
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Australia
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
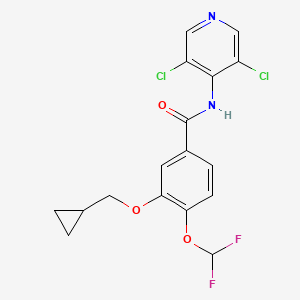

1. 3-cyclopropylmethoxy-4-difluoromethoxy-n-(3,5-di-chloropyrid-4-yl)benzamide
2. Daliresp
1. 162401-32-3
2. Daxas
3. 3-(cyclopropylmethoxy)-n-(3,5-dichloropyridin-4-yl)-4-(difluoromethoxy)benzamide
4. Daliresp
5. By217
6. Byk20869
7. By-217
8. B9302-107
9. By 217
10. 3-(cyclopropylmethoxy)-n-(3,5-dichloro-4-pyridinyl)-4-(difluoromethoxy)benzamide
11. Roflumilast (daxas)
12. Benzamide, 3-(cyclopropylmethoxy)-n-(3,5-dichloro-4-pyridinyl)-4-(difluoromethoxy)-
13. Byk-20869
14. 0p6c6zop5u
15. Chembl193240
16. Chebi:47657
17. 3-cyclopropylmethoxy-n-(3,5-dichloropyridin-4-yl)-4-(difluoromethoxy)benzamide
18. Byk 20869
19. Mfcd00938270
20. B 9302-107
21. Rof
22. Libertek
23. Roflumilast [usan]
24. B-9302-107
25. Roflumilast [usan:inn]
26. Unii-0p6c6zop5u
27. Roflumilastum
28. Roflumilast (jan/usan/inn)
29. Apta-2217
30. 1xmu
31. 1xoq
32. Daliresp (tn)
33. Roflumilast- Bio-x
34. 3g4l
35. Roflumilast [mi]
36. Roflumilast [inn]
37. Roflumilast [jan]
38. 3-cyclopropylmethoxy-4-difluoromethoxy-n-(3,5-di-chloropyrid-4-yl)benzamide
39. Roflumilast [vandf]
40. Roflumilast [mart.]
41. Schembl19158
42. Roflumilast [who-dd]
43. Mls006010074
44. Roflumilast [ema Epar]
45. Gtpl6962
46. Dtxsid8044123
47. Apta 2217
48. Bdbm14774
49. Roflumilast, >=98% (hplc)
50. Amy4219
51. Ex-a059
52. Roflumilast [orange Book]
53. Hms3655p21
54. Hms3748c19
55. Hms3884f09
56. Zinc592419
57. Act02619
58. Bcp03736
59. S2131
60. Arq-151/zoryve (roflumilast Cream)
61. Akos005146309
62. Am84550
63. Ccg-268678
64. Cs-0963
65. Db01656
66. Pb29130
67. Ncgc00346566-01
68. Ncgc00346566-09
69. Ac-23383
70. As-14120
71. Br164364
72. Hy-15455
73. Smr002530074
74. Sy008710
75. Bcp0726000146
76. Ft-0660846
77. R0193
78. Sw220196-1
79. A24672
80. D05744
81. Ab01565852_02
82. 401r323
83. Q693482
84. J-510858
85. Brd-k03194791-001-02-2
86. 3- Cyclo-propylmethoxy-4-difluoromethoxy-n- [3, 5-di-chloropyrid-4-yl]- Benzamid
87. Benzamide, 3-(cyclopropylmethoxy)-n-(3,5-dichloro-4-pyridinyl)-4-(difluoromethoxy)-
88. N-(3,5-dichloropyridin-4-yl)-4-difluoromethoxy-3-cyclopropylmethoxybenzamide
| Molecular Weight | 403.2 g/mol |
|---|---|
| Molecular Formula | C17H14Cl2F2N2O3 |
| XLogP3 | 4.6 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 7 |
| Exact Mass | 402.0349540 g/mol |
| Monoisotopic Mass | 402.0349540 g/mol |
| Topological Polar Surface Area | 60.4 Ų |
| Heavy Atom Count | 26 |
| Formal Charge | 0 |
| Complexity | 475 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Daliresp |
| PubMed Health | Roflumilast (By mouth) |
| Drug Classes | Respiratory Agent |
| Drug Label | The active ingredient in DALIRESP tablets is roflumilast. Roflumilast and its active metabolite (roflumilast N-oxide) are selective phosphodiesterase 4 (PDE4) inhibitors. The chemical name of roflumilast is N-(3,5-dichloropyridin-4-yl)-3-cyclopropylm... |
| Active Ingredient | Roflumilast |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 500mcg |
| Market Status | Prescription |
| Company | Forest Res Inst |
| 2 of 2 | |
|---|---|
| Drug Name | Daliresp |
| PubMed Health | Roflumilast (By mouth) |
| Drug Classes | Respiratory Agent |
| Drug Label | The active ingredient in DALIRESP tablets is roflumilast. Roflumilast and its active metabolite (roflumilast N-oxide) are selective phosphodiesterase 4 (PDE4) inhibitors. The chemical name of roflumilast is N-(3,5-dichloropyridin-4-yl)-3-cyclopropylm... |
| Active Ingredient | Roflumilast |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 500mcg |
| Market Status | Prescription |
| Company | Forest Res Inst |
Roflumilast is indicated as a treatment to reduce the risk of COPD exacerbations in patients with severe COPD associated with chronic bronchitis and a history of exacerbations. Roflumilast is not a bronchodilator and is not indicated for the relief of acute bronchospasm.
FDA Label
Daxas is indicated for maintenance treatment of severe chronic obstructive pulmonary disease (COPD) (FEV1 post-bronchodilator less than 50% predicted) associated with chronic bronchitis in adult patients with a history of frequent exacerbations as add-on to bronchodilator treatment.
Libertek is indicated for maintenance treatment of severe chronic obstructive pulmonary disease (COPD) (FEV1 post-bronchodilator less than 50% predicted) associated with chronic bronchitis in adult patients with a history of frequent exacerbations as add-on to bronchodilator treatment.
Daliresp is indicated for maintenance treatment of severe chronic obstructive pulmonary disease (COPD) (FEV1 post-bronchodilator less than 50% predicted) associated with chronic bronchitis in adult patients with a history of frequent exacerbations as add-on to bronchodilator treatment.
Chronic obstructive pulmonary disease
Roflumilast (and its active metabolite, roflumilast N-oxide) increases cyclic adenosine-3, 5-monophosphate (cAMP) in lung cells by inhibiting PDE4. Increased cAMP activates PKA, which inactivates transcription factors involved in inflammation. Romflumilast also decreases the amount of sputum neutrophils and eosinophils in COPD patients.
R03DX07
R03DX08
R03DX07
R03DX07
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
R - Respiratory system
R03 - Drugs for obstructive airway diseases
R03D - Other systemic drugs for obstructive airway diseases
R03DX - Other systemic drugs for obstructive airway diseases
R03DX07 - Roflumilast
Absorption
After a 500mcg dose, the bioavailability of roflumilast is about 80%. In the fasted state, maximum plasma concentrations are reached in 0.5 to 2 hours. While in the fed state, Cmax is reduced by 40%, Tmax is increased by one hour, and total absorption is unchanged.
Route of Elimination
Roflumilast is excreted 70% in the urine as roflumilast N-oxide.
Volume of Distribution
Roflumilast has a Vd of 2.9L/kg with a dose of 500mcg. Permeability of roflumilast across the blood-brain barrier appears to be poor in rat studies.
Clearance
~9.6 L/hour.
Roflumilast is metabolized to roflumilast N-oxide, the active metabolite of roflumilast in humans, by CYP3A4 and CYP1A2.
Plasma half-life of roflumilast is 17 hours and its metabolite is 30 hours (oral dose).
Roflumilast is a phosphodiesterase-4 (PDE-4) inhibitor which, due to its selective inhibition of the PDE4 isoenzyme, has potential antiinflammatory and antimodulatory effects in the pulmonary system. It is thought that the increased levels of intracellular cyclic AMP are responsible for the therapeutic actions of Roflumilast.
 Interquim comes from an international group of 50 companies active in the pharma, hospital, diagnostics, fine chemicals & feed sectors.
Interquim comes from an international group of 50 companies active in the pharma, hospital, diagnostics, fine chemicals & feed sectors.
GDUFA
DMF Review : Complete
Rev. Date : 2015-01-05
Pay. Date : 2014-07-23
DMF Number : 28471
Submission : 2014-07-31
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 27779
Submission : 2014-03-28
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2018-06-29
Pay. Date : 2018-06-04
DMF Number : 32851
Submission : 2018-06-01
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2015-01-21
Pay. Date : 2014-09-10
DMF Number : 28506
Submission : 2014-07-22
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2014-11-17
Pay. Date : 2014-08-14
DMF Number : 28516
Submission : 2014-08-27
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2014-08-27
Pay. Date : 2014-07-03
DMF Number : 27612
Submission : 2013-11-30
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2014-09-08
Pay. Date : 2014-03-13
DMF Number : 26764
Submission : 2012-12-27
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2021-04-21
Pay. Date : 2021-03-04
DMF Number : 35661
Submission : 2021-03-11
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2014-11-11
Pay. Date : 2014-08-04
DMF Number : 28440
Submission : 2014-09-04
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2015-01-05
Pay. Date : 2014-10-14
DMF Number : 28657
Submission : 2014-10-31
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
54
PharmaCompass offers a list of Roflumilast API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Roflumilast manufacturer or Roflumilast supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Roflumilast manufacturer or Roflumilast supplier.
A Roflumilast (Daxas) manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Roflumilast (Daxas), including repackagers and relabelers. The FDA regulates Roflumilast (Daxas) manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Roflumilast (Daxas) API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Roflumilast (Daxas) manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Roflumilast (Daxas) supplier is an individual or a company that provides Roflumilast (Daxas) active pharmaceutical ingredient (API) or Roflumilast (Daxas) finished formulations upon request. The Roflumilast (Daxas) suppliers may include Roflumilast (Daxas) API manufacturers, exporters, distributors and traders.
click here to find a list of Roflumilast (Daxas) suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Roflumilast (Daxas) DMF (Drug Master File) is a document detailing the whole manufacturing process of Roflumilast (Daxas) active pharmaceutical ingredient (API) in detail. Different forms of Roflumilast (Daxas) DMFs exist exist since differing nations have different regulations, such as Roflumilast (Daxas) USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Roflumilast (Daxas) DMF submitted to regulatory agencies in the US is known as a USDMF. Roflumilast (Daxas) USDMF includes data on Roflumilast (Daxas)'s chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Roflumilast (Daxas) USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Roflumilast (Daxas) suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Roflumilast (Daxas) Drug Master File in Korea (Roflumilast (Daxas) KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Roflumilast (Daxas). The MFDS reviews the Roflumilast (Daxas) KDMF as part of the drug registration process and uses the information provided in the Roflumilast (Daxas) KDMF to evaluate the safety and efficacy of the drug.
After submitting a Roflumilast (Daxas) KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Roflumilast (Daxas) API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Roflumilast (Daxas) suppliers with KDMF on PharmaCompass.
A Roflumilast (Daxas) written confirmation (Roflumilast (Daxas) WC) is an official document issued by a regulatory agency to a Roflumilast (Daxas) manufacturer, verifying that the manufacturing facility of a Roflumilast (Daxas) active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Roflumilast (Daxas) APIs or Roflumilast (Daxas) finished pharmaceutical products to another nation, regulatory agencies frequently require a Roflumilast (Daxas) WC (written confirmation) as part of the regulatory process.
click here to find a list of Roflumilast (Daxas) suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Roflumilast (Daxas) as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Roflumilast (Daxas) API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Roflumilast (Daxas) as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Roflumilast (Daxas) and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Roflumilast (Daxas) NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Roflumilast (Daxas) suppliers with NDC on PharmaCompass.
Roflumilast (Daxas) Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Roflumilast (Daxas) GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Roflumilast (Daxas) GMP manufacturer or Roflumilast (Daxas) GMP API supplier for your needs.
A Roflumilast (Daxas) CoA (Certificate of Analysis) is a formal document that attests to Roflumilast (Daxas)'s compliance with Roflumilast (Daxas) specifications and serves as a tool for batch-level quality control.
Roflumilast (Daxas) CoA mostly includes findings from lab analyses of a specific batch. For each Roflumilast (Daxas) CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Roflumilast (Daxas) may be tested according to a variety of international standards, such as European Pharmacopoeia (Roflumilast (Daxas) EP), Roflumilast (Daxas) JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Roflumilast (Daxas) USP).

