
Synopsis
Synopsis
0
KDMF
0
VMF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
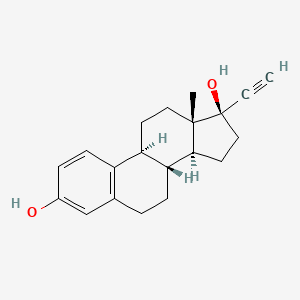

1. 19-norpregna-1,3,5(10)-trien-20-yne-3,17-diol, (17alpha)-
2. Estinyl
3. Estradiol, Ethinyl
4. Estradiol, Ethynyl
5. Ethinyl Estradiol Hemihydrate
6. Ethinyl Estradiol, (8 Alpha)-isomer
7. Ethinyl Estradiol, (8 Alpha,17 Alpha)-isomer
8. Ethinyl Estradiol, (8 Alpha,9 Beta,13 Alpha,14 Beta)-isomer
9. Ethinyl Estradiol, (9 Beta,17 Alpha)-isomer
10. Ethinyl Oestradiol Effik
11. Ethinyl-oestradiol Effik
12. Ethinylestradiol Jenapharm
13. Ethinyloestradiol
14. Ethynyl Estradiol
15. Hemihydrate, Ethinyl Estradiol
16. Jenapharm, Ethinylestradiol
17. Lynoral
18. Microfollin
19. Microfollin Forte
20. Progynon C
1. Ethynylestradiol
2. 57-63-6
3. Ethinylestradiol
4. Ethynyl Estradiol
5. Ethinyloestradiol
6. 17-ethinylestradiol
7. Estinyl
8. 17alpha-ethynylestradiol
9. Ginestrene
10. Lynoral
11. Progynon C
12. Ethinoral
13. Eticyclin
14. Eticyclol
15. Etinestrol
16. Etinestryl
17. Etinoestryl
18. Etistradiol
19. Follicoral
20. Novestrol
21. Orestralyn
22. Spanestrin
23. Amenoron
24. Dyloform
25. Ertonyl
26. Estigyn
27. Estoral
28. Estorals
29. Ethidol
30. Feminone
31. Inestra
32. Linoral
33. Menolyn
34. Oradiol
35. Primogyn
36. Esteed
37. Neo-estrone
38. Primogyn C
39. Primogyn M
40. Nogest-s
41. Eston-e
42. Palonyl
43. Perovex
44. 17alpha-ethinylestradiol
45. 17-ethynylestradiol
46. Diogyn E
47. Chee-o-genf
48. Chee-o-gen
49. 17-ethinyl-3,17-estradiol
50. Ethynyloestradiol
51. 17-ethinyl-3,17-oestradiol
52. 17-ethynyloestradiol
53. Estoral (orion)
54. 17alpha-ethinyl Estradiol
55. Diognat-e
56. Diogyn-e
57. Eticylol
58. Kolpolyn
59. Orestrayln
60. Ylestrol
61. Anovlar
62. Etivex
63. 17a-ethynylestradiol
64. Progynon M
65. Ethinylestradiolum
66. Estradiol, 17-ethynyl-
67. Nsc-10973
68. Ethinyl Estradiol [usp]
69. 17.alpha.-ethinylestradiol
70. 17.alpha.-ethynylestradiol
71. Estoral [orion]
72. 17.alpha.-ethynyloestradiol
73. Microfollin
74. 17a-ethinylestradiol
75. 17 Alpha-ethinyestradiol
76. Ethinyl Estradiol (usp)
77. Diprol
78. (8r,9s,13s,14s,17r)-17-ethynyl-13-methyl-7,8,9,11,12,13,14,15,16,17-decahydro-6h-cyclopenta[a]phenanthrene-3,17-diol
79. Ee
80. Chembl691
81. 17-alpha-ethynyl Estradiol
82. 17.alpha.-ethinyl-17.beta.-estradiol
83. Ethinyl-oestranol
84. 17.alpha.-ethynyl-17.beta.-oestradiol
85. Norinyl
86. Mls000028479
87. Aethinyloestradiolum
88. Chebi:4903
89. Ee2
90. Estopherol
91. Prosexol
92. Ethinyl E2
93. 17.alpha.-ethynylestradiol-l7.beta.
94. 423d2t571u
95. (8r,9s,13s,14s,17r)-17-ethynyl-13-methyl-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthrene-3,17-diol
96. 17.alpha.-ethynyloestradiol-17.beta.
97. Estoral (van)
98. (17beta)-17-ethynylestra-1(10),2,4-triene-3,17-diol
99. Aethinyoestradiol [german]
100. Ncgc00091533-04
101. Etinilestradiol
102. Certostat
103. Smr000058319
104. 19-norpregna-1,3,5(10)-trien-20-yne-3,17-diol, (17a)-
105. Etinilestradiolo
106. Dsstox_cid_576
107. Aethinyoestradiol
108. 19-norpregna-1,3,5(10)-trien-20-yne-3,17-diol, (17.alpha.)-
109. Etinilestradiol [inn-spanish]
110. Ee(sub 2)
111. Ethinylestradiolum [inn-latin]
112. Etinilestradiolo [dcit]
113. 17alpha-ethynyloestradiol
114. Dsstox_rid_75668
115. 17-alpha-ethynylestradiol
116. Dsstox_gsid_20576
117. 17alpha-ethynylestra-1,3,5(10)-triene-3,17beta-diol
118. 17alpha-ethinyl-estra-1,3,5(10)-triene-3,17beta-diol
119. 17-ethynyl-3,17-dihydroxy-1,3,5-oestratriene
120. 19-nor-17.alpha.-pregna-1,3,5(10)-trien-20-yne-3,17-diol
121. 17alpha-ethinylestradiol-17beta
122. Ethinylestradiol [inn:ban:jan]
123. 17-alpha-ethynylestradiol-17-beta
124. Cas-57-63-6
125. 17-alpha-ethinyl-17-beta-estradiol
126. (1s,10r,11s,14r,15s)-14-ethynyl-15-methyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadeca-2(7),3,5-triene-5,14-diol
127. 17-alpha-ethynyl-17-beta-oestradiol
128. Component Of Oracon
129. Component Of Ortrel
130. Ccris 286
131. Estinyl (tn)
132. Component Of Demulen
133. Hsdb 3587
134. Einecs 200-342-2
135. Mfcd00003690
136. Ovulen-21
137. Ovulen-28
138. Estra-1,5(10)-triene-3,17.beta.-diol, 17-ethynyl-
139. Estra-1,5[10]-triene-3,17.beta.-diol, 17-ethynyl-
140. Brn 2419975
141. Ethinyloestradiol [steroidal Oestrogens]
142. Ylestol
143. 19-nor-17.alpha.-pregna-1,5(10)-trien-20-yne-3,17-diol
144. 19-nor-17.alpha.-pregna-1,5[10]-trien-20-yne-3,17-diol
145. Ai3-52941
146. Ethinyl-estradiol
147. Ethynyl-estradiol
148. Unii-423d2t571u
149. 17alpha-ethinyl-17beta-estradiol
150. 19-norpregna-1,3,5(10)-trien-20-yne-3,17-diol, (17alpha)-
151. Component Of Ovral
152. 3wf
153. Levlen (salt/mix)
154. Oracon (salt/mix)
155. Ortrel (salt/mix)
156. 17-alpha-ethynyl-1,3,5-oestratriene-3,17-beta-diol
157. Anovlar (salt/mix)
158. Desogen (salt/mix)
159. 17a-ethynyloestradiol
160. Estoral {[orion]}
161. 17alpha-ethinylestra-1,3,5(10)-triene-3,17beta-diol
162. 17alpha-ethinyloestra-1,3,5(10)-triene-3,17beta-diol
163. Nordette (salt/mix)
164. Secrovin (salt/mix)
165. 17-alpha-ethinylestra-1,3,5(10)-triene-3,17-beta-diol
166. 17-alpha-ethynyl-1,3,5(10)-estratriene-3,17-beta-diol
167. Seasonale (salt/mix)
168. Triphasil (salt/mix)
169. 17-alpha-ethinyloestra-1,3,5(10)-triene-3,17-beta-diol
170. 17-alpha-ethynyl-1,3,5(10)-oestratriene-3,17-beta-diol
171. 17alpha-ethinyl-3,17-dihydroxy-delta(sup1,3,5)-estratriene
172. 17alpha-ethinyl-3,17-dihydroxy-delta(sup1,3,5)oestratriene
173. 3,17beta-dihydroxy-17alpha-ethynyl-1,3,5(10)-estratriene
174. 3,17beta-dihydroxy-17alpha-ethynyl-1,3,5(10)-oestratriene
175. Estopherol (salt/mix)
176. Estra-1,3,5(10)-triene-3,17beta-diol, 17alpha-ethynyl-
177. Seasonique (salt/mix)
178. 17-alpha-ethinyl-3,17-dihydroxy-delta(sup 1,3,5)-estratriene
179. 17-ethynylestradiol Ram
180. 19-nor-17alpha-pregna-1,3,5(10)-trien-20-yne-3,17-diol
181. 3,17-beta-dihydroxy-17-alpha-ethynyl-1,3,5(10)-estratriene
182. 3,17-beta-dihydroxy-17-alpha-ethynyl-1,3,5(10)-oestratriene
183. Estra-1,3,5(10)-triene-3,17-beta-diol, 17-alpha-ethynyl-
184. Ortho Evra (salt/mix)
185. Tri-levlen (salt/mix)
186. (17-alpha)-19-norpregna-1,3,5(10)-trien-20-yne-3,17,diol
187. 17alpha-ethinyl-delta(sup1,3,5(10))oestratriene-3,17-beta -diol
188. 17alpha-ethynyloestradiol-17beta; 17beta-estradiol, 17-ethynyl-
189. 19-nor-17-alpha-pregna-1,3,5(10)-triene-20-yne-3,17-diol
190. Opera_id_1808
191. 17a-ethynylestradiol-l7b
192. 17-alpha-ethinyl-delta(sup 1,3,5(10))oestratriene-3,17-beta-diol
193. Ortho-cyclen (salt/mix)
194. 17a-ethynyloestradiol-17b
195. Schembl4071
196. 17a-ethinyl-17b-estradiol
197. 17a-ethynyl-17b-oestradiol
198. Mls000758274
199. Mls001424011
200. Bidd:er0162
201. Ethinylestradiol [inn]
202. Ethinylestradiol [jan]
203. Ethinyl Estradiol [mi]
204. Ethinylestradiol (jp17/inn)
205. Ethinylestradiol [inci]
206. Gtpl7071
207. Sgcut00127
208. 17alpha-ethynyl-1,3,5(10)-estratriene-3,17beta-diol
209. Dtxsid5020576
210. Ethinyl Estradiol [hsdb]
211. Ethinylestradiol [mart.]
212. Ethinyl Estradiol [vandf]
213. Ethinylestradiol [who-dd]
214. Ethinylestradiol [who-ip]
215. Hms2051i19
216. Hms2235j09
217. Hms3715j09
218. 17alpha-ethynylestradiol, >=98%
219. Ethinyl Estradiol [usp-rs]
220. (17alpha)-19-norpregna-1,3,5(10)-trien-20-yne-3,17-diol
221. 17.beta.-estradiol, 17-ethynyl-
222. 19-nor-17-alpha-pregna-1,3,5(10)-trien-20-yne-3,17-diol
223. Hy-b0216
224. Nsc10973
225. To_000048
226. Zinc3812897
227. Tox21_111147
228. Tox21_201291
229. Tox21_300413
230. Bdbm50187243
231. Ethinyl Estradiol [ema Epar]
232. Lmst02010036
233. S1625
234. 17
235. A-ethynylestradiol;ethynylestradiol
236. Akos015894925
237. Tox21_111147_1
238. Yaz Component Ethinyl Estradiol
239. Ac-2169
240. Ccg-100819
241. Db00977
242. Ethinyl Estradiol [orange Book]
243. Ethinylestradiol [ep Monograph]
244. Ks-5257
245. Nc00069
246. 17.alpha.-ethinyl-3,3,5)oestratriene
247. Beyaz Component Ethinyl Estradiol
248. Ovral Component Ethinyl Estradiol
249. Zovia Component Ethinyl Estradiol
250. 17.alpha.-ethinyl-3,3,5)-estratriene
251. Alesse Component Ethinyl Estradiol
252. Aviane Component Ethinyl Estradiol
253. Ethinyl Estradiol [usp Monograph]
254. Ethinylestradiolum [who-ip Latin]
255. Femhrt Component Ethinyl Estradiol
256. Kariva Component Ethinyl Estradiol
257. Kelnor Component Ethinyl Estradiol
258. Levora Component Ethinyl Estradiol
259. Lybrel Component Ethinyl Estradiol
260. Ncgc00091533-01
261. Ncgc00091533-05
262. Ncgc00091533-07
263. Ncgc00091533-08
264. Ncgc00091533-09
265. Ncgc00091533-10
266. Ncgc00254514-01
267. Ncgc00258843-01
268. Portia Component Ethinyl Estradiol
269. Preven Component Ethinyl Estradiol
270. Twirla Component Ethinyl Estradiol
271. Vienva Component Ethinyl Estradiol
272. Volnea Component Ethinyl Estradiol
273. Xulane Component Ethinyl Estradiol
274. Yasmin Component Ethinyl Estradiol
275. Bekyree Component Ethinyl Estradiol
276. Desogen Component Ethinyl Estradiol
277. Enskyce Component Ethinyl Estradiol
278. Ethinyl Estradiol Component Of Yaz
279. Kurvelo Component Ethinyl Estradiol
280. Lessina Component Ethinyl Estradiol
281. Levlite Component Ethinyl Estradiol
282. Nci60_000234
283. Norinyl Component Ethinyl Estradiol
284. Pimtrea Component Ethinyl Estradiol
285. Safyral Component Ethinyl Estradiol
286. Trivora Component Ethinyl Estradiol
287. Velivet Component Ethinyl Estradiol
288. Viorele Component Ethinyl Estradiol
289. Vyfemla Component Ethinyl Estradiol
290. Altavera Component Ethinyl Estradiol
291. Annovera Component Ethinyl Estradiol
292. Aranelle Component Ethinyl Estradiol
293. Brevicon Component Ethinyl Estradiol
294. Cryselle Component Ethinyl Estradiol
295. Cyclessa Component Ethinyl Estradiol
296. Elifemme Component Ethinyl Estradiol
297. Enpresse Component Ethinyl Estradiol
298. Isibloom Component Ethinyl Estradiol
299. Levonest Component Ethinyl Estradiol
300. Marlissa Component Ethinyl Estradiol
301. Mircette Component Ethinyl Estradiol
302. Nordette Component Ethinyl Estradiol
303. Nuvaring Component Ethinyl Estradiol
304. Orsythia Component Ethinyl Estradiol
305. Previfem Component Ethinyl Estradiol
306. Quasense Component Ethinyl Estradiol
307. Setlakin Component Ethinyl Estradiol
308. Sprintec Component Ethinyl Estradiol
309. Taytulla Component Ethinyl Estradiol
310. Ethinyl Estradiol Component Of Beyaz
311. Ethinyl Estradiol Component Of Ovral
312. Introvale Component Ethinyl Estradiol
313. Quartette Component Ethinyl Estradiol
314. Seasonale Component Ethinyl Estradiol
315. Triphasil Component Ethinyl Estradiol
316. Ethinyl Estradiol Component Of Alesse
317. Ethinyl Estradiol Component Of Desogen
318. Ethinyl Estradiol Component Of Femhrt
319. Ethinyl Estradiol Component Of Kariva
320. Ethinyl Estradiol Component Of Kelnor
321. Ethinyl Estradiol Component Of Levlite
322. Ethinyl Estradiol Component Of Lybrel
323. Ethinyl Estradiol Component Of Preven
324. Ethinyl Estradiol Component Of Safyral
325. Ethinyl Estradiol Component Of Twirla
326. Ethinyl Estradiol Component Of Velivet
327. Ethinyl Estradiol Component Of Yasmin
328. Femcon Fe Component Ethinyl Estradiol
329. Ortho-cept Component Ethinyl Estradiol
330. Seasonique Component Ethinyl Estradiol
331. 17-ethynyl-3-17-dihydroxy-1,5-oestratriene
332. 17alpha-ethynylestradiol, >=98.0% (hplc)
333. Ethinyl Estradiol Component Of Altavera
334. Ethinyl Estradiol Component Of Annovera
335. Ethinyl Estradiol Component Of Aranelle
336. Ethinyl Estradiol Component Of Cryselle
337. Ethinyl Estradiol Component Of Cyclessa
338. Ethinyl Estradiol Component Of Levonest
339. Ethinyl Estradiol Component Of Mircette
340. Ethinyl Estradiol Component Of Nuvaring
341. Ethinyl Estradiol Component Of Previfem
342. Ethinyl Estradiol Component Of Quasense
343. Ethinyl Estradiol Component Of Setlakin
344. Ethinyl Estradiol Component Of Sprintec
345. Norquest Fe Component Ethinyl Estradiol
346. C07534
347. D00554
348. Estrostep Fe Component Ethinyl Estradiol
349. Ethinyl Estradiol Component Of Femcon Fe
350. Ethinyl Estradiol Component Of Introvale
351. Ethinyl Estradiol Component Of Seasonale
352. H11762
353. Loseasonique Component Ethinyl Estradiol
354. Tri-previfem Component Ethinyl Estradiol
355. Tri-sprintec Component Ethinyl Estradiol
356. 17-ethynyl-3-17-dihydroxy-1,3,5-oestratriene
357. Ab00441335-11
358. Ab00441335_12
359. Ethinyl Estradiol Component Of Ortho-cept
360. Ethinyl Estradiol Component Of Seasonique
361. Tri-legest Fe Component Ethinyl Estradiol
362. Wln: L E5 B666ttt&j E1 Fq F1uu1 Oq
363. 003e690
364. Ethinyl Estradiol Component Of Estrostep Fe
365. Ethinyl Estradiol Component Of Loseasonique
366. Ethinyl Estradiol Component Of Norquest Fe
367. Ethinyl Estradiol Component Of Tri-previfem
368. Ethinyl Estradiol Component Of Tri-sprintec
369. Lo Loestrin Fe Component Ethinyl Estradiol
370. Lo Minastrin Fe Component Ethinyl Estradiol
371. Q415563
372. Sr-01000721903
373. Tri Lo Sprintec Component Ethinyl Estradiol
374. 17-ethynylestra-1,3,5(10)-triene-3,17beta-diol
375. 17a-ethinylestradiol 100 Microg/ml In Acetonitrile
376. Ethinyl Estradiol Component Of Lo Loestrin Fe
377. Ethinyl Estradiol Component Of Tri-legest Fe
378. Minastrin 24 Fe Component Ethinyl Estradiol
379. Ortho Tri-cyclen Component Ethinyl Estradiol
380. Q-201076
381. Sr-01000721903-3
382. 17-ethynyloestra-1,5(10)-triene-3,17.beta.-diol
383. 17.alpha.-ethynyl-1,5-estratriene-3,17.beta.-diol
384. 17.alpha.-ethynyl-1,5-oestratriene-3,17.beta.-diol
385. 17a-ethinylestradiol 100 Microg/ml In Methanol/water
386. 17alpha-ethinyl-1,3,5(10)-estratriene-3,17-diol
387. Brd-k48195008-001-19-9
388. Ethinyl Estradiol Component Of Ortho Tri-cyclen
389. Ethinyl Estradiol Component Of Tri Lo Sprintec
390. 17-ethynyloestra-1,3,5(10)-triene-3,17.beta.-diol
391. 17.alpha.-ethinyl-1,3,5(10)-estratriene-3,17-diol
392. Z1541638513
393. 17.alpha.-ethinylestra-1,5(10)-triene-3,17.beta.-diol
394. 17.alpha.-ethinyloestra-1,5(10)-triene-3,17.beta.-diol
395. 17.alpha.-ethynylestra-1,5(10)-triene-3,17.beta.-diol
396. 17.alpha.-ethynyloestra-1,5(10)-triene-3,17.beta.-diol
397. 17.alpha.-ethinylestra-1,3,5(10)-triene-3,17.beta.-diol
398. 17.alpha.-ethinyloestra-1,3,5(10)-triene-3,17.beta.-diol
399. 17.alpha.-ethynyl-1,3,5(10)-estratriene-3,17.beta.-diol
400. 17.alpha.-ethynyl-1,3,5(10)-oestratriene-3,17.beta.-diol
401. 17.alpha.-ethynylestra-1,3,5(10)-triene-3,17.beta.-diol
402. 17.alpha.-ethynyloestra-1,3,5(10)-triene-3,17.beta.-diol
403. 17alpha-ethynylestradiol, Vetranal(tm), Analytical Standard
404. 19-nor-17.alpha.-pregna-1,5(10)-trien-2-yne-3,17-diol
405. Estra-1,5(10)-triene-3,17.beta.-diol, 17.alpha.-ethynyl-
406. 17.alpha.-ethinyl-3,17-dihydroxy-.delta.(sup1,3,5)-estratriene
407. 17.alpha.-ethinyl-3,17-dihydroxy-.delta.(sup1,3,5)oestratriene
408. 19-nor-17.alpha.-pregna-1,3,5(10)-trien-20-yn-3,17-diol
409. 19-norpregna-1,5(10)-trien-20-yne-3,17-diol, (17.alpha.)-
410. 3,17.beta.-dihydroxy-17.alpha.-ethynyl-1,3,5(10)-estratriene
411. 3,17.beta.-dihydroxy-17.alpha.-ethynyl-1,3,5(10)-oestratriene
412. Estra-1,3,5(10)-triene-3,17.beta.-diol, 17.alpha.-ethynyl-
413. Ethinylestradiol, European Pharmacopoeia (ep) Reference Standard
414. 17.alpha.-ethinyl-.delta.(sup1,3,5(10))oestratriene-3,17-.beta. -diol
415. 17.alpha.-ethinyl-.delta.(sup1,5(10))oestratriene-3,17-.beta.-diol
416. 19-nor-17.alpha.-pregna-1,3,5(10)-trien-20-yne-3,17.beta.-diol
417. Ethinyl Estradiol, United States Pharmacopeia (usp) Reference Standard
418. (9beta,13alpha,14beta,17alpha)-17-ethynylestra-1(10),2,4-triene-3,17-diol
419. Ethinyl Estradiol, Pharmaceutical Secondary Standard; Certified Reference Material
420. Ethinylestradiol For System Suitability, European Pharmacopoeia (ep) Reference Standard
421. 1050678-65-3
| Molecular Weight | 296.4 g/mol |
|---|---|
| Molecular Formula | C20H24O2 |
| XLogP3 | 3.7 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 1 |
| Exact Mass | 296.177630004 g/mol |
| Monoisotopic Mass | 296.177630004 g/mol |
| Topological Polar Surface Area | 40.5 Ų |
| Heavy Atom Count | 22 |
| Formal Charge | 0 |
| Complexity | 505 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 5 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Estrogens
National Library of Medicine's Medical Subject Headings online file (MeSH, 2009)
Ethinyl estradiol /is indicated in combination with an oral progestin/ for the prevention of pregnancy in women who elect to use this product as a method of contraception. /Included in US product label/
US Natl Inst Health; DailyMed. Current Medication Information CESIA (desogestrel and ethinyl estradiol) kit (February 2009). Available from, as of March 10, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=9373
Femhrt (norethindrone acetate/ethinyl estradiol) is indicated in women with an intact uterus for the treatment of moderate to severe vasomotor symptoms associated with the menopause. /Included in US product label/
US Natl Inst Health; DailyMed. Current Medication Information FEMHRT (norethindrone acetate/ethinyl estradiol) tablet (October 2008). Available from, as of March 10, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=23194
Femhrt (norethindrone acetate/ethinyl estradiol) is indicated in women with an intact uterus for the prevention of postmenopausal osteoporosis. When prescribing solely for the prevention of postmenopausal osteoporosis, therapy should only be considered for women at significant risk of osteoporosis. Non-estrogen medications should be carefully considered. /Included in US product label/
US Natl Inst Health; DailyMed. Current Medication Information FEMHRT (norethindrone acetate/ethinyl estradiol) tablet (October 2008). Available from, as of March 10, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=23194
Ethinyl estradiol is used frequently in combination with progestins for oral contraception.
Briggs, G.G, R.K. Freeman, S.J. Yaffe. A Reference Guide to Fetal and Neonatal Risk. Drugs in Pregnancy and Lactation. 4th ed. Baltimore, MD: Williams & Wilkins 1994., p. 354
/BOXED WARNING/ WARNING: Estrogens and progestins should not be used for the prevention of cardiovascular disease or dementia. The Women's Health Initiative (WHI) study reported increased risks of myocardial infarction, stroke, invasive breast cancer, pulmonary emboli, and deep vein thrombosis in postmenopausal women (50 to 79 years of age) during 5 years of treatment with oral conjugated estrogens (CE 0.625 mg) combined with medroxyprogesterone acetate (MPA 2.5 mg) relative to placebo. The Women's Health Initiative Memory Study (WHIMS), a substudy of WHI, reported increased risk of developing probable dementia in postmenopausal women 65 years of age or older during 4 years of treatment with oral conjugated estrogens plus medroxyprogesterone acetate relative to placebo. It is unknown whether this finding applies to younger postmenopausal women. Other doses of oral conjugated estrogens with medroxyprogesterone acetate, and other combinations and dosage forms of estrogens and progestins were not studied in the WHI clinical trials and, in the absence of comparable data, these risks should be assumed to be similar. Because of these risks, estrogens with or without progestins should be prescribed at the lowest effective doses and for the shortest duration consistent with treatment goals and risks for the individual woman.
US Natl Inst Health; DailyMed. Current Medication Information FEMHRT (norethindrone acetate/ethinyl estradiol) tablet (October 2008). Available from, as of March 10, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=23194
ESTROGENS INCREASE THE RISK OF ENDOMETRIAL CANCER: Close clinical surveillance of all women taking estrogens is important. Adequate diagnostic measures, including endometrial sampling when indicated, should be undertaken to rule out malignancy in all cases of undiagnosed persistent or recurring abnormal vaginal bleeding. There is no evidence that the use of "natural" estrogens results in a different endometrial risk profile than synthetic estrogens at equivalent estrogen doses.
US Natl Inst Health; DailyMed. Current Medication Information FEMHRT (norethindrone acetate/ethinyl estradiol) tablet (October 2008). Available from, as of March 10, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=23194
Other doses of oral conjugated estrogens with medroxyprogesterone acetate, and other combinations and dosage forms of estrogens and progestins were not studied in the WHI clinical trials and, in the absence of comparable data, these risks should be assumed to be similar. Because of these risks, estrogens with or without progestins should be prescribed at the lowest effective doses and for the shortest duration consistent with treatment goals and risks for the individual woman.
US Natl Inst Health; DailyMed. Current Medication Information FEMHRT (norethindrone acetate/ethinyl estradiol) tablet (October 2008). Available from, as of March 10, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=23194
FDA Pregnancy Risk Category: X /CONTRAINDICATED IN PREGNANCY. Studies in animals and or humans, or investigational or post-marketing reports, have demonstrated positive evidence of fetal abnormalities or risk which clearly outweighs any possible benefit to the patient./
US Natl Inst Health; DailyMed. Current Medication Information FEMHRT (norethindrone acetate/ethinyl estradiol) tablet (October 2008). Available from, as of March 10, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=23194
For more Drug Warnings (Complete) data for ETHINYLESTRADIOL (38 total), please visit the HSDB record page.
Ethinylestradiol is combined with other drugs for use as a contraceptive, premenstrual dysphoric disorder, moderate acne, moderate to severe vasomotor symptoms of menopause, prevention of postmenopausal osteoporosis.
FDA Label
Ethinylestradiol is a synthetic estrogen that decreases luteinizing hormone to decrease endometrial vascularization, and decreases gonadotrophic hormone to prevent ovulation. It has a long duration of action as it is taken once daily, and a wide therapeutic index as overdoses are generally not associated with serious adverse effects. Patients should be counselled regarding the risks of thrombotic events.
Contraceptives, Oral, Hormonal
Oral contraceptives which owe their effectiveness to hormonal preparations. (See all compounds classified as Contraceptives, Oral, Hormonal.)
Estrogens
Compounds that interact with ESTROGEN RECEPTORS in target tissues to bring about the effects similar to those of ESTRADIOL. Estrogens stimulate the female reproductive organs, and the development of secondary female SEX CHARACTERISTICS. Estrogenic chemicals include natural, synthetic, steroidal, or non-steroidal compounds. (See all compounds classified as Estrogens.)
G03AA07
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
G - Genito urinary system and sex hormones
G03 - Sex hormones and modulators of the genital system
G03C - Estrogens
G03CA - Natural and semisynthetic estrogens, plain
G03CA01 - Ethinylestradiol
L - Antineoplastic and immunomodulating agents
L02 - Endocrine therapy
L02A - Hormones and related agents
L02AA - Estrogens
L02AA03 - Ethinylestradiol
Absorption
A 30g oral dose of ethinylestradiol reaches a Cmax of 74.135.6pg/mL, with a Tmax of 1.50.5h, and an AUC of 487.4166.6pg\*h/mL. A 1.2mg dose delivered via a patch reaches a Cmax of 28.810.3pg/mL, with a Tmax of 8631h, and an AUC of38951423pg\*h/mL.
Route of Elimination
Ethinylestradiol is 59.2% eliminated in the urine and bile, while 2-3% is eliminated in the feces. Over 90% of ethinylestradiol is eliminated as the unchanged parent drug.
Volume of Distribution
A 30g oral dose has an apparent volume of distribution of 625.3228.7L and a 1.2mg topical dose has an apparent volume of distribution of 11745.315934.8L.
Clearance
Ethinylestradiol has an intravenous clearance of 16.47L/h, and an estimated renal clearance of approximately 2.1L/h. A 30g oral dose has a clearance of 58.019.8L/h and a 1.2mg topical dose has a clearance of 303.5100.5L/h.
Ethinyl estradiol is rapidly and almost completely absorbed. When the lowest and highest tablet strengths, 0.100 mg desogestrel/0.025 mg ethinyl estradiol and 0.150 mg desogestrel/0.025 mg ethinyl estradiol, were compared to solution, the relative bioavailability of ethinyl estradiol was 92% and 98%, respectively.
US Natl Inst Health; DailyMed. Current Medication Information CESIA (desogestrel and ethinyl estradiol) kit (February 2009). Available from, as of March 10, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=9373
The distribution of exogenous estrogens is similar to that of endogenous estrogens. Estrogens are widely distributed in the body and are generally found in higher concentrations in the sex hormone target organs. Ethinyl estradiol circulates in the blood largely bound to ... albumin. ... Although ethinyl estradiol does not bind to SHBG, it induces SHBG synthesis.
US Natl Inst Health; DailyMed. Current Medication Information FEMHRT (norethindrone acetate/ethinyl estradiol) tablet (October 2008). Available from, as of March 10, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=23194
Estradiol, estrone, and estriol are excreted in the urine along with glucuronide and sulfate conjugates.
US Natl Inst Health; DailyMed. Current Medication Information FEMHRT (norethindrone acetate/ethinyl estradiol) tablet (October 2008). Available from, as of March 10, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=23194
25 healthy women of reproductive age who had not previously used oral contraceptive steroids, were each given a single tablet containing 50, 80, or 100 ug of mestranol or 50 or 80 ug of ethinyl estradiol. Blood samples were obtained before taking the tablets and at intervals of 1, 2, 4, and 24 hours afterward. Anti-ethinyl-estradiol antibody in 1 to 100,000 initial dilution was used. Details of techniques employed are given. With ethinyl estradiol, the 1-hour sampling yielded the maximum plasma levels. At 24 hours, the plasma level was not detectable in 4 of 5 subjects given 50 ug or in 1 of 5 given 80 ug. With mestranol, the disappearance curve was more variable with the peak levels usually at 2 hours but occasionally at 4 hours. At all 3 dose levels of mestranol, measurable serum ethinyl estradiol levels were found at 24 hours. These levels were reached more slowly and were lower than when ethinyl estradiol was given. In contrast to natural estrogens ethinyl, estrogens are bound to plasma proteins chiefly by nonspecific binding and are therefore less likely to affect the metabolism of the ethinyl estrogens than are the endogenous steroids. Also, significant amounts of ethinyl estradiol are was given. In contrast to natural estrogens ethinyl, estrogens are bound di-ethynylated in vitro. The pharmacokinetics of ethinyl estrogens differ from those of natural estrogens. This complicates interpretation of plasma or urinary estrone and estradiol measurements.
PMID:1154455 de la Pena A et al; Steroids 25 (6): 773-80 (1975)
For more Absorption, Distribution and Excretion (Complete) data for ETHINYLESTRADIOL (7 total), please visit the HSDB record page.
Ethinylestradiol can be glucuronidated by UGT1A1, UGT1A3, UGT1A4, UGT1A9, and UGT2B7. Ethinylestradiol is also sulfated by SULT1A1, SULT1A3, and SULT1E1. Ethinylestradiol can also be hydroxylated at positions 2, 4, 6, 7, and 16 by CYP3A4, CYP3A5, CYP2C8, CYP2C9, and CYP1A2. These hydroxylated metabolites can be methylated by catechol-O-methyltransferase. The methoxy metabolites can in turn be sulfated or glucuronidated.
Exogenous estrogens are metabolized in the same manner as endogenous estrogens. Circulating estrogens exist in a dynamic equilibrium of metabolic interconversions. These transformations take place mainly in the liver. Estradiol is converted reversibly to estrone, and both can be converted to estriol, which is the major urinary metabolite. Estrogens also undergo enterohepatic recirculation via sulfate and glucuronide conjugation in the liver, biliary secretion of conjugates into the intestine, and hydrolysis in the gut followed by reabsorption. In postmenopausal women, a significant proportion of the circulating estrogens exist as sulfate conjugates, especially estrone sulfate, which serves as a circulating reservoir for the formation of more active estrogens.
US Natl Inst Health; DailyMed. Current Medication Information FEMHRT (norethindrone acetate/ethinyl estradiol) tablet (October 2008). Available from, as of March 10, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=23194
Ethinyl estradiol is extensively metabolized, both by oxidation and by conjugation with sulfate and glucuronide. Sulfates are the major circulating conjugates of ethinyl estradiol and glucuronides predominate in urine. The primary oxidative metabolite is 2-hydroxy ethinyl estradiol, formed by the CYP3A4 isoform of cytochrome P450. Part of the first-pass metabolism of ethinyl estradiol is believed to occur in gastrointestinal mucosa. Ethinyl estradiol may undergo enterohepatic circulation.
US Natl Inst Health; DailyMed. Current Medication Information FEMHRT (norethindrone acetate/ethinyl estradiol) tablet (October 2008). Available from, as of March 10, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=23194
Ethinyl estradiol is cleared much more slowly ... due to decreased hepatic metabolism.
Hardman, J.G., L.E. Limbird, P.B. Molinoff, R.W. Ruddon, A.G. Goodman (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 9th ed. New York, NY: McGraw-Hill, 1996., p. 1420
Studies on the metabolism of ethinylestradiol have been carried out in rats, rabbits, guinea-pigs, dogs and monkeys. It is very rapidly and effectively absorbed from rat intestine; no appreciable metabolic transformation is reported to take place during the absorption process. The main metabolic pathway of ethinylestradiol in rats is by aromatic 2-hydroxylation; hydroxylations at ring B (C-6/C-7) are of only minor importance. Rat liver forms 2-hydroxyethinylestradiol and the methyl ethers thereof, 2-methoxyethinylestradiol and 2-hydroxyethinylestradiol-3-methy1 ether, as its major metabolic products. This pathway is also important in humans. Metabolites of ethinylestradiol in rats are excreted almost exclusively in the feces.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V21 245 (1979)
For more Metabolism/Metabolites (Complete) data for ETHINYLESTRADIOL (10 total), please visit the HSDB record page.
Ethinylestradiol has known human metabolites that include 17-Ethynyl-13-methyl-7,8,9,11,12,14,15,16-octahydro-6H-cyclopenta[a]phenanthrene-2,3,17-triol and 17-Ethynyl-13-methyl-7,8,9,11,12,14,15,16-octahydro-6H-cyclopenta[a]phenanthrene-3,4,17-triol.
Ethinylestradiol is a known human metabolite of Mestranol.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
A 30g oral dose has a half life of 8.44.8h and a 1.2mg topical dose has a half life of 27.734.2h.
The pharmacokinetics of 19-nor-17 alpha-pregna-1,3,5(10)-trien-20-yne-3,17-diol (ethinylestradiol, Progynon C) (EE2) has been studied after intravenous administration of 0.1 or 0.01 mg/kg and after intragastric administration of 1 mg/kg in female rats, rabbits, beagle dogs, rhesus monkeys and baboons. After intravenous administration disposition of unchanged drug in the plasma was biphasic with initial half-lives between 0.3 and 0.5 hr and terminal half-lives between 2.3 and 3.0 hr. Total plasma clearance was of the same magnitude as total plasma liver flow or even higher rat) indicating a rapid biotransformation of the estrogen in the liver. Systemic availability of intragastric EE2 amounted to 3% in the rat, 0.3% in the rabbit, 9% in the dog, 0.6% in rhesus monkeys and 2% in the baboon and was considerably lower than in humans (40%). Differences in the pharmacokinetics and in the systemic availability of EE2 between laboratory animals and man should be taken into account in the retrospective interpretation of pharmacological and toxicological data and in the design of new studies.
PMID:3778555 Dusterberg B et al; Arzneimittelforschung 36 (8): 1187-90 (1986)
... The elimination phase half-life has been reported ... to be 13 to 27 hours.
Hardman, J.G., L.E. Limbird, P.B. Molinoff, R.W. Ruddon, A.G. Goodman (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 9th ed. New York, NY: McGraw-Hill, 1996., p. 1420
Ethinylestradiol is a synthetic estrogenic compound. Use of estrogens have a number of effects on the body including reduced bone density. Combined oral contraceptives suppress ovulation by suppressing gonadotrophic hormone, thickening cervical mucus to prevent the travel of sperm, and preventing changes in the endometrium required for implantation of a fertilized egg. Ethinylestradiol decreases luteinizing hormone, decreasing vascularity in the endometrium. It also increases sex hormone binding globulin.
Endogenous estrogens are largely responsible for the development and maintenance of the female reproductive system and secondary sexual characteristics. Although circulating estrogens exist in a dynamic equilibrium of metabolic interconversions, estradiol is the principal intracellular human estrogen and is substantially more potent than its metabolites estrone and estriol at the receptor level. ... After menopause, most endogenous estrogen is produced by conversion of androstenedione, secreted by the adrenal cortex, to estrone by peripheral tissues. Thus, estrone and the sulfate conjugated form, estrone sulfate, are the most abundant circulating estrogens in postmenopausal women. The pharmacologic effects of ethinyl estradiol are similar to those of endogenous estrogens. Estrogens act through binding to nuclear receptors in estrogen-responsive tissues. To date, two estrogen receptors have been identified. These vary in proportion from tissue to tissue. Circulating estrogens modulate the pituitary secretion of the gonadotropins, luteinizing hormone (LH) and follicle stimulating hormone (FSH) through a negative feedback mechanism. Estrogens act to reduce the elevated levels of these hormones seen in postmenopausal women.
US Natl Inst Health; DailyMed. Current Medication Information FEMHRT (norethindrone acetate/ethinyl estradiol) tablet (October 2008). Available from, as of March 10, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=23194
Estrogens have an important role in the reproductive, skeletal, cardiovascular, and central nervous systems in women, and act principally by regulating gene expression. Biologic response is initiated when estrogen binds to a ligand-binding domain of the estrogen receptor resulting in a conformational change that leads to gene transcription through specific estrogen response elements (ERE) of target gene promoters; subsequent activation or repression of the target gene is mediated through 2 distinct transactivation domains (ie, AF-1 and AF-2) of the receptor. The estrogen receptor also mediates gene transcription using different response elements (ie, AP-1) and other signal pathways. Recent advances in the molecular pharmacology of estrogen and estrogen receptors have resulted in the development of selective estrogen receptor modulators (eg, clomiphene, raloxifene, tamoxifen, toremifene), agents that bind and activate the estrogen receptor but that exhibit tissue-specific effects distinct from estrogen. Tissue-specific estrogen-agonist or -antagonist activity of these drugs appears to be related to structural differences in their estrogen receptor complex (eg, specifically the surface topography of AF-2 for raloxifene) compared with the estrogen (estradiol)-estrogen receptor complex. A second estrogen receptor also has been identified, and existence of at least 2 estrogen receptors (ER-alpha, ER-beta) may contribute to the tissue-specific activity of selective modulators. While the role of the estrogen receptor in bone, cardiovascular tissue, and the CNS continues to be studied, emerging evidence indicates that the mechanism of action of estrogen receptors in these tissues differs from the manner in which estrogen receptors function in reproductive tissue. /Estrogen General Statement/
American Society of Health System Pharmacists; AHFS Drug Information 2010. Bethesda, MD. (2010), p. 3130
Intracellular cytosol-binding proteins for estrogens have been identified in estrogen-responsive tissues including the female genital organs, breasts, pituitary, and hypothalamus. The estrogen-binding protein complex (ie, cytosol-binding protein and estrogen) distributes into the cell nucleus where it stimulates DNA, RNA, and protein synthesis. The presence of these receptor proteins is responsible for the palliative response to estrogen therapy in women with metastatic carcinoma of the breast. /Estrogen General Statement/
American Society of Health System Pharmacists; AHFS Drug Information 2010. Bethesda, MD. (2010), p. 3130
Estrogens have generally favorable effects on blood cholesterol and phospholipid concentrations. Estrogens reduce LDL-cholesterol and increase HDL-cholesterol concentrations in a dose-related manner. The decrease in LDL-cholesterol concentrations associated with estrogen therapy appears to result from increased LDL catabolism, while the increase in triglyceride concentrations is caused by increased production of large, triglyceride-rich, very-low-density lipoproteins (VLDLs); changes in serum HDL-cholesterol concentrations appear to result principally from an increase in the cholesterol and apolipoprotein A-1 content of HDL2- and a slight increase in HDL3-cholesterol. /Estrogen General Statement/
American Society of Health System Pharmacists; AHFS Drug Information 2010. Bethesda, MD. (2010), p. 3130
For more Mechanism of Action (Complete) data for ETHINYLESTRADIOL (7 total), please visit the HSDB record page.
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
16
PharmaCompass offers a list of Ethinyl Estradiol API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ethinyl Estradiol manufacturer or Ethinyl Estradiol supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Ethinyl Estradiol manufacturer or Ethinyl Estradiol supplier.
A PREVIFEM manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of PREVIFEM, including repackagers and relabelers. The FDA regulates PREVIFEM manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. PREVIFEM API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of PREVIFEM manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A PREVIFEM supplier is an individual or a company that provides PREVIFEM active pharmaceutical ingredient (API) or PREVIFEM finished formulations upon request. The PREVIFEM suppliers may include PREVIFEM API manufacturers, exporters, distributors and traders.
click here to find a list of PREVIFEM suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A PREVIFEM DMF (Drug Master File) is a document detailing the whole manufacturing process of PREVIFEM active pharmaceutical ingredient (API) in detail. Different forms of PREVIFEM DMFs exist exist since differing nations have different regulations, such as PREVIFEM USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A PREVIFEM DMF submitted to regulatory agencies in the US is known as a USDMF. PREVIFEM USDMF includes data on PREVIFEM's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The PREVIFEM USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of PREVIFEM suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The PREVIFEM Drug Master File in Japan (PREVIFEM JDMF) empowers PREVIFEM API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the PREVIFEM JDMF during the approval evaluation for pharmaceutical products. At the time of PREVIFEM JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of PREVIFEM suppliers with JDMF on PharmaCompass.
A PREVIFEM CEP of the European Pharmacopoeia monograph is often referred to as a PREVIFEM Certificate of Suitability (COS). The purpose of a PREVIFEM CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of PREVIFEM EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of PREVIFEM to their clients by showing that a PREVIFEM CEP has been issued for it. The manufacturer submits a PREVIFEM CEP (COS) as part of the market authorization procedure, and it takes on the role of a PREVIFEM CEP holder for the record. Additionally, the data presented in the PREVIFEM CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the PREVIFEM DMF.
A PREVIFEM CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. PREVIFEM CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of PREVIFEM suppliers with CEP (COS) on PharmaCompass.
A PREVIFEM written confirmation (PREVIFEM WC) is an official document issued by a regulatory agency to a PREVIFEM manufacturer, verifying that the manufacturing facility of a PREVIFEM active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting PREVIFEM APIs or PREVIFEM finished pharmaceutical products to another nation, regulatory agencies frequently require a PREVIFEM WC (written confirmation) as part of the regulatory process.
click here to find a list of PREVIFEM suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing PREVIFEM as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for PREVIFEM API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture PREVIFEM as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain PREVIFEM and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a PREVIFEM NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of PREVIFEM suppliers with NDC on PharmaCompass.
PREVIFEM Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of PREVIFEM GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right PREVIFEM GMP manufacturer or PREVIFEM GMP API supplier for your needs.
A PREVIFEM CoA (Certificate of Analysis) is a formal document that attests to PREVIFEM's compliance with PREVIFEM specifications and serves as a tool for batch-level quality control.
PREVIFEM CoA mostly includes findings from lab analyses of a specific batch. For each PREVIFEM CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
PREVIFEM may be tested according to a variety of international standards, such as European Pharmacopoeia (PREVIFEM EP), PREVIFEM JP (Japanese Pharmacopeia) and the US Pharmacopoeia (PREVIFEM USP).

