
Synopsis
0
CEP/COS
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
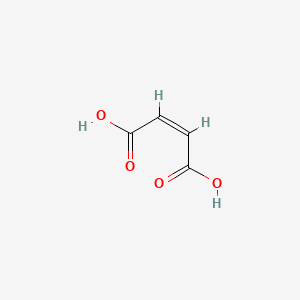

1. Hydrogen Maleate
2. Maleate
3. Maleic Acid, Ammonium Salt
4. Maleic Acid, Calcium Salt
5. Maleic Acid, Dipotassium Salt
6. Maleic Acid, Disodium Salt
7. Maleic Acid, Iron Salt
8. Maleic Acid, Monoammonium Salt
9. Maleic Acid, Monocopper (2+) Salt
10. Maleic Acid, Monosodium Salt
11. Maleic Acid, Neodymium Salt
12. Maleic Acid, Potassium Salt
13. Maleic Acid, Sodium Salt
14. Sodium Maleate
1. 110-16-7
2. Cis-butenedioic Acid
3. Toxilic Acid
4. Maleinic Acid
5. Malenic Acid
6. (2z)-but-2-enedioic Acid
7. 2-butenedioic Acid (2z)-
8. (z)-butenedioic Acid
9. 2-butenedioic Acid (z)-
10. Maleate
11. Cis-1,2-ethylenedicarboxylic Acid
12. Kyselina Maleinova
13. (z)-2-butenedioic Acid
14. Butenedioic Acid, (z)-
15. 2-butenedioic Acid, (z)-
16. 2-butenedioic Acid
17. 1,2-ethylenedicarboxylic Acid, (z)
18. Ccris 1115
19. Hsdb 666
20. Cis-2-butenedioic Acid
21. Malezid Cm
22. H2male
23. (z)-but-2-enedioic Acid
24. Ai3-01002
25. Cis-but-2-enedioic Acid
26. Mfcd00063177
27. Scotchbond Multipurpose Etchant
28. Chebi:18300
29. 91xw058u2c
30. Nsc-25940
31. 2-butenedioic Acid (2z)-, Monocastor Oil Alkyl Esters
32. Dsstox_cid_1517
33. (z)-2-butenedioate
34. Dsstox_rid_76194
35. Dsstox_gsid_21517
36. 68307-91-5
37. Mae
38. (2z)-but-2-enedioate
39. Kyselina Maleinova [czech]
40. Cas-110-16-7
41. Maleic Acid [nf]
42. Einecs 203-742-5
43. Na2215
44. Nsc 25940
45. (2z)-2-butenedioic Acid
46. Brn 0605762
47. Maleic-acid
48. Unii-91xw058u2c
49. Cis-butenedioate
50. 2-butenedioate
51. (z)-butenedioate
52. 1ahy
53. 1tok
54. Ncgc00091192-02
55. Cis-2-butenedioate
56. Bis-hydrogen Maleate
57. Maleic Acid [na2215] [corrosive]
58. Cis-but-2-enedioate
59. Maleic Acid, 99%
60. Maleic Acid (8ci)
61. Maleic Anhydride, Mono(castor Oil) Ester
62. (2z)-2-butenedioate
63. Butenedioic Acid,(z)-
64. (2z)-butene-2-dioate
65. Maleic Acid [ii]
66. Maleic Acid [mi]
67. Schembl613
68. Bmse000212
69. Ec 203-742-5
70. Maleic Acid [na2215]
71. Wln: Qv1u1vq-c
72. Maleic Acid [hsdb]
73. Maleic Acid [inci]
74. (2z)-butene-2-dioic Acid
75. Maleic Acid [mart.]
76. 4-02-00-02199 (beilstein Handbook Reference)
77. Mls002153468
78. 2-butenedioic Acid, (2z)-
79. Maleic Acid [usp-rs]
80. Maleic Acid [who-dd]
81. (2z)-2-butenedioic Acid #
82. Chembl539648
83. Gtpl9630
84. Dtxsid8021517
85. 1-(o-tolyl)piperazinehydrochloride
86. Hms2236f09
87. Maleic Acid, For Synthesis, 99%
88. Maleic Acid [ep Monograph]
89. Maleic Acid [usp Impurity]
90. Hy-y0367
91. Nsc25940
92. Str03481
93. Tox21_113105
94. Tox21_202193
95. Tox21_303045
96. 1,2-ethylenedicarboxylic Acid, Cis-
97. 2-butenedioic Acid (2z)- (9ci)
98. S3086
99. Zinc12358683
100. Akos000268822
101. Tox21_113105_1
102. Db04299
103. Maleic Acid [na2215] [corrosive]
104. Ncgc00090970-01
105. Ncgc00090970-02
106. Ncgc00090970-03
107. Ncgc00257215-01
108. Ncgc00259742-01
109. Maleic Acid, Tested According To Ph.eur.
110. Smr001230824
111. Maleic Acid 100 Microg/ml In Acetonitrile
112. Malic Acid Impurity B [ep Impurity]
113. Cs-0015079
114. M0006
115. Maleic Acid, Saj Special Grade, >=99.0%
116. Maleic Acid, Vetec(tm) Reagent Grade, 98%
117. 1, (z)
118. C01384
119. Q42038
120. A802156
121. Maleic Acid, Reagentplus(r), >=99.0% (hplc)
122. J-521667
123. J-610085
124. Sodium Aurothiomalate Impurity A [ep Impurity]
125. F2191-0240
126. 3b685b85-c1f3-4e32-9dbc-3854162a8de1
127. Maleic Acid, European Pharmacopoeia (ep) Reference Standard
128. Maleic Acid, Standard For Quantitative Nmr, Tracecert(r)
129. Maleic Acid, United States Pharmacopeia (usp) Reference Standard
130. Maleic Acid, Pharmaceutical Secondary Standard; Certified Reference Material
| Molecular Weight | 116.07 g/mol |
|---|---|
| Molecular Formula | C4H4O4 |
| XLogP3 | -0.3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 2 |
| Exact Mass | 116.01095860 g/mol |
| Monoisotopic Mass | 116.01095860 g/mol |
| Topological Polar Surface Area | 74.6 Ų |
| Heavy Atom Count | 8 |
| Formal Charge | 0 |
| Complexity | 119 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Enzyme Inhibitors
Compounds or agents that combine with an enzyme in such a manner as to prevent the normal substrate-enzyme combination and the catalytic reaction. (See all compounds classified as Enzyme Inhibitors.)
Kidney damage has been observed as a systemic toxic action of maleic acid. Morphological-functional changes in the kidneys were produced by intraperitoneal injection (100-350 mg/kg; rats, dogs) and also by inhalation exposure to ca. 720 mg/mc (rats). The damage to distal and proximal tubular resorption produced by maleic acid is reminiscent of the known (congenital or acquired) Fanconi syndrome in humans. This damage seems to be caused by the interaction of maleic acid with glutathione in tubular renal cells, leading to intolerable concentration of free radicals and peroxides. In addition to the resultant cellular damage, maleic acid appears to affect Na+ and H+ ion transport in the proximal tubes [RE5]. After chronic exposure of male rats (250, 500, 750 mg/kg /per/ day for up to two years), an increased mortality as well as kidney damage and delayed growth was observed in all dosage groups. Liver and testicular damage has also been found in highest dosage group. No signs of carcinogenic activity of maleic acid were discovered with long-term dosage, which however was not designed as a carcinogenicity study.
European Commission/European Chemical Substances Information System (ESIS); IUCLID Dataset, Maleic acid (CAS 110-16-7) p. 31 (2000). Available from, as of February 19, 2015: https://esis.jrc.ec.europa.eu/
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
79
PharmaCompass offers a list of Maleic Acid API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Maleic Acid manufacturer or Maleic Acid supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Maleic Acid manufacturer or Maleic Acid supplier.
A Maleic Acid manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Maleic Acid, including repackagers and relabelers. The FDA regulates Maleic Acid manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Maleic Acid API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Maleic Acid manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Maleic Acid supplier is an individual or a company that provides Maleic Acid active pharmaceutical ingredient (API) or Maleic Acid finished formulations upon request. The Maleic Acid suppliers may include Maleic Acid API manufacturers, exporters, distributors and traders.
click here to find a list of Maleic Acid suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Maleic Acid DMF (Drug Master File) is a document detailing the whole manufacturing process of Maleic Acid active pharmaceutical ingredient (API) in detail. Different forms of Maleic Acid DMFs exist exist since differing nations have different regulations, such as Maleic Acid USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Maleic Acid DMF submitted to regulatory agencies in the US is known as a USDMF. Maleic Acid USDMF includes data on Maleic Acid's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Maleic Acid USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Maleic Acid suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Maleic Acid Drug Master File in Japan (Maleic Acid JDMF) empowers Maleic Acid API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Maleic Acid JDMF during the approval evaluation for pharmaceutical products. At the time of Maleic Acid JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Maleic Acid suppliers with JDMF on PharmaCompass.
Maleic Acid Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Maleic Acid GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Maleic Acid GMP manufacturer or Maleic Acid GMP API supplier for your needs.
A Maleic Acid CoA (Certificate of Analysis) is a formal document that attests to Maleic Acid's compliance with Maleic Acid specifications and serves as a tool for batch-level quality control.
Maleic Acid CoA mostly includes findings from lab analyses of a specific batch. For each Maleic Acid CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Maleic Acid may be tested according to a variety of international standards, such as European Pharmacopoeia (Maleic Acid EP), Maleic Acid JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Maleic Acid USP).

