
Synopsis
Synopsis
0
EU WC
0
KDMF
0
VMF
0
FDA Orange Book
0
Australia
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
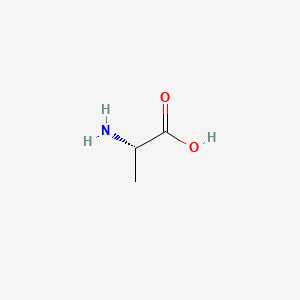

1. Abufne
2. Alanine, L Isomer
3. Alanine, L-isomer
4. L Alanine
5. L-alanine
6. L-isomer Alanine
1. L-alanine
2. 56-41-7
3. (s)-alanine
4. H-ala-oh
5. L-alpha-alanine
6. (s)-2-aminopropanoic Acid
7. L-2-aminopropionic Acid
8. (2s)-2-aminopropanoic Acid
9. L-(+)-alanine
10. L-2-aminopropanoic Acid
11. 2-aminopropionic Acid
12. Alpha-alanine
13. L-alpha-aminopropionic Acid
14. Alpha-aminopropionic Acid
15. L-2-aminopropionsaeure
16. Alaninum [latin]
17. (s)-(+)-alanine
18. L-s-aminopropionic Acid
19. (l)-alanine
20. Alanine (van)
21. (s)-2-aminopropionsaeure
22. Poly-l-alanine
23. 2-aminopropanoic Acid, L-
24. (s)-alpha-aminopropionsaeure
25. Alanine [usan:inn]
26. Alanina [dcit,spanish]
27. Alanine, L-
28. L-alanine, Homopolymer
29. L-a-aminopropionic Acid
30. L-alanin
31. (s)-2-aminopropionic Acid
32. Propanoic Acid, 2-amino-, (s)-
33. 25191-17-7
34. Hsdb 1801
35. Nsc 206315
36. L-alanine Homopolymer
37. Mfcd00064410
38. Of5p57n2zx
39. Chembl279597
40. Chebi:16977
41. Alanine (l-alanine)
42. Polyalanine
43. Alaninum
44. A-alanine
45. Poly-dl-alanine
46. L-a-alanine
47. 14c-l-alanine
48. A-aminopropionic Acid
49. (2s)-2-aminopropanoate
50. (c14)l-alanine
51. .alpha.-alanine
52. 18875-37-1
53. L-.alpha.-alanine
54. L-&alpha-alanine
55. Alanine (usp)
56. L-alanine (9ci)
57. 2-ammoniopropanoate
58. Propanoic Acid, 2-amino-
59. .alpha.-aminopropionic Acid
60. L-alanine, N-coco Alkyl Derivs.
61. L-alanine, Labeled With Carbon-14
62. Einecs 200-273-8
63. L-.alpha.-aminopropionic Acid
64. Unii-of5p57n2zx
65. Ritalanine
66. Racemic Alanine
67. A-aminopropionate
68. L-alanine, Labeled With Tritium
69. 3h-l-alanine
70. L-alanine Powder
71. Propanoic Acid, 2-amino-, (s)
72. [3h]alanine
73. L-a-aminopropionate
74. L-ala
75. [14c]alanine
76. [3h]-alanine
77. Alpha-aminopropanoate
78. Alpha-aminopropionate
79. L-2-aminopropanoate
80. L-2-aminopropionate
81. L-alanine,(s)
82. [14c]-alanine
83. L-alpha-aminopropionate
84. Tocris-0205
85. 2-ammoniopropanoic Acid
86. Ala-oh
87. Starbld0003382
88. L-alanine (jp17)
89. (s)-2-aminopropanoate
90. Alanine [vandf]
91. Alanine [hsdb]
92. Alanine [inci]
93. Alanine [usan]
94. Alanine [inn]
95. Alpha-aminopropanoic Acid
96. Alanine [ii]
97. Alanine [mi]
98. L-ala-2
99. L-alanine [fcc]
100. L-alanine [jan]
101. (s)-2-amino-propanoate
102. Alanine [who-dd]
103. L-alanine, >=99%
104. Bmse000028
105. Bmse000994
106. Ec 200-273-8
107. L-ch3ch(nh2)cooh
108. L-alanine [usp-rs]
109. Gtpl720
110. (s)-2-amino-propanoic Acid
111. 26336-61-8
112. (2s)-2-azanylpropanoic Acid
113. Alanine [ep Monograph]
114. Alanine, L- (7ci,8ci)
115. Gtpl4542
116. Gtpl4543
117. Alanine [usp Monograph]
118. Dtxsid20873899
119. L-alanine, >=98% (tlc)
120. L-alanine, 99%, Natural, Fg
121. F4f207ff-8ff8-4789-99a1-147ae0a36673
122. Hy-n0229
123. Str01663
124. Zinc4658553
125. L-alanine, >=99.0% (nt)
126. Ac-014
127. Bdbm50000099
128. S5631
129. (s)-2-aminopropanoic Acid;h-ala-oh
130. Akos010367904
131. Akos015840030
132. Ccg-266017
133. Cs-w020002
134. Db00160
135. L-alanine, Purum, >=98.0% (nt)
136. Serine Impurity A [ep Impurity]
137. Valine Impurity A [ep Impurity]
138. Ncgc00024494-01
139. 130380-93-7
140. 6898-94-8
141. Ac-24031
142. Bp-13281
143. L-alanine, Bioultra, >=99.5% (nt)
144. Db-029940
145. L-alanine, Saj Special Grade, >=99.0%
146. A0179
147. Am20100374
148. A15652
149. C00041
150. D00012
151. D84362
152. Lysine Acetate Impurity C [ep Impurity]
153. L-alanine, Cell Culture Reagent (h-l-ala-oh)
154. 064a410
155. A803529
156. Q218642
157. Sr-01000597687
158. J-015860
159. Q-201274
160. Sr-01000597687-3
161. L-alanine, Certified Reference Material, Tracecert(r)
162. Alanine, European Pharmacopoeia (ep) Reference Standard
163. F0001-2354
164. Z940713326
165. L-alanine, United States Pharmacopeia (usp) Reference Standard
166. L-alanine, Pharmaceutical Secondary Standard; Certified Reference Material
167. 115967-49-2
168. 77160-91-9
169. L-alanine, From Non-animal Source, Meets Ep, Usp Testing Specifications, Suitable For Cell Culture, >=98.5%
170. L-alanine, Pharmagrade, Ajinomoto, Ep, Jp, Usp, Manufactured Under Appropriate Gmp Controls For Pharma Or Biopharmaceutical Production, Suitable For Cell Culture
| Molecular Weight | 89.09 g/mol |
|---|---|
| Molecular Formula | C3H7NO2 |
| XLogP3 | -3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 1 |
| Exact Mass | 89.047678466 g/mol |
| Monoisotopic Mass | 89.047678466 g/mol |
| Topological Polar Surface Area | 63.3 Ų |
| Heavy Atom Count | 6 |
| Formal Charge | 0 |
| Complexity | 61.8 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Exptl Ther: ... Therapy of acute water diarrhea is reported in 94 male patients, aged 6-59 yr, of whom 49 were given a standard dextrose (glucose) based formula, to a total dose of 105-1719 ml/kg, and 45 were given the same soln with added alanine 90 uM/l, to a total dose of 60-1035 ml/kg. All patients were also given iv injections of replacement soln and oral tetracycline. The median stool output/kg body weight during the initial 24 hr of treatment until diarrhea stopped was reduced in those given the soln containing alanine as compared to controls. Intake of oral rehydration soln was reduced in those given alanine. Two patients given alanine soln and 18 given the standard treatment required unscheduled rapid iv replacement soln to correct signs of dehydration during oral therapy.
Patra FC et al; Br Med J 298: 1353-6 (1989)
DIETARY SUPPLEMENT
Lewis, R.J., Sr (Ed.). Hawley's Condensed Chemical Dictionary. 12th ed. New York, NY: Van Nostrand Rheinhold Co., 1993, p. 29
Used in pharmaceutical preparations for injection or infusion; dietary supplement and flavor compounds in maillard reaction products; stimulant of glucagon secretion
Gerhartz, W. (exec ed.). Ullmann's Encyclopedia of Industrial Chemistry. 5th ed.Vol A1: Deerfield Beach, FL: VCH Publishers, 1985 to Present., p. VA2 80-4
Used for protein synthesis.
Is an important source of energy for muscle tissue, the brain and central nervous system; strengthens the immune system by producing antibodies; helps in the metabolism of sugars and organic acids.
L-Alanine is a non-essential amino acid that occurs in high levels in its free state in plasma. It is produced from pyruvate by transamination. It is involved in sugar and acid metabolism, increases immunity, and provides energy for muscle tissue, brain, and the central nervous system. BCAAs are used as a source of energy for muscle cells. During prolonged exercise, BCAAs are released from skeletal muscles and their carbon backbones are used as fuel, while their nitrogen portion is used to form another amino acid, Alanine. Alanine is then converted to Glucose by the liver. This form of energy production is called the Alanine-Glucose cycle, and it plays a major role in maintaining the body's blood sugar balance.
Related Excipient Companies

Excipients by Applications
Market Place

REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
72
PharmaCompass offers a list of L-Alanine API API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right L-Alanine API manufacturer or L-Alanine API supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred L-Alanine API manufacturer or L-Alanine API supplier.
A L-Alanine API manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of L-Alanine API, including repackagers and relabelers. The FDA regulates L-Alanine API manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. L-Alanine API API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of L-Alanine API manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A L-Alanine API supplier is an individual or a company that provides L-Alanine API active pharmaceutical ingredient (API) or L-Alanine API finished formulations upon request. The L-Alanine API suppliers may include L-Alanine API API manufacturers, exporters, distributors and traders.
click here to find a list of L-Alanine API suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A L-Alanine API DMF (Drug Master File) is a document detailing the whole manufacturing process of L-Alanine API active pharmaceutical ingredient (API) in detail. Different forms of L-Alanine API DMFs exist exist since differing nations have different regulations, such as L-Alanine API USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A L-Alanine API DMF submitted to regulatory agencies in the US is known as a USDMF. L-Alanine API USDMF includes data on L-Alanine API's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The L-Alanine API USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of L-Alanine API suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The L-Alanine API Drug Master File in Japan (L-Alanine API JDMF) empowers L-Alanine API API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the L-Alanine API JDMF during the approval evaluation for pharmaceutical products. At the time of L-Alanine API JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of L-Alanine API suppliers with JDMF on PharmaCompass.
A L-Alanine API CEP of the European Pharmacopoeia monograph is often referred to as a L-Alanine API Certificate of Suitability (COS). The purpose of a L-Alanine API CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of L-Alanine API EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of L-Alanine API to their clients by showing that a L-Alanine API CEP has been issued for it. The manufacturer submits a L-Alanine API CEP (COS) as part of the market authorization procedure, and it takes on the role of a L-Alanine API CEP holder for the record. Additionally, the data presented in the L-Alanine API CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the L-Alanine API DMF.
A L-Alanine API CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. L-Alanine API CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of L-Alanine API suppliers with CEP (COS) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing L-Alanine API as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for L-Alanine API API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture L-Alanine API as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain L-Alanine API and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a L-Alanine API NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of L-Alanine API suppliers with NDC on PharmaCompass.
L-Alanine API Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of L-Alanine API GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right L-Alanine API GMP manufacturer or L-Alanine API GMP API supplier for your needs.
A L-Alanine API CoA (Certificate of Analysis) is a formal document that attests to L-Alanine API's compliance with L-Alanine API specifications and serves as a tool for batch-level quality control.
L-Alanine API CoA mostly includes findings from lab analyses of a specific batch. For each L-Alanine API CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
L-Alanine API may be tested according to a variety of international standards, such as European Pharmacopoeia (L-Alanine API EP), L-Alanine API JP (Japanese Pharmacopeia) and the US Pharmacopoeia (L-Alanine API USP).

