
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
VMF
DRUG PRODUCT COMPOSITIONS
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
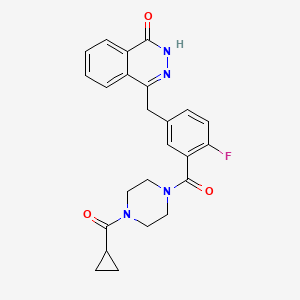

1. Azd 2281
2. Azd-2281
3. Azd221
4. Azd2281
5. Lynparza
1. 763113-22-0
2. Lynparza
3. Azd2281
4. Azd-2281
5. Ku-0059436
6. Azd 2281
7. 1-(cyclopropylcarbonyl)-4-[5-[(3,4-dihydro-4-oxo-1-phthalazinyl)methyl]-2-fluorobenzoyl]piperazine
8. Olaparib (azd-2281)
9. 4-(3-(4-(cyclopropanecarbonyl)piperazine-1-carbonyl)-4-fluorobenzyl)phthalazin-1(2h)-one
10. Ku-59436
11. Olaparib (azd2281, Ku-0059436)
12. 4-[[3-[4-(cyclopropanecarbonyl)piperazine-1-carbonyl]-4-fluorophenyl]methyl]-2h-phthalazin-1-one
13. Az2281
14. Mfcd13185161
15. Woh1jd9ar8
16. Nsc-747856
17. C24h23fn4o3
18. Chebi:83766
19. 4-(3-{[4-(cyclopropylcarbonyl)piperazin-1-yl]carbonyl}-4-fluorobenzyl)phthalazin-1(2h)-one
20. 4-[3-(4-cyclopropanecarbonyl-piperazine-1-carbonyl)-4-fluoro-benzyl]-2h-phthalazin-1-one
21. Az-2281
22. Keylynk-010 Component Olaparib
23. Ku59436
24. Olaparib Component Of Keylynk-010
25. Olaparib [inn]
26. Olaparib Cpd
27. Olaparib (azd2281; Ku-0059436)
28. Olaparib [usan:inn]
29. Unii-woh1jd9ar8
30. Olaparib (azd2281)
31. Acylpiperazine Analogue, 47
32. Olaparibum
33. Azd221
34. 4-[3-[4-(cyclopropanecarbonyl)piperazine-1-carbonyl]-4-fluorobenzyl]phthalazin-1(2h)-one
35. Olaparib- Bio-x
36. Lynparza (tn)
37. 09l
38. 4-((3-((4-(cyclopropylcarbonyl)piperazin-1-yl)carbonyl)-4-fluorophenyl)methyl)phthalazin-1(2h)-one
39. 4-((3-{(4-(cyclopropylcarbonyl)piperazin-1-yl)carbonyl}-4-fluorophenyl)methyl)phthalazin-1(2h)-one
40. 4-[(3-{[4-(cyclopropylcarbonyl)piperazin-1-yl]carbonyl}-4-fluorophenyl)methyl]phthalazin-1(2h)-one
41. Ku 59436
42. Olaparib [usan]
43. Olaparib [jan]
44. Ku0059436
45. Olaparib [mi]
46. Olaparib [vandf]
47. Olaparib - Azd2281
48. Olaparib [mart.]
49. Olaparib [who-dd]
50. Azd-2281 (olaparib)
51. Olaparib (jan/usan/inn)
52. Mls006010185
53. Schembl426568
54. Olaparib [orange Book]
55. Chembl521686
56. Gtpl7519
57. Bdbm27566
58. Dtxsid60917988
59. Ex-a002
60. Bcpp000360
61. Hms3295i09
62. Hms3426c03
63. Hms3654g13
64. Hms3746k07
65. Hms3870h03
66. Amy10295
67. Bcp01872
68. 763113-22-0, Lynparza,
69. Nsc747856
70. Nsc753686
71. S1060
72. Zinc40430143
73. Akos005145764
74. Ac-7939
75. Bcp9000363
76. Ccg-264799
77. Cs-0075
78. Db09074
79. Ex-7210
80. Nsc 747856
81. Nsc-753686
82. Sb14617
83. Ss-4573
84. Azd2281,olaparib, Ku-0059436
85. Ncgc00238451-01
86. Ncgc00238451-02
87. Ncgc00238451-08
88. Ncgc00238451-09
89. Ncgc00238451-11
90. 4-[(3-{[4-cyclopropylcarbonyl)piperazin-4-yl]carbonyl}-4-fluorophenyl)methyl]phtalazin-1(2h)-one
91. 4-[[3-[[4-(cyclopropylcarbonyl)-1-piperazinyl]carbonyl]-4-fluorophenyl]methyl]-1(2h)-phthalazinone
92. Bo164169
93. Hy-10162
94. Smr004701291
95. Sy040527
96. Olaparib(azd2281,kudosku-0059436)
97. A9666
98. Bb 0260909
99. Ft-0651458
100. Ku 0059436
101. Sw218142-2
102. Ec-000.2324
103. D09730
104. J-503540
105. Q7083106
106. Brd-k02113016-001-08-9
107. Brd-k02113016-001-09-7
108. 1-(cyclopropylcarbonyl)-4-[5-[(3,4-dihydro-4-oxo-1-phthalazine
109. 4-(3-(1-(cyclopropanecarbonyl)piperazine-4-carbonyl)-4-fluorobenzyl)phthalazin-1(2h)-one
110. (2h)-phthalazinone, 4-((3-((4-(cyclopropylcarbonyl)-1-piperazinyl)carbonyl)-4-fluorophenyl)methyl)-
111. 1(2h)-phthalazinone, 4-((3-((4-(cyclopropylcarbonyl)-1-piperazinyl)carbonyl)-4-fluorophenyl)methyl)-
112. 1021843-02-6
113. 4-({3-[(4-cyclopropanecarbonylpiperazin-1-yl)carbonyl]-4-fluorophenyl}methyl)-1,2-dihydrophthalazin-1-one
114. Piperazine, 1-(cyclopropylcarbonyl)-4-(5-((3,4-dihydro-4-oxo-1-phthalazinyl)methyl)-2-fluorobenzoyl)-
| Molecular Weight | 434.5 g/mol |
|---|---|
| Molecular Formula | C24H23FN4O3 |
| XLogP3 | 1.9 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Exact Mass | 434.17541877 g/mol |
| Monoisotopic Mass | 434.17541877 g/mol |
| Topological Polar Surface Area | 82.1 Ų |
| Heavy Atom Count | 32 |
| Formal Charge | 0 |
| Complexity | 790 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Olaparib is a poly (ADP-ribose) polymerase (PARP) inhibitor indicated for the treatment of: - Ovarian cancer, in which the medication is intended for [a] the maintenance treatment of adult patients with recurrent epithelial ovarian, fallopian tube or primary peritoneal cancer, who are in complete or partial response to platinum-based chemotherapy, or [b] for the treatment of adult patients with deleterious or suspected deleterious germline BRCA-mutated (gBRCAm) advanced ovarian cancer who have been treated with three or more prior lines of chemotherapy. Select patients for therapy based on an FDA-approved companion diagnostic for olaparib. - Breast cancer, in which the medication is intended for use in patients with deleterious or suspected deleterious gBRCAm, human epidermal growth factor receptor 2 (HER2)-negative metastatic breast cancer who have previously been treated with chemotherapy in the neoadjuvant, adjuvant or metastatic setting. Patients with hormone receptor (HR)-positive breast cancer should have been treated with a prior endocrine therapy or be considered inappropriate for endocrine treatment. Select patients for therapy based on an FDA-approved companion diagnostic for olaparib.
FDA Label
* Ovarian cancer :
Lynparza is indicated as monotherapy for the:
- maintenance treatment of adult patients with advanced (FIGO stages III and IV) BRCA1/2-mutated (germline and/or somatic) high-grade epithelial ovarian, fallopian tube or primary peritoneal cancer who are in response (complete or partial) following completion of first-line platinum-based chemotherapy.
- maintenance treatment of adult patients with platinum sensitive relapsed high grade epithelial ovarian, fallopian tube, or primary peritoneal cancer who are in response (complete or partial) to platinum based chemotherapy.
Lynparza in combination with bevacizumab is indicated for the:
- maintenance treatment of adult patients with advanced (FIGO stages III and IV) high-grade epithelial ovarian, fallopian tube or primary peritoneal cancer who are in response (complete or partial) following completion of first-line platinum-based chemotherapy in combination with bevacizumab and whose cancer is associated with homologous recombination deficiency (HRD) positive status defined by either a BRCA1/2 mutation and/or genomic instability (see section 5. 1).
* Breast cancer :
Lynparza is indicated as monotherapy for the treatment of adult patients with germline BRCA1/2-mutations, who have HER2 negative locally advanced or metastatic breast cancer . Patients should have previously been treated with an anthracycline and a taxane in the (neo)adjuvant or metastatic setting unless patients were not suitable for these treatments (see section 5. 1).
Patients with hormone receptor (HR)-positive breast cancer should also have progressed on or after prior endocrine therapy, or be considered unsuitable for endocrine therapy.
* Adenocarcinoma of the pancreas:
Lynparza is indicated as monotherapy for the maintenance treatment of adult patients with germline BRCA1/2-mutations who have metastatic adenocarcinoma of the pancreas and have not progressed after a minimum of 16 weeks of platinum treatment within a first-line chemotherapy regimen.
* Prostate cancer :
Lynparza is indicated as monotherapy for the treatment of adult patients with metastatic castration-resistant prostate cancer and BRCA1/2-mutations (germline and/or somatic) who have progressed following prior therapy that included a new hormonal agent.
Lynparza is indicated as monotherapy for the maintenance treatment of adult patients with platinum sensitive relapsed BRCA mutated (germline and/or somatic) high grade serous epithelial ovarian, fallopian tube, or primary peritoneal cancer who are in response (complete response or partial response) to platinum based chemotherapy.
The effect of olaparib on cardiac repolarization was assessed in 119 patients following a single dose of 300 mg and in 109 patients following multiple dosing of 300 mg twice daily. No clinically relevant effect of olaparib on QT interval was observed.
Antineoplastic Agents
Substances that inhibit or prevent the proliferation of NEOPLASMS. (See all compounds classified as Antineoplastic Agents.)
Poly(ADP-ribose) Polymerase Inhibitors
Chemicals and drugs that inhibit the action of POLY(ADP-RIBOSE)POLYMERASES. (See all compounds classified as Poly(ADP-ribose) Polymerase Inhibitors.)
L01XK01
L - Antineoplastic and immunomodulating agents
L01 - Antineoplastic agents
L01X - Other antineoplastic agents
L01XK - Poly (adp-ribose) polymerase (parp) inhibitors
L01XK01 - Olaparib
Absorption
Following oral administration, the absorption of olaparib is very rapid and can reach a peak concentration ranging between 4.7 and 9.1 mcg/ml after 1-3 hours. The reported AUC of olaparib after a dose of 200 mg is of 25.8 mcg.h/L and this AUC can be increased by 26% with constant administration. The consumption of a high-fat diet with olaparib can only decrease the tmax but do not have an effect in the peak concentration.
Route of Elimination
From the administered dose, approximately 86% of the administered dose is recovered after 7 days from which 44% is found in the urine and 42% is obtained in feces.
Volume of Distribution
After administration of a dose of 100 mg/kg, the reported volume of distribution was of 40.3 L.
Clearance
The total clearance of olaparib was reported to be 4.6 L/h.
Olaparib is extensively metabolized in the liver by the action of CYP3A isoenzymes. From the administered dose, the unchanged form of olaparib accounted for 70% of the circulating dose and it was considered the major component in urine and feces. The metabolic pathway of olaparib is mainly attributable to oxidation reactions with subsequent glucuronide and sulfate conjugation. However, the over 20 metabolites found in plasma, urine, and feces represented a minor portion of the administered dose. The major circulating metabolites were represented by the mono-oxygenated form and the piperazin-3-ol form.
The reported elimination half-life ranges between 5 to 11 hours.
Olaparib is an inhibitor of poly (ADP-ribose) polymerase (PARP) enzymes, including PARP1, PARP2, and PARP3. PARP enzymes are involved in normal cellular homeostasis, such as DNA transcription, cell cycle regulation, and DNA repair. Olaparib has been shown to inhibit growth of select tumor cell lines in vitro and decrease tumor growth in mouse xenograft models of human cancer both as monotherapy or following platinum-based chemotherapy. Increased cytotoxicity and anti-tumor activity following treatment with olaparib were noted in cell lines and mouse tumor models with deficiencies in BRCA. In vitro studies have shown that olaparib-induced cytotoxicity may involve inhibition of PARP enzymatic activity and increased formation of PARP-DNA complex, resulting in disruption of cellular homeostasis and cell death.
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
62
PharmaCompass offers a list of Olaparib API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Olaparib manufacturer or Olaparib supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Olaparib manufacturer or Olaparib supplier.
A Olaparib manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Olaparib, including repackagers and relabelers. The FDA regulates Olaparib manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Olaparib API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Olaparib manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Olaparib supplier is an individual or a company that provides Olaparib active pharmaceutical ingredient (API) or Olaparib finished formulations upon request. The Olaparib suppliers may include Olaparib API manufacturers, exporters, distributors and traders.
click here to find a list of Olaparib suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Olaparib DMF (Drug Master File) is a document detailing the whole manufacturing process of Olaparib active pharmaceutical ingredient (API) in detail. Different forms of Olaparib DMFs exist exist since differing nations have different regulations, such as Olaparib USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Olaparib DMF submitted to regulatory agencies in the US is known as a USDMF. Olaparib USDMF includes data on Olaparib's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Olaparib USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Olaparib suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Olaparib Drug Master File in Korea (Olaparib KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Olaparib. The MFDS reviews the Olaparib KDMF as part of the drug registration process and uses the information provided in the Olaparib KDMF to evaluate the safety and efficacy of the drug.
After submitting a Olaparib KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Olaparib API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Olaparib suppliers with KDMF on PharmaCompass.
A Olaparib written confirmation (Olaparib WC) is an official document issued by a regulatory agency to a Olaparib manufacturer, verifying that the manufacturing facility of a Olaparib active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Olaparib APIs or Olaparib finished pharmaceutical products to another nation, regulatory agencies frequently require a Olaparib WC (written confirmation) as part of the regulatory process.
click here to find a list of Olaparib suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Olaparib as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Olaparib API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Olaparib as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Olaparib and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Olaparib NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Olaparib suppliers with NDC on PharmaCompass.
Olaparib Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Olaparib GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Olaparib GMP manufacturer or Olaparib GMP API supplier for your needs.
A Olaparib CoA (Certificate of Analysis) is a formal document that attests to Olaparib's compliance with Olaparib specifications and serves as a tool for batch-level quality control.
Olaparib CoA mostly includes findings from lab analyses of a specific batch. For each Olaparib CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Olaparib may be tested according to a variety of international standards, such as European Pharmacopoeia (Olaparib EP), Olaparib JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Olaparib USP).

