Canada

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
86
PharmaCompass offers a list of Trametinib API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Trametinib manufacturer or Trametinib supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Trametinib manufacturer or Trametinib supplier.
A Trametinib Dimethyl Sulfoxide manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Trametinib Dimethyl Sulfoxide, including repackagers and relabelers. The FDA regulates Trametinib Dimethyl Sulfoxide manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Trametinib Dimethyl Sulfoxide API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Trametinib Dimethyl Sulfoxide manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Trametinib Dimethyl Sulfoxide supplier is an individual or a company that provides Trametinib Dimethyl Sulfoxide active pharmaceutical ingredient (API) or Trametinib Dimethyl Sulfoxide finished formulations upon request. The Trametinib Dimethyl Sulfoxide suppliers may include Trametinib Dimethyl Sulfoxide API manufacturers, exporters, distributors and traders.
click here to find a list of Trametinib Dimethyl Sulfoxide suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Trametinib Dimethyl Sulfoxide DMF (Drug Master File) is a document detailing the whole manufacturing process of Trametinib Dimethyl Sulfoxide active pharmaceutical ingredient (API) in detail. Different forms of Trametinib Dimethyl Sulfoxide DMFs exist exist since differing nations have different regulations, such as Trametinib Dimethyl Sulfoxide USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Trametinib Dimethyl Sulfoxide DMF submitted to regulatory agencies in the US is known as a USDMF. Trametinib Dimethyl Sulfoxide USDMF includes data on Trametinib Dimethyl Sulfoxide's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Trametinib Dimethyl Sulfoxide USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Trametinib Dimethyl Sulfoxide suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Trametinib Dimethyl Sulfoxide Drug Master File in Korea (Trametinib Dimethyl Sulfoxide KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Trametinib Dimethyl Sulfoxide. The MFDS reviews the Trametinib Dimethyl Sulfoxide KDMF as part of the drug registration process and uses the information provided in the Trametinib Dimethyl Sulfoxide KDMF to evaluate the safety and efficacy of the drug.
After submitting a Trametinib Dimethyl Sulfoxide KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Trametinib Dimethyl Sulfoxide API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Trametinib Dimethyl Sulfoxide suppliers with KDMF on PharmaCompass.
A Trametinib Dimethyl Sulfoxide written confirmation (Trametinib Dimethyl Sulfoxide WC) is an official document issued by a regulatory agency to a Trametinib Dimethyl Sulfoxide manufacturer, verifying that the manufacturing facility of a Trametinib Dimethyl Sulfoxide active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Trametinib Dimethyl Sulfoxide APIs or Trametinib Dimethyl Sulfoxide finished pharmaceutical products to another nation, regulatory agencies frequently require a Trametinib Dimethyl Sulfoxide WC (written confirmation) as part of the regulatory process.
click here to find a list of Trametinib Dimethyl Sulfoxide suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Trametinib Dimethyl Sulfoxide as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Trametinib Dimethyl Sulfoxide API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Trametinib Dimethyl Sulfoxide as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Trametinib Dimethyl Sulfoxide and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Trametinib Dimethyl Sulfoxide NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Trametinib Dimethyl Sulfoxide suppliers with NDC on PharmaCompass.
Trametinib Dimethyl Sulfoxide Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Trametinib Dimethyl Sulfoxide GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Trametinib Dimethyl Sulfoxide GMP manufacturer or Trametinib Dimethyl Sulfoxide GMP API supplier for your needs.
A Trametinib Dimethyl Sulfoxide CoA (Certificate of Analysis) is a formal document that attests to Trametinib Dimethyl Sulfoxide's compliance with Trametinib Dimethyl Sulfoxide specifications and serves as a tool for batch-level quality control.
Trametinib Dimethyl Sulfoxide CoA mostly includes findings from lab analyses of a specific batch. For each Trametinib Dimethyl Sulfoxide CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Trametinib Dimethyl Sulfoxide may be tested according to a variety of international standards, such as European Pharmacopoeia (Trametinib Dimethyl Sulfoxide EP), Trametinib Dimethyl Sulfoxide JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Trametinib Dimethyl Sulfoxide USP).

