
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
Australia
0
South Africa
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
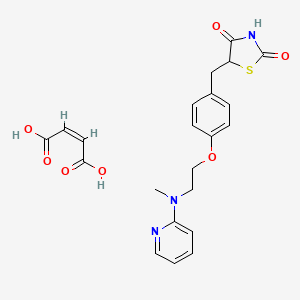

1. 5-((4-(2-methyl-2-(pyridinylamino)ethoxy)phenyl)methyl)-2,4-thiazolidinedione-2-butenedioate
2. Avandia
3. Brl 49653
4. Brl-49653
5. Brl49653
6. Rosiglitazone
1. 155141-29-0
2. Avandia
3. Rosiglitazone (maleate)
4. Rosiglitazone Maleate [usan]
5. Brl-49653c
6. Brl 49653c
7. Rosiglitzazone Maleate
8. 5-(4-(2-(methyl(pyridin-2-yl)amino)ethoxy)benzyl)thiazolidine-2,4-dione Maleate
9. Brl-49653-c
10. Nsc-717764
11. Kx2339dp44
12. Avandamet
13. 2,4-thiazolidinedione, 5-((4-(2-(methyl-2-pyridinylamino)ethoxy)phenyl)methyl)-, (2z)-2-butenedioate (1:1)
14. Nyracta
15. Rosiglitazone Xr
16. Venvia
17. Chebi:8892
18. Brl 49653-c
19. (2z)-but-2-enedioic Acid; 5-[(4-{2-[methyl(pyridin-2-yl)amino]ethoxy}phenyl)methyl]-1,3-thiazolidine-2,4-dione
20. (z)-but-2-enedioic Acid;5-[[4-[2-[methyl(pyridin-2-yl)amino]ethoxy]phenyl]methyl]-1,3-thiazolidine-2,4-dione
21. 5-[[4-[2-[methyl(pyridin-2-yl)amino]ethoxy]phenyl]methyl]-1,3-thiazolidine-2,4-dione Maleate
22. Smr000471612
23. Avandia (tn)
24. Unii-kx2339dp44
25. 5-[4-[2-(n-methyl-n-(2-pyridyl)amino)ethoxy]benzyl]thiazolidine-2,4-dione Maleate
26. 5-[4-[2-[n-methyl-n-(2-pyridyl)amino)ethoxy]benzyl]thiazolidine-2,4-dione Maleate
27. Rosiglitazonemaleate
28. 2,4-thiazolidinedione, 5-[[4-[2-(methyl-2-pyridinylamino)ethoxy]phenyl]methyl]-, (2z)-2-butenedioate (1:1)
29. (+-)-5-(p-(2-(methyl-2-pyridylamino)ethoxy)benzyl)-2,4-thiazolidinedione Maleate (1:1)
30. 1217260-35-9
31. 5-[(4-{2-[methyl(pyridin-2-yl)amino]ethoxy}phenyl)methyl]-1,3-thiazolidine-2,4-dione; But-2-enedioic Acid
32. Dsstox_cid_3569
33. Chembl843
34. Dsstox_rid_77086
35. Dsstox_gsid_23569
36. Schembl19023
37. Rosiglitazone Maleate- Bio-x
38. Mls001306472
39. Mls001401426
40. Dtxsid2023569
41. Rosiglitazone Maleate (jan/usp)
42. Hms2051b11
43. Hms2233i10
44. Hms3884h04
45. Rosiglitazone Maleate [mi]
46. Rosiglitazone Maleate [jan]
47. Tox21_302367
48. Ac-737
49. Mfcd03427306
50. Nsc717764
51. Rosiglitazone Maleate [vandf]
52. S2505
53. Rosiglitazone Maleate [mart.]
54. Akos015961534
55. Rosiglitazone Maleate [usp-rs]
56. Rosiglitazone Maleate [who-dd]
57. Ccg-100943
58. Ccg-208126
59. Cs-1692
60. Ks-5027
61. Nc00193
62. Nsc 717764
63. Ncgc00255233-01
64. 5-{[4-({2-[methyl(pyridin-2-yl)amino]ethyl}oxy)phenyl]methyl}-1,3-thiazolidine-2,4-dione (2z)-but-2-enedioate
65. Br164376
66. Hy-14600
67. Rosiglitazone Maleate [orange Book]
68. Cas-155141-29-0
69. Rosiglitazone Maleate [usp Monograph]
70. Sw197573-4
71. D00596
72. Rosiglitazone Maleate 100 Microg/ml In Methanol
73. 141r290
74. A809615
75. Q27888042
76. 2, 5-[[4-[2-(methyl-2-pyridinylamino) Ethoxy]phenyl]methyl]-, (2z)-2-butenedioate (1:1)
77. 5-[4-[2-[n-methyl-n-(2-pyridyl)amino]ethoxy]phenyl Methyl]thiazolidine-2,4-dione Maleate
78. 5-{4-[2-(methylpyridin-2-ylamino)ethoxy]benzyl}thiazolidine-2,4-dione Maleate
79. (+/-)-5-(p-(2-(methyl-2-pyridylamino)ethoxy)benzyl)-2,4-thiazolidinedione Maleate (1:1)
80. (z)-but-2-enedioate;2-[4-[(2,4-dioxo-1,3-thiazolidin-3-ium-5-yl)methyl]phenoxy]ethyl-methyl-pyridin-2-ylazanium
81. 5-((4-(2-(n-methyl-n-2-pyridylamino)ethoxy)phenyl)methyl)thiazolidine-2,4-dione Maleate
| Molecular Weight | 473.5 g/mol |
|---|---|
| Molecular Formula | C22H23N3O7S |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 9 |
| Exact Mass | 473.12567125 g/mol |
| Monoisotopic Mass | 473.12567125 g/mol |
| Topological Polar Surface Area | 171 Ų |
| Heavy Atom Count | 33 |
| Formal Charge | 0 |
| Complexity | 588 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 6 | |
|---|---|
| Drug Name | Avandaryl |
| PubMed Health | Rosiglitazone (By mouth) |
| Drug Classes | Antidiabetic |
| Active Ingredient | rosiglitazone maleate; Glimepiride |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 4mg; 2mg; 1mg; 8mg |
| Market Status | Prescription |
| Company | Sb Pharmco |
| 2 of 6 | |
|---|---|
| Drug Name | Avandia |
| Drug Label | AVANDIA (rosiglitazone maleate) is an oral antidiabetic agent which acts primarily by increasing insulin sensitivity. AVANDIA improves glycemic control while reducing circulating insulin levels. Rosiglitazone maleate is not chemically or functionally... |
| Active Ingredient | Rosiglitazone maleate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 4mg base; eq 2mg base; eq 8mg base |
| Market Status | Prescription |
| Company | Sb Pharmco |
| 3 of 6 | |
|---|---|
| Drug Name | Rosiglitazone maleate |
| PubMed Health | Rosiglitazone/Glimepiride (By mouth) |
| Drug Classes | Antidiabetic |
| Drug Label | AVANDIA (rosiglitazone maleate) is an oral antidiabetic agent which acts primarily by increasing insulin sensitivity. AVANDIA improves glycemic control while reducing circulating insulin levels. Rosiglitazone maleate is not chemically or functionally... |
| Active Ingredient | Rosiglitazone maleate |
| Dosage Form | Tablet |
| Route | oral; Oral |
| Strength | eq 4mg base; eq 2mg base; 8mg; 4mg; 2mg; eq 8mg base |
| Market Status | Tentative Approval; Prescription |
| Company | Mylan Pharma; Teva; Sandoz; Hikma Pharms; Roxane |
| 4 of 6 | |
|---|---|
| Drug Name | Avandaryl |
| PubMed Health | Rosiglitazone (By mouth) |
| Drug Classes | Antidiabetic |
| Active Ingredient | rosiglitazone maleate; Glimepiride |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 4mg; 2mg; 1mg; 8mg |
| Market Status | Prescription |
| Company | Sb Pharmco |
| 5 of 6 | |
|---|---|
| Drug Name | Avandia |
| Drug Label | AVANDIA (rosiglitazone maleate) is an oral antidiabetic agent which acts primarily by increasing insulin sensitivity. AVANDIA improves glycemic control while reducing circulating insulin levels. Rosiglitazone maleate is not chemically or functionally... |
| Active Ingredient | Rosiglitazone maleate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 4mg base; eq 2mg base; eq 8mg base |
| Market Status | Prescription |
| Company | Sb Pharmco |
| 6 of 6 | |
|---|---|
| Drug Name | Rosiglitazone maleate |
| PubMed Health | Rosiglitazone/Glimepiride (By mouth) |
| Drug Classes | Antidiabetic |
| Drug Label | AVANDIA (rosiglitazone maleate) is an oral antidiabetic agent which acts primarily by increasing insulin sensitivity. AVANDIA improves glycemic control while reducing circulating insulin levels. Rosiglitazone maleate is not chemically or functionally... |
| Active Ingredient | Rosiglitazone maleate |
| Dosage Form | Tablet |
| Route | oral; Oral |
| Strength | eq 4mg base; eq 2mg base; 8mg; 4mg; 2mg; eq 8mg base |
| Market Status | Tentative Approval; Prescription |
| Company | Mylan Pharma; Teva; Sandoz; Hikma Pharms; Roxane |
AVANDAMET is indicated in the treatment of type 2 diabetes mellitus patients, particularly overweight patients:
- who are unable to achieve sufficient glycaemic control at their maximally tolerated dose of oral metformin alone.
- in triple oral therapy with sulphonylurea in patients with insufficient glycaemic control despite dual oral therapy with their maximally tolerated dose of metformin and a sulphonylurea (see section 4. 4).
Rosiglitazone is indicated in the treatment of type 2 diabetes mellitus:
as monotherapy
-in patients (particularly overweight patients) inadequately controlled by diet and exercise for whom metformin is inappropriate because of contraindications or intolerance
as dual oral therapy in combination with
-metformin, in patients (particularly overweight patients) with insufficient glycaemic control despite maximal tolerated dose of monotherapy with metformin
-a sulphonylurea, only in patients who show intolerance to metformin or for whom metformin is contraindicated, with insufficient glycaemic control despite monotherapy with a sulphonylurea
as triple oral therapy in combination with
-metformin and a sulphonylurea, in patients (particularly overweight patients) with insufficient glycaemic control despite dual oral therapy (see section 4. 4).
Rosiglitazone is indicated as oral monotherapy in type 2 diabetes mellitus patients, particularly overweight patients, inadequately controlled by diet and exercise for whom metformin is inappropriate because of contraindications or intolerance.
Rosiglitazone is also indicated for oral combination treatment in type 2 diabetes mellitus patients with insufficient glycaemic control despite maximal tolerated dose of oral monotherapy with either metformin or a sulphonylurea:
- in combination with metformin particularly in overweight patients.
- in combination with a sulphonylurea only in patients who show intolerance to metformin or for whom metformin is contraindicated.
Rosiglitazone is indicated as oral monotherapy in type 2 diabetes mellitus patients, particularly overweight patients, inadequately controlled by diet and exercise for whom metformin is inappropriate because of contraindications or intolerance.
Rosiglitazone is also indicated for oral combination treatment in type 2 diabetes mellitus patients with insufficient glycaemic control despite maximal tolerated dose of oral monotherapy with either metformin or a sulphonylurea:
- in combination with metformin particularly in overweight patients.
- in combination with a sulphonylurea only in patients who show intolerance to metformin or for whom metformin is contraindicated.
Alzheimer's Disease
Hypoglycemic Agents
Substances which lower blood glucose levels. (See all compounds classified as Hypoglycemic Agents.)
A10BD03
A10BG02
A10BG02
A10BG02
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
92
PharmaCompass offers a list of Rosiglitazone Maleate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Rosiglitazone Maleate manufacturer or Rosiglitazone Maleate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Rosiglitazone Maleate manufacturer or Rosiglitazone Maleate supplier.
A Rosiglitazone Maleate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Rosiglitazone Maleate, including repackagers and relabelers. The FDA regulates Rosiglitazone Maleate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Rosiglitazone Maleate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Rosiglitazone Maleate manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Rosiglitazone Maleate supplier is an individual or a company that provides Rosiglitazone Maleate active pharmaceutical ingredient (API) or Rosiglitazone Maleate finished formulations upon request. The Rosiglitazone Maleate suppliers may include Rosiglitazone Maleate API manufacturers, exporters, distributors and traders.
click here to find a list of Rosiglitazone Maleate suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Rosiglitazone Maleate DMF (Drug Master File) is a document detailing the whole manufacturing process of Rosiglitazone Maleate active pharmaceutical ingredient (API) in detail. Different forms of Rosiglitazone Maleate DMFs exist exist since differing nations have different regulations, such as Rosiglitazone Maleate USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Rosiglitazone Maleate DMF submitted to regulatory agencies in the US is known as a USDMF. Rosiglitazone Maleate USDMF includes data on Rosiglitazone Maleate's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Rosiglitazone Maleate USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Rosiglitazone Maleate suppliers with USDMF on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Rosiglitazone Maleate as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Rosiglitazone Maleate API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Rosiglitazone Maleate as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Rosiglitazone Maleate and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Rosiglitazone Maleate NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Rosiglitazone Maleate suppliers with NDC on PharmaCompass.
Rosiglitazone Maleate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Rosiglitazone Maleate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Rosiglitazone Maleate GMP manufacturer or Rosiglitazone Maleate GMP API supplier for your needs.
A Rosiglitazone Maleate CoA (Certificate of Analysis) is a formal document that attests to Rosiglitazone Maleate's compliance with Rosiglitazone Maleate specifications and serves as a tool for batch-level quality control.
Rosiglitazone Maleate CoA mostly includes findings from lab analyses of a specific batch. For each Rosiglitazone Maleate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Rosiglitazone Maleate may be tested according to a variety of international standards, such as European Pharmacopoeia (Rosiglitazone Maleate EP), Rosiglitazone Maleate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Rosiglitazone Maleate USP).

